Abstract
Purpose. The aim was to assess the feasibility of cutaneousmicrodialysis as a method to study percutaneous penetration of methyl nicotinatethrough human skin in vitro and in vivo.
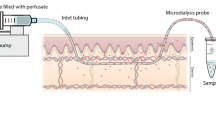
Methods. Microdialysis was applied in vitro in excised human skin,in isolated dermis, in reconstructed human epidermis and in vivo inthe volar forearm skin of volunteers using methyl nicotinate (MN) asa model compound. After topical application of MN, aliquots of theperfusate were collected and analyzed for the presence of MNspectrophotometrically and by HPLC. In vivo, visual scoring and laser Dopplerperfusion imaging (LDPI) were used to monitor the effects on skinblood flow.
Results. In vitro, MN was detected in the dialysate after a 1 minexposure of excised skin to concentrations as low as 25 mM. Higherconcentrations up to 500 mM showed increased levels. Prolongationof the application time to 60 min resulted in increased levels of MNin the perfusate as the duration of application increased. Reconstructedepidermis and isolated dermis showed an almost 2- and 20-fold higherpenetration compared to excised skin, respectively. In vivo, LDPImeasurements showed a rapid increase in skin blood flow afterapplication of 25 to 100 mM MN for 1 min. MN was only detectable inthe microdialysate after application of 100 mM for 10 min (two ofthree subjects).
Conclusions. Cutaneous microdialysis may be a tool for comparativestudies linking responses in human skin in vivo to in vitro data usingthe same technique and endpoint.
Similar content being viewed by others
REFERENCES
J. L. Fox, C. D. Yu, W. I. Higuchi, and N. F. H. Ho. General physical model for simultaneous diffusion and metabolism in biological membranes. The computational approach for steady-state case. Int. J. Pharm. 2:41-57 (1979).
J. Hadgraft. Percutaneous absorption: possibilities and problems. Int. J. Pharm. 16:255-270 (1983).
R. L. Bronaugh and H. I. Maibach. In vitro models for human percutaneous adsorption. In H. I. Maibach and N. J. Lowe (eds), Models in Dermatology, Karger, Basel, Switzerland, 1985, pp. 178-188
D. Averbeck, S. Averbeck, J. Blais, A. Moysan, G. Huppe, Moliere, P. Prognon, P. Vigny, and L. Dubertret. Suction blister fluid: its use for pharmacodynamic and toxicological studies of drugs and metabolites in vivo in human skin after topical or systemic administration. In H. I. Maibach, N. J. Lowe (eds), Models in Dermatology, Karger, Basel, Switzerland, 1989; pp. 5-11.
A. Zesch and H. Schaefer. Penetrationskinetik von radiomarkiertem hydrocortison aus verschiedenartigen Salbengrundlagen in die menschlichen Haut II in vivo. Arch. Dermatol. Forsch. 225:245-256 (1975).
C. Anderson, C. Svensson, F. Sjogren, T. Andersson, and K. Wardell. Human in vivo microdialysis technique can be used to measure cytokines in contact reactions. Curr. Probl. Dermatol. 23:121-130 (1995).
U. Ungerstedt. Microdialysis-principles and applications for studies in animals and man. J. Int. Med. 230:365-373 (1991).
L. Groth. Cutaneous microdialysis. Methodology and validation. Acta. Derm. Venereol. (Stockh) 76:5-61 (1996).
C. Anderson, T. Andersson, and M. Molander. Ethanol absorption across human skin measured by in vivo microdialysis technique. Acta. Derm. Venereol. (Stockh) 71:389-393 (1991).
L. Groth. Cutaneous microdialysis. A new technique for the assessment of skin penetration, Curr. Probl. Dermatol. 26:90-98 (1998).
L. Hegemann, C. Forstinger, B. Partsch, I. Lagler, S. Krotz, and K. Wolff. Microdialysis in cutaneous pharmacology: kinetic analysis of transdermally delivered nicotine. J. Invest. Dermatol. 104:839-843 (1995).
Anderson C, Andersson T, and Andersson RGG. In vivo microdialysis estimation of histamine in human skin. Skin. Pharmacol. 5:177-183 (1992)
L. J. Petersen, L. K. Poulsen, J. Sondergaard, and P. S. Skov. The use of cutaneous microdialysis to measure substance-P induced histamine release in intact human skin in vivo. Allergy 47:635-637 (1992).
R. H. Guy, R. C. Wester, E. Tur, and H. I. Maibach. Noninvasive assessments of the percutaneous absorption of methyl nicotinate in humans. J. Pharm. Sci. 72:1077-1079 (1983).
R. H. Guy, E. M. Carlstrom, D. A. W. Bucks, R. S. Hinz, and H. I. Maibach. Percutaneous penetration of nicotinates: in vivo and in vitro measurements. J. Pharm. Sci. 75:968-972 (1986).
R. Kohli, W. I. Archer, and A. Li Wan Po. Laser velocimetry for the non-invasive assessment of the percutaneous absorption of nicotinates. Int. J. Pharm. 36:91-98 (1987).
M. Ponec, A. Weerheim, J. Kempenaar, A. Mulder, G. S. Gooris, J. Bouwstra, and A. M. Mommaas. The formation of competent barrier lipids in reconstructed human epidermis requires the presence of vitamin C. J. Invest. Dermatol. 109:348-355 (1997).
A. Jakobsson and G. E. Nilsson. Prediction of sampling depth and photon pathlength in laser Doppler flowmetry. Med. Biol. Eng. Comput. 31:301-307 (1993).
K. Wårdell, A. Jakobsson, and G. E. Nilsson. Laser Doppler perfusion imaging by dynamic light scattering. IEEE Trans. Bio-med. Eng. 40:309-316 (1993).
M. Linden, K. Wårdell, T. Andersson, and C. Anderson. High resolution laser Doppler perfusion imaging for investigation of blood circulation changes after microdialysis probe insertion. Skin Res. Technol. 3:227-232 (1997).
L. Lafforgue, I. Eynard, F. Falson, and J. Hadgraft. Physicochemical and physiological parameters of methyl nicotinate percutaneous penetration. Prediction of Percutaneous Penetration, vol 4B;106-108 (1996).
J. D. Morrow, J. A. Awad, J. A. Oates, and L. J. Roberts II. Identification of skin as a major site of prostaglandin D2 release following oral administration of niacin in humans. J. Invest. Dermatol. 98:812-815 (1992).
E. Tur, R. H. Guy, M. Tur, and H. I. Maibach. Noninvasive assessment of local nicotinate pharmacodynamics by photople-thysmography. J. Invest. Dermatol. 80:499-503 (1983).
J. P. Fulton, E. M. Farver, and A. P. Moreci. The mechanism of action of rubefacients. J. Invest. Dermatol. 33:317-325 (1959).
J. K. Wilkin, G. Fortner, L. A. Reinhardt, O. V. Flowers, S. J. Kilpatrick, and W. C. Streeter. Prostaglandins and nicotinate-provoked increase in cutaneous blood flow. Clin. Pharmacol. Ther. 38:273-277 (1985).
J. D. Morrow, W. G. Parsons, and L. J. Roberts. Release of markedly increased quantities of prostaglandin D2 in vivo in humans following the administration of nicotinic acid. Prostaglandins 38:263-274 (1989).
A. Zesch, H. Schaefer, and G. Stuttgen. The quantitative distribution of percutaneously applied caffeine in human skin. Arch. Dermatol. Res. 266:277-283 (1979).
J. M. Ault, C. M. Riley, N. M. Meltzer, and C. E. Lunte. Dermal microdialysis sampling in vivo. Pharm. Res. 11:1631-1639 (1994).
C. J. Gean, H. I. Maibach, and R. H. Guy. Cutaneous response to topical methyl nicotinate in black, oriental, and caucasian subjects Arch. Dermatol. Res. 281:95-98 (1989).
E. Tur, H. I. Maibach, and R. H. Guy. Percutaneous penetration of methyl nicotinate at three anatomic sites: evidence for an appendageal contribution to transport? Skin Pharmacol. 4:230-234 (1991).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Boelsma, E., Anderson, C., Karlsson, A.M.J. et al. Microdialysis Technique as a Method to Study the Percutaneous Penetration of Methyl Nicotinate Through Excised Human Skin, Reconstructed Epidermis, and Human Skin In Vivo. Pharm Res 17, 141–147 (2000). https://doi.org/10.1023/A:1007505011474
Issue Date:
DOI: https://doi.org/10.1023/A:1007505011474