Abstract
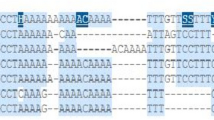
DNA methylation is an epigenetic event that occurs frequently in colorectal cancer (CRC). Increased glucose level is a strong risk factor for CRC. Protein phosphatase 1 regulatory subunit 3C (PPP1R3C) modulates glycogen metabolism, particularly glycogen synthesis. The aim of this study was to investigate the effect of high glucose levels on DNA methylation of PPP1R3C in CRC. PPP1R3C was significantly hypermethylated in CRC tissues (76/105, 72.38%, P < 0.05) and colon cancer cell lines (P < 0.05). CRC tissues obtained from patients with high glucose levels showed that the methylation of PPP1R3C was lower than in patients who had normal levels of glucose. When DLD-1 cells were cultured under conditions of high glucose, the methylation of PPP1R3C was repressed. The expression of PPP1R3C was inversely related to methylation status. In addition, a promoter luciferase assay showed that the transcriptional activity of PPP1R3C was increased in high glucose culture conditions. The number of cells decreased when PPP1R3C was silenced in DLD-1 cells. These results suggest that PPP1R3C, a novel hypermethylated gene in CRC, may play a critical role in cancer cell growth in association with glucose levels.






Similar content being viewed by others
References
Ahlquist D. A., Zou H., Domanico M., Mahoney D. W., Yab T. C., Taylor W. R. et al. 2012 Next-generation stool DNA test accurately detects colorectal cancer and large adenomas. Gastroenterology 142, 248–256.
Ali N. A., O’Brien Jr J. M., Blum W., Byrd J. C., Klisovic R. B., Marcucci G. et al. 2007 Hyperglycemia in patients with acute myeloid leukemia is associated with increased hospital mortality. Cancer 110, 96–102.
Annibaldi A. and Widmann C. 2010 Glucose metabolism in cancer cells. Curr. Opin. Clin. Nutr. Metab. Care 13, 466–470.
Berger B. M., Schroy 3rd P. C., Rosenberg J. L., Lai-Goldman M., Eisenberg M., Brown T. et al. 2006 Colorectal cancer screening using stool DNA analysis in clinical practice: early clinical experience with respect to patient acceptance and colonoscopic follow-up of abnormal tests. Clin. Colorectal Cancer 5, 338–343.
Center M. M., Jemal A. and Ward E. 2009 International trends in colorectal cancer incidence rates. Cancer Epidemiol. Biomarkers Prev. 18, 1688–1694.
Cheng A., Zhang M., Crosson S. M., Bao Z. Q. and Saltiel A. R. 2006 Regulation of the mouse protein targeting to glycogen (PTG) promoter by the FoxA2 forkhead protein and by 3\(^{\prime }\),5\(^{\prime }\)-cyclic adenosine 5\(^{\prime }\)-monophosphate in H4IIE hepatoma cells. Endocrinology 147, 3606–3612.
Doherty M. J., Young P. R. and Cohen P. T. 1996 Amino acid sequence of a novel protein phosphatase 1 binding protein (R5) which is related to the liver- and muscle-specific glycogen binding subunits of protein phosphatase 1. FEBS Lett. 399, 339–343.
Eschwège E. and Balkau B. 2001 Hyperglycaemia: link to excess mortality. Int. J. Clin. Pract. Suppl. 123, 3–6.
Gort E. H., Suijkerbuijk K. P., Roothaan S. M., Raman V., Vooijs M., van der Wall E. et al. 2008 Methylation of the TWIST1 promoter, TWIST1 mRNA levels, and immunohistochemical expression of TWIST1 in breast cancer. Cancer Epidemiol. Biomarkers Prev. 17, 3325–3330.
Grady W. M. and Carethers J. M. 2008 Genomic and epigenetic instability in colorectal cancer pathogenesis. Gastroenterology 135, 1079–1099.
Greenberg C. C., Meredith K. N., Yan L. and Brady M. J. 2003 Protein targeting to glycogen overexpression results in the specific enhancement of glycogen storage in 3T3-L1 adipocytes. J. Biol. Chem. 278, 30835–30842.
Greenberg C. C., Danos A. M. and Brady M. J. 2006 Central role for protein targeting to glycogen in the maintenance of cellular glycogen stores in 3T3-L1 adipocytes. Mol. Cell Biol. 26, 334–342.
Gupta C. and Tikoo K. 2013 High glucose and insulin differentially modulates proliferation in MCF-7 and MDA-MB-231 cells. J. Mol. Endocrinol. 51, 119–129.
Hamanaka R. B. and Chandel N. S. 2012 Targeting glucose metabolism for cancer therapy. J. Exp. Med. 209, 211–215.
Han L., Ma Q., Li J., Liu H., Li W., Ma G. et al. 2011 High glucose promotes pancreatic cancer cell proliferation via the induction of EGF expression and transactivation of EGFR. PLoS One 6, e27074.
Hoffmann M. J., Engers R., Florl A. R., Otte A. P., Muller M. and Schulz W. A. 2007 Expression changes in EZH2, but not in BMI-1, SIRT1, DNMT1 or DNMT3B are associated with DNA methylation changes in prostate cancer. Cancer Biol. Ther. 6, 1403–1412.
Jones P. A. and Baylin S. B. 2007 The epigenomics of cancer. Cell 128, 683–692.
Kel A. E., Niehof M., Matys V., Zemlin R. and Borlak J. 2008 Genome wide prediction of HNF4alpha functional binding sites by the use of local and global sequence context. Genome Biol. 9, R36.
Kim M. S., Lee J. and Sidransky D. 2010 DNA methylation markers in colorectal cancer. Cancer Metastasis Rev. 29, 181–206.
Kitajima Y. and Miyazaki K. 2013 The critical impact of HIF-1a on gastric cancer biology. Cancers (Basel) 5, 15–26.
Liang H., Zhong Y., Huang Y. and Chen G. 2012 Type 1 receptor parathyroid hormone (PTH1R) influences breast cancer cell proliferation and apoptosis induced by high levels of glucose. Med. Oncol. 29, 439–445.
Masur K., Vetter C., Hinz A., Tomas N., Henrich H., Niggemann B. et al. 2011 Diabetogenic glucose and insulin concentrations modulate transcriptome and protein levels involved in tumour cell migration, adhesion and proliferation. Br. J. Cancer 104, 345–352.
Mohindra V., Tripathi R. K., Singh R. K. and Lal K. K 2013 Molecular characterization and expression analysis of PPP1R3C in hypoxia-tolerant Indian catfish, Clarias batrachus (Linnaeus, 1758) under hypoxia. Gene 530, 127– 133.
Okada T., Suehiro Y., Ueno K., Mitomori S., Kaneko S., Nishioka M., et al. 2010 TWIST1 hypermethylation is observed frequently in colorectal tumours and its overexpression is associated with unfavorable outcomes in patients with colorectal cancer. Genes Chromosomes Cancer 49, 452–462.
Oster B., Thorsen K., Lamy P., Wojdacz T. K., Hansen L. L., Birkenkamp-Demtröder K. et al. 2011 Identification and validation of highly frequent CpG island hypermethylation incolorectal adenomas and carcinomas. Int. J. Cancer 129, 2855– 2866.
Park H. C., Shin A., Kim B. W., Jung K. W., Won Y. J., Oh J. H. et al. 2013 Data on the characteristics and the survival of korean patients with colorectal cancer from the Korea central cancer registry. Ann. Coloproctol. 29, 144–149.
Pescador N., Villar D., Cifuentes D., Garcia-Rocha M., Ortiz-Barahona A., Vazquez S. et al. 2010 Hypoxia promote glycogen accumulation through hypoxia inducible factor (HIF)-mediated induction of glycogen synthax 1. PLoS One 12, e9644.
Shaw R. J. 2006 Glucose metabolism and cancer. Curr. Opin. Cell Biol. 18, 598–608.
Shen G. M., Zhang F. L., Liu X. L. and Zhang J. W. 2010a Hypoxia-inducible factor 1-mediated regulation of PPP1R3C promotes glycogen accumulation in human MCF-7 cells under hypoxia. FEBS Lett. 584, 4366–4372.
Shen G. M., Zhang F. L., Liu X. L. and Zhang J. W. 2010b Hypoxia promotes glycogen accumulation through hypoxia inducible factor (HIF)-mediated induction of glycogen synthase 1. PLoS One 5, e9644.
Vernia S., Solaz-Fuster M. C., Gimeno-Alcañiz J. V., Rubio T., García-Haro L., Foretz M. et al. 2009 AMP-activated protein kinase phosphorylates R5/PTG, the glycogen targeting subunit of the R5/PTG-protein phosphatase 1 holoenzyme, and accelerates its down-regulation by the laforin-malin complex. J. Biol. Chem. 284, 8247–8255.
Warfel N. A., Dolloff N. G., Dicker D. T., Malysz J. and El-Deiry W. S. 2013 CDK1 stabilizes HIF-1alpha via direct phosphorylation of Ser668 to promote tumour growth. Cell Cycle 12, 3689–3701.
Zhang W., Wu Y., Yan Q., Ma F., Shi X., Zhao Y. et al. 2014 Deferoxamine enhances cell migration and invasion through promotion of HIF-1alpha expression and epithelial-mesenchymal transition in colorectal cancer. Oncol. Rep. 31, 111–116.
Zhao Y., Butler E. B. and Tan M. 2013 Targeting cellular metabolism to improve cancer therapeutics. Cell Death Dis. 4, e532.
Acknowledgements
This study was supported by the Basic Science Research Programme through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (grant number: 2010-0024117).
Author information
Authors and Affiliations
Corresponding author
Additional information
[Lee S. K., Moon J. W., Lee Y. W., Lee J. O., Kim S. J., Kim N., Kim J., Kim H. S. and Park S.-H. 2015 The effect of high glucose levels on the hypermethylation of protein phosphatase 1 regulatory subunit 3C (PPP1R3C) gene in colorectal cancer. J. Genet. 94, xx–xx]
Rights and permissions
About this article
Cite this article
LEE, S.K., MOON, J.W., LEE, Y.W. et al. The effect of high glucose levels on the hypermethylation of protein phosphatase 1 regulatory subunit 3C (PPP1R3C) gene in colorectal cancer. J Genet 94, 75–85 (2015). https://doi.org/10.1007/s12041-015-0492-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12041-015-0492-2