Abstract
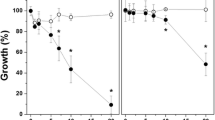
Due to the increased number of immunocompromised patients, the infections associated with the pathogen of the genus Candida and other fungi have increased dramatically. Photodynamic antimicrobial chemotherapy (PACT) has been presented as a potential antimicrobial therapy, in a process that combines light and a photosensitizing drug, which promotes a phototoxic response by the treated cells. In this work, we studied the effects of the different medium conditions during PACT, using either methylene blue (MB) or toluidine blue (TB) on Candida albicans. The inhibition of the growth produced by PACT was decreased for different pH values (6.0, 7.0, and 8.0) in a buffered medium. The phototoxic effects were observed only in the presence of saline (not buffered medium). PACT was modulated by calcium in a different manner using either MB or TB. Also when using MB both verapamil or sodium azide were able to decrease the phototoxic effects on the C. albicans. These results show that PACT is presented as a new and promising antifungal therapy, however, new studies are necessary to understand the mechanism by which this event occurs.
Similar content being viewed by others
References
Almeida, R.D., B.J. Manadas, A.P. Carvalho, and C.B. Duarte. 2004. Intracellular signaling mechanisms in photodynamic therapy. Biochim. Biophys. Acta. 1704, 59–86.
Barchiesi, F., A.L. Colombo, D.A. McGough, A.W. Fothergill, and M.G. Rinaldi. 1994. In vitro activity of itraconazole aginst fluconazole-susceptible and -resistant Candida albicans isolates from oral cavities from patients infected with the human immunodeficiency virus. Antimicrob. Agents Chemother. 38, 1530–1533.
Bennett, J.E., K. Izumikawa, and K.A. Marr. 2004. Mechanism of increased fluconazole resistance in Candida glabrata during prophylaxis. Antimicrob. Agents Chemother. 48, 1773–1777.
Bhatti, M., A. MacRobert, S. Meghji, B. Henderson, and M. Wilson. 1998. A study of the uptake of toluidine blue O by Porphyromonas gingivalis and the mechanism of lethal photosensitization. Photochem. Photobiol. 68, 370–376.
Bliss, J.M., C.E. Bigelow, T.H. Foster, and C.G. Haidaris. 2004. Susceptibility of Candida species to photodynamic effects of photofrin. Antimicrob. Agents Chemother. 48, 2000–2006.
Böhmer, R.M. and G. Morstyn. 1985. Uptake of hematoporphyrin derivative by normal and malignant cells: Effect of serum, pH, temperature, and cell size. Cancer Res. 45, 5328–5334.
Calderone, R.A. 2002. Candida and Candidiasis. Washington D.C. ASM Press, USA.
Cannon, R.D., E. Lamping, A.R. Holmes, K. Niimi, K. Tanabe, M. Niimi, and B.C. Monk. 2007. Candida albicans drug resistance — another way to cope with stress. Microbiology 153, 3211–3217.
Chabrier-Roselló, Y., T.H. Foster, N. Pérez-Nazario, S. Mitra, and C.G. Haidaris. 2005. Sensitivity of Candida albicans germ tubes and biofilms to photofrin-mediated phototoxicity. Antimicrob. Agents Chemother. 49, 4288–4295.
Cormick, M.P., M.G. Alvarez, M. Rovera, and E.N. Durantini. 2009. Photodynamic inactivation of Candida albicans sensitized by tri- and tetra-cationic porphyrin derivatives. Eur. J. Med. Chem. 44, 1592–1599.
Demidova, T.N. and M.R. Hamblin. 2005. Effect of cell-photo-sensitizer binding and cell density on microbial photoinactivation. Antimicrob. Agents Chemother. 49, 2329–2335.
Gad, F., T. Zahra, T. Hasan, and M.R. Hamblin. 2004. Effects of growth phase and extracellular slime on photodynamic inactivation of Gram-positive pathogenic bacteria. Antimicrob. Agents Chemother. 48, 2173–2178.
Gudlaugsson, O., S. Gillespie, K. Lee, J. Vande Berg, J. Hu, S. Messer, L. Herwaldt, M. Pfaller, and D. Diekema. 2003. Attributable mortality of nosocomial candidemia, revisited. Clin. Infect. Dis. 37, 1172–1177.
Hamblin, M.R., D.A. O’Donnell, N. Murthy, C.H. Contag, and T. Hasan. 2002. Rapid control of wound infections by targeted photodynamic therapy monitored by in vivo bioluminescence imaging. Photochem. Photobiol. 75, 51–57.
Hamblin, M.R., J.L. Miller, I. Rizvi, H.G. Loew, and T. Hasan. 2001. Pegylation of a chlorine6 polymer conjugate increases tumor targeting of photosensitizer. Cancer Res. 61, 7155–7162.
Horsburgh, C.R. and C.H. Kirkpatrick. 1983. Long-term therapy of chronic mucocutaneous candidiasis with ketoconazole: experience with twenty-one patients. Am. J. Med. 74, 23–29.
Jori, G. 2006. Photodynamic therapy of microbial infections: state of the art and perspectives. J. Environ. Pathol. Toxicol. Oncol. 25, 505–519.
Komerik, N., H. Nakanishi, A.J. MacRobert, B. Henderson, and P. Speight. 2003. In vivo killing of Porphyromonas gingivalis by toluidine blue-mediated photosensitization in an animal model. Antimicrob. Agents Chemother. 47, 932–940.
Lambrechts, S.A.G., M.G. Aalders, and J. Van Marle. 2005. Mechanistic study of the photodynamic inactivation of Candida albicans by a cationic porphyrin. Antimicrob. Agents Chemother. 49, 2026–2034.
Lambrechts, S.A., T.N. Demidova, M.C. Aalders, T. Hasan, and M.R. Hamblin. 2005. Photodynamic therapy for Staphylococcus aureus infected burn wounds in mice. Photochem. Photobiol. Sci. 4, 503–509.
MacDonald, I. and T.J. Dougherty. 2001. Basic principles of photodynamic therapy. J. Porphyrins Phthalocyanines 5, 105–129.
Morgan, J., M.I. Meltzer, B.D. Plikaytis, A.N. Sofair, S. Huie-White, S. Wilcox, L.H. Harrison, E.C. Seaberg, R.A. Hajjeh, and S.M. Teutsch. 2005. Excess mortality, hospital stay, and cost due to candidemia: a case-control study using data from population-based candidemia surveillance. Infect. Control Hosp. Epidemiol. 26, 540–547.
Munin, E., L.M. Giroldo, L.P. Alves, and M.S. Costa. 2007. Study of germ tube formation by Candida albicans after photodynamic antimicrobial chemotherapy (PACT). J. Photochem. Photobiol. 88, 16–20.
Peloi, L.S., R.R. Soares, C.E. Biondo, V.R. Souza, N. Hioka, and E. Kimura. 2008. Photodynamic effect of light-emitting diode light on cell growth inhibition induced by methylene blue. J. Biosci. 33, 231–237.
Pfaller, M.A., S.A. Messer, R.J. Hollis, R.N. Jones, and D.J. Diekema. 2002. In vitro activities of ravuconazole and voriconazole compared with those of four approved systemic antifungal agents against 6,970 clinical isolates of Candida spp. Antimicrob. Agents Chemother. 46, 1723–1727.
Posteraro, B., M. Tumbarello, M. La Sorda, T. Spanu, E.M. Trecarichi, F. De Bernardis, G. Scoppettuolo, M. Sanguinetti, and G. Fadda. 2006. Azole resistance of Candida glabrata in a case of recurrent fungemia. J. Clin. Microbiol. 44, 3046–3047.
Ruhnke, M., A. Schmidt-Westhausen, and J. Morschhäuser. 2000. Development of simultaneous resistance to fluconazole in Candida albicans and Candida dubliniensis in a patient with AIDS. J. Antimicrob. Chemother. 46, 291–295.
Sanguinetti, M., B. Posteraro, B. Fiori, S. Ranno, R. Torelli, and G. Fadda. 2005. Mechanisms of azole resistance in clinical isolates of Candida glabrata collected during a hospital survey of antifungal resistance. Antimicrob. Agents Chemother. 49, 668–679.
Sharma, M., A. Dube, H. Bansal, and P.K. Gupta. 2004. Effect of pH on uptake and photodynamic action of chlorin p6 on human colon and breast adenocarcinoma cell lines. Photochem. Photobiol. Sci. 3, 231–235.
Sharma, M., K. Sahu, A. Dube, and P.K. Gupta. 2005. Extracellular pH influences the mode of cell death in human colon adenocarcinoma cells subjected to photodynamic treatment with chlorin p6. J. Photochem. Photobiol. B 81, 107–113.
Smijs, T.G.M., J.A. Bouwstra, M. Talebi, and S. Pavel. 2007. Investigation of conditions involved in the susceptibility of the dermatophyte Trichophyton rubrum to photodynamic treatment. J. Antimicrob. Chemother. 60, 750–759.
So, C.W., P.W. Tsang, P.C. Lo, C.J. Seneviratne, L.P. Samaranayake, and W.P. Fong. 2009. Photodynamic inactivation of Candida albicans by BAM-SiPc. Mycoses. Feb 26. [in press].
Souza, S.C., J.C. Junqueira, I. Balducci, C.Y. Koga-Ito, E. Munin, and A.O.C. Jorge. 2006. Photosensitization of different Candida species by low power laser light. J. Photochem. Photibiol. 83, 34–38.
Tegos, G.P. and M.R. Hamblin. 2006. Phenothiazinium antimicrobial photosensitizers are substrates of bacterial multidrug resistance pumps. Antimicrob. Agents Chemother. 50, 196–203.
Uzdensky, A., A. Lobanov, M. Bibov, and Y. Petin. 2007. Involvement of Ca2+- and cyclic adenosine monophosphate-mediated signaling pathways in photodynamic injury of isolated crayfish neuron and satellite glial cells. J. Neurosci. Res. 85, 860–870.
Zheng, J.H., D. Shi, Y. Zhao, and Z.L. Chen. 2006. Role of calcium signal in apoptosis and protective mechanism of colon cancer cell line SW480 in response to 5-aminolevulinic acid-photodynamic therapy. Ai Zheng. 25, 683–688.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Carvalho, G.G., Felipe, M.P. & Costa, M.S. The photodynamic effect of methylene blue and toluidine blue on Candida albicans is dependent on medium conditions. J Microbiol. 47, 619–623 (2009). https://doi.org/10.1007/s12275-009-0059-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-009-0059-0