Abstract
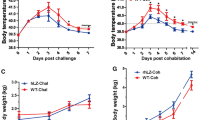
Probiotics form a promising strategy to maintain intestinal health. Milks fermented with probiotic strains, such as the Lactobacillus paracasei ST11, are largely commercialized in Brazil and form a low-cost alternative to probiotic pharmaceutical formulations. In this study, we assessed the probiotic effects of milk fermented by L. paracasei ST11 (administered through fermented milk) in a Salmonella typhimurium infection model in BALB/c mice. We observed in this murine model that the applied probiotic conferred protective effects against S. typhimurium infection, since its administration reduced mortality, weight loss, translocation to target organs (liver and spleen) and ileum injury. Moreover, a reduction in the mRNA expression of pro-inflammatory cytokines such as IFN-γ, IL-6, TNF-α and IL-17 in animals that received the probiotic before challenge was observed. Additionally, the ileum microbiota was better preserved in these animals. The present study highlights a multifactorial protective aspect of this commercial probiotic strain against a common gastrointestinal pathogen.





Similar content being viewed by others
References
Lawley TD, Bouley DM, Hoy YE, Lai YY, Gerke C, Relman DA, Monack DM (2008) Host transmission of Salmonella enterica serovar typhimurium is controlled by virulence factors and indigenous intestinal microbiota. Infect Immun 76:403–416. https://doi.org/10.1128/IAI.01189-07
Barman M, Unold D, Shifley K, Amir E, Hung K, Bos N, Salzman N (2008) Enteric salmonellosis disrupts the microbial ecology of the murine gastrointestinal tract. Infect Immun 76(3):907–915. https://doi.org/10.1128/IAI.01432-07
Crump JA, Sjölund-Karlsson M, Gordon MA, Parry CM (2015) Epidemiology, clinical presentation, laboratory diagnosis, antimicrobial resistance, and antimicrobial management of invasive Salmonella infections. Clin Microbiol Rev 28(4):901–937. https://doi.org/10.1128/CMR.00002-15
FAO/WHO (2002) Working group guidelines for the evaluation of probiotics in food. FAO/WHO (Food and Agriculture Organization/World Health Organization). WHO, London, pp 1–11
Santos RL (2014) Pathobiology of Salmonella, intestinal microbiota, and the host immune response. Front Immunol 5. https://doi.org/10.3389/fimmu.2014.00252
Neeser JR, Reniero R, Servin A (2004) Lactobacillus paracasei strain for preventing diarrhea caused by pathogenic bacteria. U.S. patent 6835376 B1, December 28, 2004
Govender M, Choonara YE, Kumar P, du Toit LC, van Vuuren S, Pillay V (2014) A review of the advancements in probiotic delivery: conventional vs. non-conventional formulations for intestinal flora supplementation. AAPS PharmSciTech 15(1):29–43. https://doi.org/10.1208/s12249-013-0027-1
Perrin Y, Nutten S, Audran R, Berger B, Biblioni R, Wassenberg J, Barbier N, Aubert V, Moulin J, Singh A, Magiola C, Mercenier A, Spertini F (2014) Comparison of two oral probiotic preparations in a randomized crossover trial highlights a potentially beneficial effect of Lactobacillus paracasei NCC2461 in patients with allergic rhinitis. Clin Transl Allergy 4(1):1. https://doi.org/10.1186/2045-7022-4-1
Nembrini C, Singh A, Castro CA, Mercenier A, Nutten S (2015) Oral administration of Lactobacillus paracasei NCC 2461 for the modulation of grass pollen allergic rhinitis: a randomized, placebo-controlled study during the pollen season. Clin Transl Allergy 5:41. https://doi.org/10.1186/s13601-015-0085-4
Benyacoub J, Bosco N, Blanchard C, Demont A, Philippe D, Castiel-Higounenc I, Guéniche A (2014) Immune modulation property of Lactobacillus paracasei NCC2461 (ST11) strain and impact on skin defences. Benefic Microbes 5:129–136. https://doi.org/10.3920/BM2013.0014
Reyagne P, Bastien P, Couavoux MP, Philippe D, Renouf M, Castiel-Higounenc I, Gueniche A (2017) The positive benefit of Lactobacillus paracasei NCC2461 ST11 in healthy volunteers with moderate to severe dandruff. Benefic Microbes 8:671–680. https://doi.org/10.3920/BM2016.0144
Mendonça AH, Cerqueira MMOP, Nicoli JR, Sousa SMM, Nardi RMD, Souza FN, Fonseca LM, Leite MO, Arantes RME (2014) A Lactobacillus rhamnosus strain induces protection in different sites after Salmonella enterica subsp. enterica serovar Typhimurium challenge in gnotobiotic and conventional mice. Arq Bras Med Vet Zootec 66:347–354. https://doi.org/10.1590/1678-41625517
Alvim LB, Sandes SHC, Silva BC, Steinberg RS, Camps MHA, Acurcio LB, Arantes RME, Nicoli JR, Neumann E, Nunes AC (2016) Weissella paramesenteroides WpK4 reduces gene expression of intestinal cytokines, and hepatic and splenic injuries in a murine model of typhoid fever. Benefic Microbes 7:61–73. https://doi.org/10.3920/BM2015.0093
Acurcio LB, Bastos RW, Sandes SHC, Guimarães ACC, Alves CG, Reis DC, Wuyts S, Nunes AC, Cassali GD, Lebeer S, Souza MR, Nicoli JR (2017) Protective effect of milk fermented by Lactobacillus plantarum B7 from Brazilian artisanal cheese on a Salmonella enterica serovar Typhimurium infection in BALB/c mice. J Funct Foods 33:436–445. https://doi.org/10.1016/j.jff.2017.04.010
Ornellas RMS, Santos TT, Acurcio LB, Sandes SHC, Oliveira MM, Dias CV, Silva SC, Uetanabaro APT, Vinderola G, Nicoli JR (2017) Selection of lactic acid bacteria with probiotic potential isolated from the fermentation process of “Cupuacu” (Theobroma grandiflorum). Adv Exp Med Biol. https://doi.org/10.1007/5584_2017_5
Giulietti A, Overbergh L, Valckx D, Decallonne B, Bouillon R, Mathieu C (2001) An overview of real-time quantitative PCR: applications to quantify cytokine gene expression. Methods 25(4):386–401. https://doi.org/10.1006/meth.2001.1261
Hellemans J, Mortier G, de Paepe A, Speleman F, Vandesompele J (2007) qBase relative quantification framework and software for management and automated analysis of real-time quantitative PCR data. Genome Biol 8(R19). https://doi.org/10.1186/gb-2007-8-2-r19
Kozich JJ, Westcott SL, Baxter NT, Highlander SK, Schloss PD (2013) Development of a dual-index sequencing strategy and curation pipeline for analyzing amplicon sequence data on the miseq illumina sequencing platform. Appl Environ Microbiol 79:5112–5120. https://doi.org/10.1128/AEM.01043-13
McMurdie PJ, Holmes S (2013) Phyloseq: an R package for reproducible interactive analysis and graphics of microbiome census data. PLoS One 8. https://doi.org/10.1371/journal.pone.0061217
Yoon S, Ha S, Kwoon S, Lim J, Kim Y, Seo H, Chun J (2017) Introducing EzBioCloud: a taxonomically united database of 16S rRNA gene sequences and whole-genome assemblies. Int J Syst Evol Microbiol 67:1613–1617. https://doi.org/10.1099/ijsem.0.001755
Tsai CC, Hsih HY, Chiu HH, Lai YY, Liu JH, Yu B, Tsen HY (2005) Antagonistic activity against Salmonella infection in vitro and in vivo for two Lactobacillus strains from swine and poultry. Int J Food Microbiol 102:185–194. https://doi.org/10.1016/j.ijfoodmicro.2004.12.014
Asahara T, Shimizu K, Takada T, Kado S, Yuki N, Morotomi M, Tanaka R, Nomoto K (2011) Protective effect of Lactobacillus casei strain Shirota against lethal infection with multi-drug resistant Salmonella enterica serovar Typhimurium DT104 in mice. J Appl Microbiol 110:163–173. https://doi.org/10.1111/j.1365-2672.2010.04884.x
Castillo NA, de Moreno de Leblanc A, Galdeano CM, Perdigón G (2012) Comparative study of the protective capacity against Salmonella infection between probiotic and non-probiotic lactobacilli. J Appl Microbiol 114(3):861–876. https://doi.org/10.1111/jam.12074
Hoque R, Farooq A, Ghani A, Gorelick F, Mehal WZ (2014) Lactate reduces liver and pancreatic injury in toll-like receptor– and inflammasome-mediated inflammation via GPR81-mediated suppression of innate immunity. Gastroenterology 146:1763–1774. https://doi.org/10.1053/j.gastro.2014.03.014
De Keersmaecker SCJ, Verhoeven TLA, Desair J, Marchal K, Vanderleyden J, Nagy I (2006) Strong antimicrobial activity of Lactobacillus rhamnosus GG against Salmonella typhimurium is due to accumulation of lactic acid. FEMS Microbiol Lett 259:89–96. https://doi.org/10.1111/j.1574-6968.2006.00250.x
Perdigón G, Macias MEN, Alvarez S, Oliver G, Ruiz Holgado AP (1988) Systemic augmentation of the immune response in mice by feeding fermented milks with Lactobacillus casei and Lactobacillus acidophilus. Immunology 63:17–23
Brenchley JM, Douek DC (2012) Microbial translocation across the GI tract. Annu Rev Immunol 30:149–173. https://doi.org/10.1146/annurev-immunol-020711-075001
LeBlanc AM, Castillo NA, Perdigon G (2010) Anti-infective mechanisms induced by a probiotic Lactobacillus strain against Salmonella enterica serovar Typhimurium infection. Int J Food Microbiol 138:223–231. https://doi.org/10.1016/j.ijfoodmicro.2010.01.020
Kuzminska-Bajor M, Grzymajlo K, Ugorski M (2015) Type 1 fimbriae are important factors limiting the dissemination and colonization of mice by Salmonella Enteritidis and contribute to the induction of intestinal inflammation during Salmonella invasion. Front Microbiol 6:1–10. https://doi.org/10.3389/fmicb.2015.00276
Thiennimitr P, Winter SE, Bäumler AJ (2012) Salmonella, the host and its microbiota. Curr Opin Microbiol 15:108–114. http:// https://doi.org/10.1016/j.mib.2011.10.002
Winter SE, Thiennimitr P, Winter MG, Butler BP, Huseby DL, Crawford RW, Russell JM, Bevins CL, Adams LG, Tsolis RM, Roth JR, Bäumler AJ (2010) Gut inflammation provides a respiratory electron acceptor for Salmonella. Nature 23:426–429. https://doi.org/10.1038/nature09415
Castillo NA, Perdigon G, Leblanc AM (2011) Oral administration of a probiotic Lactobacillus modulates cytokine production and TLR expression improving the immune response against Salmonella enterica serovar Typhimurium infection in mice. BMC Microbiol 11:177. https://doi.org/10.1186/1471-2180-11-177
Chandrasekar B, Deobagkar-Lele M, Victor ES, Nandi D (2013) Regulation of chemokines, ccl3 and ccl4, by interferon γ and nitric oxide synthase 2 in mouse macrophages and during Salmonella enterica serovar Typhimurium infection. J Infect Dis 207:1556–1568. https://doi.org/10.1093/infdis/jit067
Rydström A, Wick MJ (2007) Monocyte recruitment, activation, and function in the gut-associated lymphoid tissue during oral Salmonella infection. J Immunol 178:5789–5801. https://doi.org/10.4049/jimmunol.178.9.5789
Peña JA, Versalovic J (2003) Lactobacillus rhamnosus GG decreases TNF-α production in lipopolysaccharide-activated murine macrophages by a contact-independent mechanism. Cell Microbiol 5:277–285. https://doi.org/10.1046/j.1462-5822.2003.t01-1-00275.x
Lin WH, Yu B, Lin CK, Hwang WZ, Tsen HY (2007) Immune effect of heat-killed multistrain of Lactobacillus acidophilus against Salmonella typhimurium invasion to mice. J Appl Microbiol 102:22–31. https://doi.org/10.1111/j.1365-2672.2006.03073.x
Ivanov II, Atarashi K, Manel N, Brodie EL, Shima T, Karaoz U, Wei D, Goldfarb KC, Santee CA, Lynch SV, Tanoue T, Imaoka A, Itoh K, Takeda K, Umesaki Y, Honda K, Littman DR (2009) Induction of intestinal Th17 cells by segmented filamentous bacteria. Cell 139:485–498. https://doi.org/10.1016/j.cell.2009.09.033
Mayuzumi H, Inagaki-Ohara K, Uyttenhove C, Okamoto Y, Matsuzaki G (2010) Interleukin-17A is required to suppress invasion of Salmonella enterica serovar Typhimurium to enteric mucosa. Immunology 131:377–385. https://doi.org/10.1111/j.1365-2567.2010.03310.x
Shimizu K, Ogura H, Goto M, Asahara T, Nomoto K, Morotomi M, Yoshiya K, Matsushima A, Sumi Y, Kuwagata Y, Tanaka H, Shimazu T, Sugimoto H (2006) Altered gut flora and environment in patients with severe SIRS. J Trauma 60:126–133. https://doi.org/10.1097/01.ta.0000197374.99755.fe
Castillo NA, LeBlanc AM, Galdeano CM, Pérdigon G (2012) Comparative study of the protective capacity against Salmonella infection between probiotic and nonprobiotic lactobacilli. J Appl Microbiol 114:861–876. https://doi.org/10.1111/jam.12074
Das S, Sarkar A, Choudhury SS, Owen KA, Castillo V, Fox S, Eckmann L, Elliot MR, Casanova JE, Ernst PB (2015) Engulfment and cell motility protein 1 (ELMO1) has an essential role in the internalization of Salmonella Typhimurium into enteric macrophages that impact disease outcome. Cell Mol Gastroenterol Hepatol 3:311–324. https://doi.org/10.1016/j.jcmgh.2015.02.003
Symonds EL, O’Mahony C, Laphtorne S, O’Mahony D, Sharry JM, O’Mahony L, Shanahan F (2012) Bifidobacterium infantis 35624 protects against Salmonella-induced reductions in digestive enzyme activity in mice by attenuation of the host inflammatory response. Clin Transl Gastroenterol 3(e15). https://doi.org/10.1038/ctg.2012.9
Schulz SM, Köhler G, Holscher C, Iwakura Y, Alber G (2008) IL-17A is produced by Th17, gammadelta T cells and other CD4- lymphocytes during infection with Salmonella enterica serovar Enteritidis and has a mild effect in bacterial clearance. Int Immunol 9:1129–1138. https://doi.org/10.1093/intimm/dxn069
Funding
This work was supported by the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) (grant number 470451/2011-4) and Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG) (grant number APQ-26-11 and BDS-00115-15). We thank Clélia N. Silva for valuable technical support. We also thank ENdEMIC staff, especially to the PhD students Eline Oerlemans, Ilke de Boeck and Wenke Smets for their technical support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Statement
All experimental procedures were carried out according to the Brazilian National Council for the Control of Animal Experimentation (CONCEA), which are in accordance with the guidelines for the care and use of laboratory animals described by the US National Institutes of Health.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix 1
Appendix 1
Histopathological aspects of a liver with numerous inflammatory foci (indicated by arrows) (A) and with no inflammatory foci (B). Figure A is from Salmonella challenged group with no probiotic treatment, and B is from a group challenged that received the milk fermented by L. paracasei ST11. Scale: 100 μm.
Rights and permissions
About this article
Cite this article
Acurcio, L.B., Wuyts, S., de Cicco Sandes, S.H. et al. Milk Fermented by Lactobacillus paracasei NCC 2461 (ST11) Modulates the Immune Response and Microbiota to Exert its Protective Effects Against Salmonella typhimurium Infection in Mice. Probiotics & Antimicro. Prot. 12, 1398–1408 (2020). https://doi.org/10.1007/s12602-020-09634-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12602-020-09634-x