Abstract
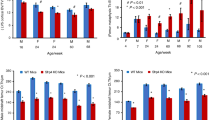
The resilience and strength of bone is due to the orderly mineralization of a specialized extracellular matrix (ECM) composed of type I collagen (90%) and a host of non-collagenous proteins that are, in general, also found in other tissues. Biglycan (encoded by the gene Bgn) is an ECM proteoglycan that is enriched in bone1,2,3 and other non-skeletal connective tissues. In vitro studies indicate that Bgn may function in connective tissue metabolism by binding to collagen fibrils4 and TGF-ß (Refs 5,6), and may promote neuronal survival7. To study the role of Bgn in vivo, we generated Bgn-deficient mice. Although apparently normal at birth, these mice display a phenotype characterized by a reduced growth rate and decreased bone mass due to the absence of Bgn. To our knowledge, this is the first report in which deficiency of a non-collagenous ECM protein leads to a skeletal phenotype that is marked by low bone mass that becomes more obvious with age. These mice may serve as an animal model to study the role of ECM proteins in osteoporosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Fisher, L.W. et al. Proteoglycans of developing bone. J. Biol. Chem. 258, 6588–6594 (1983).
Bianco, P., Fisher, L.W., Young, M.F., Termine, J.D. & Robey, P.G. Expression and localization of the two small proteoglycans biglycan and decorin in developing human skeletal and non-skeletal tissues. J. Histochem. Cytochem. 38, 1549–1563 (1990).
Fisher, L.W., Termine, J.D. & Young, M.F. Deduced protein sequence of bone small proteoglycan I (biglycan) shows homology with proteoglycan II (decorin) and several nonconnective tissue proteins in a variety of species. J. Biol. Chem. 264, 4571–4576 (1989).
Schönherr, E. et al. Interaction of biglycan with type I collagen. J. Biol. Chem. 270, 2776–2783 (1995).
Yamaguchi, Y., Mann, D.M. & Ruoslahti, E. Negative regulation of transforming growth factor-beta by the proteoglycan decorin. Nature 346, 281–284 (1990).
Hildebrand, A. et al. Interaction of the small interstitial proteoglycans biglycan, decorin and fibromodulin with transforming growth factor beta. Biochem. J. 302, 527–534 (1994).
Kappler, J. et al. Chondroitin/dermatan sulphate promotes the survival of neurons from rat embryonic neocortex. Eur. J. Neurosci. 9, 306–318 (1997).
Iozzo, R.V. & Murdoch, A.D. Proteoglycans of the extracellular environment: clues from the gene and protein side offer novel perspectives in molecular diversity and function. FASEB J. 10, 598–614 (1996).
Ruoslahti, E. Proteoglycans in cell regulation. J. Biol. Chem. 264, 13369–13372 (1989).
Ruoslahti, E. & Yamaguchi, Y. Proteoglycans as modulators of growth factor activities. Cell 64, 867–869 (1991).
Vetter, U.K. et al. in Basic and Clinical Approach to Turner Syndrome (eds Hibi, I. & Takano, K.) 143–146 (Elsevier, Amsterdam,1993).
Parfitt, A.M. et al. Bone histomorphometry: standardization of nomenclature, symbols, and units. Report of the ASBMR Histomorphometry Nomenclature Committee. J. Bone Miner. Res. 2, 595–610 (1987).
Boskey, A.L., Pleshko, N., Doty, S. & Mendelsohn, R. Applications of Fourier Transform InfraRed (FT-IR) microscopy to the study of mineralization in bone and cartilage. Cells & Materials 2, 209–220 (1992).
Boskey, A.L., Spevak, L., Doty, S.B. & Rosenberg, L. Effects of bone CS-proteoglycans, DS-decorin, and DS-biglycan on hydroxyapatite formation in a gelatin gel. Calcif. Tissue Int. 61, 298–305 (1997).
Bonadio, J. et al. A murine skeletal adaptation that significantly increases cortical bone mechanical properties. Implications for human skeletal fragility. J. Clin. Invest. 92, 1697–1705 (1993).
Kuro-o, M. et al. Mutation of the mouse klotho gene leads to a syndrome resembling aging. Nature 390, 45–51 (1997).
Lewis, D.B. et al. Osteoporosis induced in mice by overproduction of interleukin 4. Proc. Natl Acad. Sci. USA 90, 11618–11622 (1993).
Erlebacher, A. & Derynck, R. Increased expression of TGF-beta 2 in osteoblasts results in an osteoporosis-like phenotype. J. Cell Biol. 132, 195–210 (1996).
Takahashi, T., Wada, T., Mori, M., Kokai, Y. & Ishii, S. Overexpression of the granulocyte colony-stimulating factor gene leads to osteoporosis in mice. Lab Invest. 74, 827–834 (1996).
Riggs, B.L. & Melton, L.J. 3rd. Involutional osteoporosis. N. Engl. J. Med. 314, 1676–1686 (1986).
Bucay, N. et al. osteoprotegerin-deficient mice develop early onset osteoporosis and arterial calcification. Genes Dev. 12, 1260–1268 (1998).
Prockop, D.J. & Kivirikko, K.I. Collagens: molecular biology, diseases, and potentials for therapy. Annu. Rev. Biochem. 64, 403–434 (1995).
Danielson, K.G. et al. Targeted disruption of decorin leads to abnormal collagen fibril morphology and skin fragility. J. Cell Biol. 136, 729–743 (1997).
Gilmour, D.T. et al. Mice deficient for the secreted glycoprotein SPARC/osteonectin/BM40 develop normally but show severe age-onset cataract formation and disruption of the lens. EMBO J. 17, 1860–1870 (1998).
Ducy, P. et al. Increased bone formation in osteocalcin-deficient mice. Nature 382, 448–452 (1996).
Tybulewicz, V.L., Crawford, C.E., Jackson, P.K., Bronson, R.T. & Mulligan, R.C. Neonatal lethality and lymphopenia in mice with a homozygous disruption of the c-abl proto-oncogene. Cell 65, 1153–1163 (1991).
Li, E., Bestor, T.H. & Janeisch, R. Targeted mutation of the DNA methyl transferase gene results in embryonic lethality. Cell 69, 915–926 (1992).
Love, P.E., Tremblay, M.L. & Westphal, H. Targeting of the T-cell receptor zeta-chain gene in embryonic stem cells: strategies for generating multiple mutations in a single gene. Proc. Natl Acad. Sci. USA 89, 9929–9933 (1992).
Bradley, A. in Teratocarcinomas and Embryonic Stem Cells (ed. Robinson, E.J.) 113–151 (IRL, Oxford, 1987).
Fisher, L.W., Stubbs, J.T. Jr. & Young, M.F. Antisera and cDNA probes to human and certain animal model bone matrix noncollagenous proteins. Acta Orthop. Scand. Suppl. 266, 61–65 (1995).
Vignery, A. & Baron, R. Dynamic histomorphometry of alveolar bone remodeling in the adult rat. Anat. Rec. 196, 191–200 (1980).
Boyce, B.F., Yoneda, T., Lowe, C., Soriano, P. & Mundy, G.R. Requirement of pp60c-src expression for osteoclasts to form ruffled borders and resorb bone in mice. J. Clin. Invest. 90, 1622–1627 (1992).
Andersson, G. & Ek-Rylander, B. The tartrate-resistant purple acid phosphatase of bone osteoclasts—a protein phosphatase with multivalent substrate specificity and regulation. Acta Orthop. Scand. Suppl. 266, 189–194 (1995).
Acknowledgements
We thank D. Caden and M. Mankani for X-ray analysis, D. Porter for advice with gene targeting vector and D. Eanes, N. Marino, D. Paget, J. Liang and S. Dieúdonne for help with the project.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xu, T., Bianco, P., Fisher, L. et al. Targeted disruption of the biglycan gene leads to an osteoporosis-like phenotype in mice. Nat Genet 20, 78–82 (1998). https://doi.org/10.1038/1746
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/1746
This article is cited by
-
Spatial transcriptomics reveal markers of histopathological changes in Duchenne muscular dystrophy mouse models
Nature Communications (2023)
-
Regenerating zebrafish scales express a subset of evolutionary conserved genes involved in human skeletal disease
BMC Biology (2022)
-
Inhibition of stromal biglycan promotes normalization of the tumor microenvironment and enhances chemotherapeutic efficacy
Breast Cancer Research (2021)
-
Biglycan expression and its function in human ligamentum flavum
Scientific Reports (2021)
-
An in situ hybridization study of decorin and biglycan mRNA in mouse osteoblasts in vivo
Anatomical Science International (2021)