Abstract
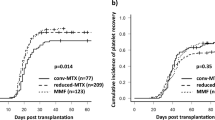
Umbilical cord blood transplantation with a reduced-intensity conditioning regimen (RIC-UCBT) is used increasingly in patients who have comorbid organ functions and lack human leukocyte antigen-identical donors. We compared the outcomes in 35 patients who received mycophenolate mofetil plus cyclosporine (MMF/CSP, n = 17) or MMF plus tacrolimus (MMF/TAC, n = 18) for graft-versus-host disease (GVHD) prophylaxis after RIC-UCBT. Cumulative incidence of neutrophil engraftment was 94 and 89 % in MMF/CSP and MMF/TAC groups, respectively (p = 0.34). The incidence of pre-engraftment immune reaction did not differ between the MMF/CSP (41 %) and MMF/TAC (39 %, p = 1.00) groups; however, patients in the MMF/TAC group tended to have a lower incidence of grade II–IV acute GVHD than those in MMF/CSP group (28 vs 53 %, p = 0.11). Overall survival (OS) at 1 year was 43 and 60 % in MMF/CSP and MMF/TAC groups, respectively (p = 0.39). Progression-free survival, non-relapse mortality, and relapse rate were comparable between the two groups (p = 0.76, 0.59, and 0.88, respectively). In multivariate analyses, MMF/TAC GVHD prophylaxis was closely associated with improved OS, but not with incidence of engraftment and acute GVHD. These results suggest that more intensive GVHD prophylaxis with MMF/TAC decreased acute GVHD without affecting other clinical outcomes, resulting in improved OS after RIC-UCBT.




Similar content being viewed by others
References
Brunstein CG, Wagner JE. Cord blood transplantation for adults. Vox Sang. 2006;91(3):195–205.
Slavin S, Nagler A, Naparstek E, Kapelushnik Y, Aker M, Cividalli G, et al. Nonmyeloablative stem cell transplantation and cell therapy as an alternative to conventional bone marrow transplantation with lethal cytoreduction for the treatment of malignant and nonmalignant hematologic diseases. Blood. 1998;91(3):756–63.
Giralt S, Thall PF, Khouri I, Wang X, Braunschweig I, Ippolitti C, et al. Melphalan and purine analog-containing preparative regimens: reduced-intensity conditioning for patients with hematologic malignancies undergoing allogeneic progenitor cell transplantation. Blood. 2001;97(3):631–7.
Goggins TF, Rizzieri DR. Nonmyeloablative allogeneic stem cell transplantation using alternative donors. Cancer Control. 2004;11(2):86–96.
Brunstein CG, Barker JN, Weisdorf DJ, DeFor TE, Miller JS, Blazar BR, et al. Umbilical cord blood transplantation after nonmyeloablative conditioning: impact on transplantation outcomes in 110 adults with hematologic disease. Blood. 2007;110(8):3064–70.
Uchida N, Wake A, Takagi S, Yamamoto H, Kato D, Matsuhashi Y, et al. Umbilical cord blood transplantation after reduced-intensity conditioning for elderly patients with hematologic diseases. Biol Blood Marrow Transplant. 2008;14(5):583–90.
Kishi Y, Kami M, Miyakoshi S, Kanda Y, Murashige N, Teshima T, et al. Early immune reaction after reduced-intensity cord-blood transplantation for adult patients. Transplantation. 2005;80(1):34–40.
Wang X, Liu H, Li L, Geng L, Ding K, Liu X, et al. Pre-engraftment syndrome after unrelated donor umbilical cord blood transplantation in patients with hematologic malignancies. Eur J Haematol. 2012;88(1):39–45.
Bolwell B, Sobecks R, Pohlman B, Andresen S, Rybicki L, Kuczkowski E, et al. A prospective randomized trial comparing cyclosporine and short course methotrexate with cyclosporine and mycophenolate mofetil for GVHD prophylaxis in myeloablative allogeneic bone marrow transplantation. Bone Marrow Transplant. 2004;34(7):621–5.
Pohlreich D, Vitek A, Maalouf J, Cetkovsky P. Decreased risk of acute gastrointestinal toxicity when substituting methotrexate with mycophenolate mofetil in the prevention of graft-versus-host disease in stem cell transplantation following myeloablative conditioning regimens. Bone Marrow Transplant. 2006;37(2):235–6 (author reply 6–7).
Cutler C, Li S, Kim HT, Laglenne P, Szeto KC, Hoffmeister L, et al. Mucositis after allogeneic hematopoietic stem cell transplantation: a cohort study of methotrexate- and non-methotrexate-containing graft-versus-host disease prophylaxis regimens. Biol Blood Marrow Transplant. 2005;11(5):383–8.
Uchida N, Wake A, Nakano N, Ishiwata K, Takagi S, Tsuji M, et al. Mycophenolate and tacrolimus for graft-versus-host disease prophylaxis for elderly after cord blood transplantation: a matched pair comparison with tacrolimus alone. Transplantation. 2011;92(3):366–71.
Kanda J, Kaynar L, Kanda Y, Prasad VK, Parikh SH, Lan L, et al. Pre-engraftment syndrome after myeloablative dual umbilical cord blood transplantation: risk factors and response to treatment. Bone Marrow Transplant. 2013;48(7):926–31.
Ratanatharathorn V, Nash RA, Przepiorka D, Devine SM, Klein JL, Weisdorf D, et al. Phase III study comparing methotrexate and tacrolimus (prograf, FK506) with methotrexate and cyclosporine for graft-versus-host disease prophylaxis after HLA-identical sibling bone marrow transplantation. Blood. 1998;92(7):2303–14.
Nash RA, Antin JH, Karanes C, Fay JW, Avalos BR, Yeager AM, et al. Phase 3 study comparing methotrexate and tacrolimus with methotrexate and cyclosporine for prophylaxis of acute graft-versus-host disease after marrow transplantation from unrelated donors. Blood. 2000;96(6):2062–8.
Hiraoka A, Ohashi Y, Okamoto S, Moriyama Y, Nagao T, Kodera Y, et al. Phase III study comparing tacrolimus (FK506) with cyclosporine for graft-versus-host disease prophylaxis after allogeneic bone marrow transplantation. Bone Marrow Transplant. 2001;28(2):181–5.
Yanada M, Emi N, Naoe T, Sakamaki H, Takahashi S, Hirabayashi N, et al. Tacrolimus instead of cyclosporine used for prophylaxis against graft-versus-host disease improves outcome after hematopoietic stem cell transplantation from unrelated donors, but not from HLA-identical sibling donors: a nationwide survey conducted in Japan. Bone Marrow Transplant. 2004;34(4):331–7.
Sakai R, Taguri M, Oshima K, Mori T, Ago H, Adachi S, et al. A comparison of tacrolimus and cyclosporine combined with methotrexate for graft-versus-host disease prophylaxis, stratified by stem cell source: a retrospective nationwide survey. Int J Hematol. 2016;103(3):322–33.
Przepiorka D, Weisdorf D, Martin P, Klingemann HG, Beatty P, Hows J, et al. 1994 Consensus conference on acute GVHD grading. Bone Marrow Transplant. 1995;15(6):825–8.
Miyakoshi S, Yuji K, Kami M, Kusumi E, Kishi Y, Kobayashi K, et al. Successful engraftment after reduced-intensity umbilical cord blood transplantation for adult patients with advanced hematological diseases. Clin Cancer Res. 2004;10(11):3586–92.
Kanda Y. Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transplant. 2013;48(3):452–8.
Long GD, Laughlin M, Madan B, Kurtzberg J, Gasparetto C, Morris A, et al. Unrelated umbilical cord blood transplantation in adult patients. Biol Blood Marrow Transplant. 2003;9(12):772–80.
Ballen KK. New trends in umbilical cord blood transplantation. Blood. 2005;105(10):3786–92.
Laughlin MJ, Barker J, Bambach B, Koc ON, Rizzieri DA, Wagner JE, et al. Hematopoietic engraftment and survival in adult recipients of umbilical-cord blood from unrelated donors. N Engl J Med. 2001;344(24):1815–22.
Bejanyan N, Rogosheske J, DeFor T, Lazaryan A, Esbaum K, Holtan S, et al. Higher dose of mycophenolate mofetil reduces acute graft-versus-host disease in reduced-intensity conditioning double umbilical cord blood transplantation. Biol Blood Marrow Transplant. 2015;21(5):926–33.
Miyakoshi S, Kusumi E, Matsumura T, Hori A, Murashige N, Hamaki T, et al. Invasive fungal infection following reduced-intensity cord blood transplantation for adult patients with hematologic diseases. Biol Blood Marrow Transplant. 2007;13(7):771–7.
Narimatsu H, Terakura S, Matsuo K, Oba T, Uchida T, Iida H, et al. Short-term methotrexate could reduce early immune reactions and improve outcomes in umbilical cord blood transplantation for adults. Bone Marrow Transplant. 2007;39(1):31–9.
Barker JN, Weisdorf DJ, DeFor TE, Blazar BR, Miller JS, Wagner JE. Rapid and complete donor chimerism in adult recipients of unrelated donor umbilical cord blood transplantation after reduced-intensity conditioning. Blood. 2003;102(5):1915–9.
Sanz J, Picardi A, Hernandez Boluda JC, Martin C, Ferra C, Nozzoli C, et al. Impact of graft-versus-host disease prophylaxis on outcomes after myeloablative single-unit umbilical cord blood transplantation. Biol Blood Marrow Transplant. 2013;19(9):1387–92.
Miyakoshi S, Kami M, Tanimoto T, Yamaguchi T, Narimatsu H, Kusumi E, et al. Tacrolimus as prophylaxis for acute graft-versus-host disease in reduced intensity cord blood transplantation for adult patients with advanced hematologic diseases. Transplantation. 2007;84(3):316–22.
Kanda Y, Kobayashi T, Mori T, Tanaka M, Nakaseko C, Yokota A, et al. A randomized controlled trial of cyclosporine and tacrolimus with strict control of blood concentrations after unrelated bone marrow transplantation. Bone Marrow Transplant. 2016;51(1):103–9.
Jacobson P, Rogosheske J, Barker JN, Green K, Ng J, Weisdorf D, et al. Relationship of mycophenolic acid exposure to clinical outcome after hematopoietic cell transplantation. Clin Pharmacol Ther. 2005;78(5):486–500.
Acknowledgments
We thank the medical and nursing staff working on the Fukuoka Blood and Marrow Transplantation Group for providing patients information. This work was supported, in part, by a Grant-in-Aid for Scientific Research (No. 16H05340 to T.M.) and Grant-in-Aid for Scientific Research on Innovative Areas “Stem Cell Aging and Disease” (No. 25115002 to T.M.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Miyamoto, T., Takashima, S., Kato, K. et al. Comparison of cyclosporine and tacrolimus combined with mycophenolate mofetil in prophylaxis for graft-versus-host disease after reduced-intensity umbilical cord blood transplantation. Int J Hematol 105, 92–99 (2017). https://doi.org/10.1007/s12185-016-2093-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-016-2093-0