Abstract
Purpose. The objective of this investigation was to determine, using the Caco-2 cell culture model, the extent to which the paracellular and transcellular routes contributed to the transport of passively absorbed drugs. An effort was also made to determine the controlling factors in this process.
Methods. We selected a heterologous series of drugs with varying physicochemical parameters for the investigation. Effective permeability coefficients of the model drugs (naproxen, phenytoin, salicylic acid, chlorothiazide, furosemide, propranolol, diltiazem, ephedrine, and cimetidine), at pH 7.2 and pH 5.4, were estimated using confluent monolayers of Caco-2 cells. The biophysical model approach, based on molecular size restricted diffusion within an electrostatic field of force, used by Adson et al. (1 ,2), was employed to estimate the permeability coefficients of the ionized and unionized forms of the drugs for the paracellular and transcellular route.
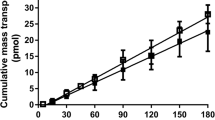
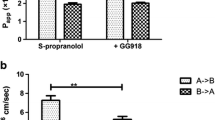
Results and Conclusions. The permeability coefficients of the acidic drugs was greater at pH 5.4, whereas that of the basic drugs was greater at pH 7.2 and the transcellular pathway was the favored pathway for most drugs, probably due to its larger accessible surface area. The paracellular permeability of the drugs was size and charge dependent. The permeability of the drugs through the tight junctions decreased with increasing molecular size. Further, the pathway also appeared to be cation-selective, with the positively charged cations of weak bases permeating the aqueous pores of the paracellular pathway at a faster rate than the negatively charged anions of weak acids. Thus, the extent to which the paracellular and transcellular routes are utilized in drug transport is influenced by the fraction of ionized and unionized species (which in turn depends upon the pKa of the drug and the pH of the solution), the intrinsic partition coefficient of the drug, the size of the molecule and its charge.
Similar content being viewed by others
REFERENCES
A. Adson, T. J. Raub, P. S. Burton, C. L. Barsuhn, A. R. Hilgers, K. L. Audus, and N. F. H. Ho. J. Pharm. Sci. 83:1529–1536 (1994).
A. Adson, P. S. Burton, T. J. Raub, C. L. Barsuhn, K. L. Audus, and N. F. H. Ho. J. Pharm. Sci. 84:1197–1204 (1995).
A. R. Hilgers, R. A. Conradi, and P. S. Burton. Pharm. Res. 7:902–910 (1990).
G. Wilson, I. F. Hassan, C. J. Dix, I. Williamson, R. Shah, and M. Mackay. J. Controlled Release 11:25–40 (1990).
P. Artursson. J. Pharm. Sci. 79:476–482 (1990).
J. N. Cogburn, M. Donovan, and C. Schasteen. Pharm. Res. 8:210–216 (1991).
I. J. Hidalgo, T. J. Raub, and R. T. Borchardt. Gastroenterology 96:736–749 (1989).
M. Rousset. Biochimie 68:1035–1040 (1986).
M. Pinto, S. Leon-Robine, M. D. Appay, M. Kedinger, N. Triadou, E. Dussaulx, B. Lacroix, P. Assmann-Simon, K. Haffen, J. Fogh, and A. Zweibaum. Biol. Cell. 47:323–330 (1983).
T. Teorell. Prog. Biophys. Mol. Biol. 3:305–369 (1953).
R. S. Pearlman. Molecular Surface Area and Volume. In: W. J. Dunn, J. H. Block, R. S. Pearlman, editors. Partition Coefficient. Determination and Estimation. New York: Pergamon Press, 1986:3–20.
E. L. Cussler. Diffusion: Mass Transfer in Fluid Systems, Cambridge University Press, NY (1986).
E. M. Renkin. J. Gen. Physiol. 38:225–243 (1955).
F. E. Curry. Mechanisms and thermodynamics of transcapillary exchange. In: E. M. Renkin, C. C. Michel, editors. The Cardiovascular System. Vol. IV. Microcirculation. Part 1. Bethesda, MD: Am. Physiol. Society, 1977:309–374.
P. A. Shore, B. B. Brodie, and C. A. M. Hogben. J. Pharmacol. Exp. Ther. 119:361–369 (1957).
J. L. Madara and K. Dharmsathaphorn. J. Cell Biol. 101:2124–2133 (1985).
K. H. Soergel Gastroenterology 105:1247–1250 (1993).
N. F. H. Ho, D. P. Thompson, T. G. Geary, T. J. Raub, and C. L. Barsuhn. Molec. Biochem. Parasitol. 41:153–166 (1990).
S. T. Ballard, J. H. Hunter, and A. E. Taylor. Annu. Rev. Nutr. 15:35–55 (1995).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pade, V., Stavchansky, S. Estimation of the Relative Contribution of the Transcellular and Paracellular Pathway to the Transport of Passively Absorbed Drugs in the Caco-2 Cell Culture Model. Pharm Res 14, 1210–1215 (1997). https://doi.org/10.1023/A:1012111008617
Issue Date:
DOI: https://doi.org/10.1023/A:1012111008617