Abstract
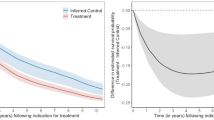
A dynamic treatment regimen is a rule or set of rules which define how a subject’s treatment at repeated visits depends on their evolving history of time-dependent covariates. In this manuscript we focus on regimens that are characterized by intermediate target ranges [b 1,b 2] which are used clinically to modify treatment such that longitudinal measures of the intermediate stay within a tolerance defined by [b 1,b 2]. For observational data, survival under a particular regimen can be consistently estimated by artificially censoring subjects when they become nonadherent to the regimen and then weighting subjects by the inverse probability of remaining uncensored. In many settings subjects are not identified at baseline as individuals who are following a pre-specified regimen. In addition, it may be possible for subjects to be adherent to multiple regimens at the same time. In order to compare alternative regimens we present a data augmentation methodology in which regimen membership is stochastically imputed multiple times and parameter estimates are aggregated to provide a final point estimate of a causal hazard ratio, and for which sandwich variance estimation methods provide consistent inference. The method is illustrated through simulation results as well as a preliminary analysis comparing epoetin therapy regimens with different target hemoglobin ranges in a cohort of hemodialysis subjects from the United States Renal Data System.
Similar content being viewed by others
References
Cain LE, Robins JM, Lanoy E, Logan R, Costagliola D, Hernán MA (2010) When to start treatment? A systematic approach to the comparison of dynamic regimes using observational data. Int J Biostat 6(2):18
Cole S, Hernán M (2004) Adjusted survival curves with inverse probability weights. Comput Methods Programs Biomed 75:45–49
Drüeke T, Locatelli F, Clyne N, Eckardt KU, Macdougall I, Tsakiris D, Burger HU, Scherhad A (2006) Normalization of hemoglobin level in patients with chronic kidney disease and anemia. N Engl J Med 355(20):2071–2084
Federal Drug Administration (2007) FDA strengthens safety information for erythropoiesis-stimulating agents (ESAs). URL http://www.fda.gov/bbs/topics/NEWS/2007/NEW01582.html. March 9, 2007
Hernán M, Brumback B, Robins J (2000) Marginal structural models to estimate the causal effect of zidovudine on the survival of HIV-positive men. Epidemiology 11(5):561–570
Hernán M, Brumback B, Robins J (2001) Marginal structural models to estimate the joint causal effect of nonrandomized treatments. J Am Stat Assoc 96(454):440–448
Hernán M, Lanoy E, Costagliola D, Robins J (2006) Comparison of dynamic treatment regimes via inverse probability weighting. Basic Clin Pharmacol Toxicol 98:237–242
Jones M, Ibels L, Schenkel B, Zagari M (2004) Impact of epoetin alfa on clinical end points in patients with chronic renal failure: a meta-analysis. Kidney Int 65:767–775
Li S, Collins A (2004) Association of hematocrit value with cardiovascular morbidity and mortality in incident hemodialysis patients. Kidney Int 65:626–633
Liang K, Zeger S (1986) Longitudinal data analysis using generalized linear models. Biometrika 73(1):13–22
Ofsthun N, Labrecque J, Lacson E, Keen M, Lazarus J (2003) The effects of higher hemoglobin levels on mortality and hospitalization in hemodialysis patients. Kidney Int 63:1908–1914
Orellana L, Rotnitzky A, Robins JM (2010) Dynamic regime marginal structural mean models for estimation of optimal dynamic treatment regimes. Part I: Main content. Int J Biostat 6(2):8
Pierce D (1982) The asymptotic effect of substituting estimators for parameters in certain types of statistics. Ann Stat 10(2):475–478
Robins J (1993) Information recovery and bias adjustment in proportional hazards regression analysis of randomized trials using surrogate markers. In: Proceedings of the biopharmaceutical section, American statistical association. American Statistical Association, Alexandria, pp 24–33
Robins J (1998) Marginal structural models. In: 1997 Proceedings of the section on Bayesian statistical science. American Statistical Association, Alexandria, pp 1–10
Robins J (1999) Marginal structural models versus structural nested models as tools for causal inference. In: Halloran ME, Berry D (eds) Statistical models in epidemiology: the environment and clinical trials. Springer, New York, pp 95–134
Robins J, Finkelstein D (2000) Correcting for noncompliance and dependent censoring in an aids clinical trial with inverse probability of censoring weighted (IPCW) log-rank tests. Biometrics 56(3):779–788
Robins JM, Hernán MA (2009) Estimation of the causal effects of time-varying exposures. In: Fitzmaurice G, Davidian M, Verbeke G, Molenberghs G (eds) Longitudinal data analysis. Chapman and Hall/CRC Press, New York, pp 553–599
Robins J, Hernán M, Brumback B (2000) Marginal structural models and causal inference in epidemiology. Epidemiology 11(5):550–560
Robins J, Orellana L, Rotnitzky A (2008) Estimation and extrapolation of optimal treatment and testing strategies. Stat Med 27(23):4678–4721
Robins J, Rotnitzky A (1992) Recovery of information and adjustment for dependent censoring using surrogate markers. In: Jewel N, Dietz K, Farewell V (eds) AIDS epidemiology—methodological issues, Proceedings of the American statistical association. American Statistical Association, Alexandria, p 2433
Robins J, Rotnitzky A, Zhao L (1995) Analysis of semiparametric regression models for repeated outcomes in the presence of missing data. J Am Stat Assoc 90:106–121
Rubin D, Schenker N (1986) Multiple imputation for interval estimation from simple random samples with ignorable nonresponse. J Am Stat Assoc 81(394):366–374
Singh A, Szczech L, Tang K, Barnhart H, Sapp S, Wolfson M, Reddan D (2006) Correction of anemia with epoetin alfa in chronic kidney disease. N Engl J Med 355(20):2085–2098
Zhang Y, Thamer M, Stefanik K, Kaufman J, Cotter D (2004) Epoetin requirements predict mortality in hemodialysis patients. Am J Kidney Dis 44(5):866–876
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cotton, C.A., Heagerty, P.J. A Data Augmentation Method for Estimating the Causal Effect of Adherence to Treatment Regimens Targeting Control of an Intermediate Measure. Stat Biosci 3, 28–44 (2011). https://doi.org/10.1007/s12561-011-9038-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12561-011-9038-1