Abstract
Purpose. The long-term stability and nasal absorption characteristics of a basic nasal formulation of azetirelin, a thyrotropin-releasing hormone analog and its absorption enhancement by incorporation of acylcarnitines in the formulation were investigated.
Methods. The long-term stability of basic nasal azetirelin formulations at 25° C was predicted by calculation from the Arrhenius plot of the data on 6 months' storage at 40, 50 and 60° C. Nasal azetirelin absorption characteristics were kinetically examined by intranasal administration to rats, determination of plasma azetirelin level by radioimmunoassay, and fitting the data to a two-compartment model including absorption rate.
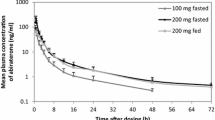
Results. Basic nasal azetirelin formulations of pH 4.0 and pH 5.1 were predicted to be highly stable. Residual azetirelin after 2 years storage at 25° C was greater than 95%. Nasal absorption characteristics of this formulation in the pH 4.0−6.3 range showed pH-dependency, with pH 4.0 showing the highest absolute bioavailability (Bioav) of 17.1%. This nasal Bioav was 21 times greater than that of oral administration (0.8%). Acylcarnitines with 12 or more carbon atoms in the acyl chain greatly enhanced nasal absorption of azetirelin: Bioavs with lauroylcarnitine chloride (LCC) and palmitoylcarnitine chloride were 96.9% and 72.9%, respectively. This enhancement by LCC plateaued at the low concentration of 0.1%.
Conclusions. The basic nasal azetirelin formulation at pH 4.0 is stable and shows adequate absorption, with nasal absorption having greater Bioav than oral absorption. The 12-carbon acylate LCC was the strongest enhancer among acylcarnitines and provided near-total delivery of the administered dose to the blood.
Similar content being viewed by others
REFERENCES
M. Yamamoto and M. Shimizu. Naunyn Schmiedebergs Arch. Pharmacol. 336:561–565 (1987).
E. Nakamura, S. Higuchi, M. Terai, and T. Yamaguchi. Oyo Yakuri 41:227–234 (1991).
I. Sasaki, T. Fujita, M. Murakami, A. Yamamoto, E. Nakamura, H. Imasaki, and S. Muranishi. Biol. Pharm. Bull. 17:1256–1261 (1994).
F. W. Merkus, N. G. Schipper, W. A. Hermens, S. G. Romeijn, and J. C. Verhoef. J. Control. Rel. 24:201–208 (1993).
L. H. Opie. Am. Heart J. 97:375–388 (1979).
J. A. Fix, K. Engle, P. A. Porter, P. S. Leppert, S. J. Selk, C. R. Gardner, and J. Alexander. Am. J. Physiol. 251:G332–340 (1986).
J. H. Hochman, J. A. Fix, and E. L. Lecluyse. J. Pharmacol. Exp. Ther. 269:813–822 (1994).
E. Nakamura, K. Yokoi, S. Kobayashi, S. Higuchi. Oyo Yakuri 41:205–212 (1991).
S. Yokohama, K. Yamashita, H. Toguchi, J. Takeuchi, and N. Kitamori. J. Pharmacobio-Dyn., 7:101–111 (1984).
T. Mitsuma and T. Nogimori. Acta. Endocrinologica. 107:207–212 (1984).
J. Sandow and W. Petri. In Y. W. Chien (Ed.), Transnasal Systemic Medications, pp. 184–186, 1985, Elsevier, Amsterdam.
S. Hirai, T. Ikenaga, and T. Matsuzawa. Diabetes 27:296–299 (1978).
T. Ohwaki, H. Ando, F. Kakimoto, K. Uesugi, S. Watanabe, Y. Miyake, and M. Kayano. J. Pharm. Sci., 76:695–698 (1987).
E. Stratford and V. H. L. Lee. Int. J. Pharm. 30:73–82 (1986).
P. Langguth, H. P. Merkle, and G. L. Amidon. Pharm. Res. 11:528–535 (1994).
S. Kagatani, T. Shinoda, M. Fukui, T. Ohmura, S. Hasumi, and T. Sonobe. Pharm. Res. 13:739–743 (1996).
E. L. LeCluyse, L. E. Appel, and S. C. Sutton. Pharm. Res. 8:84–87 (1991).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kagatani, S., Inaba, N., Fukui, M. et al. Nasal Absorption Kinetic Behavior of Azetirelin and Its Enhancement by Acylcarnitines in Rats. Pharm Res 15, 77–81 (1998). https://doi.org/10.1023/A:1011952804479
Issue Date:
DOI: https://doi.org/10.1023/A:1011952804479