Abstract
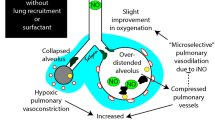
Surfactant treatment in preterm infants and term newborns with (acute respiratory distress syndrome) ARDS-like severe respiratory failure has become part of an individualized treatment strategy in many intensive care units around the world. These babies constitute heterogeneous groups of gestational ages, lung maturity, as well as of the underlying disease processes and postnatal interventions. The pathophysiology of respiratory failure in preterm infants is characterized by a combination of primary surfactant deficiency and surfactant inactivation as a result of plasma proteins leaking into the airways from areas of epithelial disruption and injury. Various pre- and postnatal factors, such as exposure to chorioamnionitis, pneumonia, sepsis and asphyxia, induce an injurious inflammatory response in the lungs of preterm infants, which may subsequently affect surfactant function, synthesis and alveolar stability. Surfactant inactivation—and dysfunction—is also a hallmark in newborns with meconium aspiration syndrome (MAS), pneumonia and other disorders affecting the pulmonary function. Although for the majority of suggested indications no data from randomized controlled trials exist, a surfactant replacement that counterbalances surfactant inactivation seems to improve oxygenation and lung function in many babies with ARDS without any apparent negative side effects. Newborns with MAS will definitely benefit from a reduced need for extracorporeal membrane oxygenation (ECMO). Clinical experience seems to justify surfactant treatment in neonates with ARDS.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Halliday HL . History of Surfactant from 1980. Biol Neonate 2005; 87: 317–322.
Ramanathan R . Surfactant therapy in preterm infants with respiratory distress syndrome and in near-term or term newborns with acute RDS. J Perinatol 2006; 26: S51–S56.
Robertson B . New targets for surfactant replacement therapy: experimental and clinical aspects. Arch Dis Child Fetal Neonatal Ed 1996; 75: F1–F3.
Jobe AH . Mechanisms to explain surfactant response. Biol Neonate 2006; 89: 298–302.
Speer CP . Role of inflammation in the evolution of bronchopulmonary dysplasia. Drug Discovery Today Dis Mech 2006; 3 (4): 409–414.
Ho NK . Factors affecting responses of infants with respiratory distress syndrome to exogenous surfactant therapy. Singapore Med J 1993; 34 (1): 74–77.
Jobe AH . Pulmonary surfactant therapy. New Engl J Med 1993; 328 (12): 861–868.
Collaborative European Multicenter Study Group. Factors influencing the clinical response to surfactant replacement therapy in babies with severe respiratory distress syndrome. Eur J Pediatr 1991; 150: 433–439.
Speer CP, Robertson B, Curstedt T, Halliday HL, Compagnone D, Gefeller O et al. Randomized European multicenter trial of surfactant replacement therapy for severe neonatal respiratory distress syndrome: single versus multiple doses of Curosurf. Pediatrics 1992; 89: 13–20.
Speer CP, Gefeller O, Groneck P, Laufkötter E, Roll C, Hanssler L et al. Randomised clinical trial of two treatment regimens of natural surfactant preparations in neonatal respiratory distress syndrome. Arch Dis Child 1995; 72: F8–F13.
Segerer H, Stevens P, Schadow B, Maier R, Kattner E, Schwarz H et al. Surfactant substitution in ventilated very low birth weight infants: factors related to response types. Pediatr Res 1991; 30: 591–596.
Charon A, Taeusch HW, Fitzgibbon C, Smith GB, Treves ST, Phelps DS . Factors associated with surfactant treatment response in infants with severe respiratory distress syndrome. Pediatrics 1998; 83: 348–354.
Lahra MM, Jeffery HE . A fetal response to chorioamnionitis is associated with early survival after preterm birth. Am J Obstet Gynecol 2004; 190: 147–151.
Schmidt B, Cao L, Mackensen-Haen S, Kendziorra H, Klingel K, Speer CP . Chorioamnionitis and inflammation of the fetal lung. Am J Obstet Gynecol 2001; 185: 173–177.
Speer CP . Inflammation and bronchopulmonary dysplasia: a continuing story. Semin Fetal Neonatal Med 2006; 11 (5): 354–362.
Wirbelauer J, Schmidt B, Klingel K, Cao L, Lang F, Speer CP . Serum and glucocorticoid-inducible kinase in pulmonary tissue of preterm fetuses exposed to chorioamnionitis. Neonatology 2007; 93: 257–262.
May M, Marx A, Seidenspinner S, Speer CP . Apoptosis and proliferation in lungs of human fetuses exposed to chorioamnionitis. Histopathology 2004; 45: 283–290.
Herting E, Gefeller O, Land M, van Sonderen L Harms K, Robertson B, Members of the Collaborative European Multicenter Study Group. Surfactant treatment of neonates with respiratory failure and group B streptococcal infection. Pediatrics 2000; 106: 957–996.
Björklund LJ, Ingimarsson J, Curstedt T, John J, Robertson B, Werner O et al. Manual ventilation with a few large breaths at birth compromises the therapeutic effect of subsequent surfactant replacement in immature lambs. Pediatr Res 1997; 42: 348–355.
Ferreira PJ, Bunch TJ, Albertine KH, Carlton DP . Circulating neutrophil concentration and respiratory distress in premature infants. J Pediatr 2000; 136: 466–472.
Jaarsma A, Braaksma MA, Geven WB, van Oeveren W, Bambang Oetomo S . Activation of the inflammatory reaction within minutes after birth in ventilated preterm lambs with neonatal respiratory distress syndrome. Biol Neonate 2004; 86: 1–5.
Turunen R, Nupponen I, Siitonen S, Repo H, Andersson S . Onset of mechanical ventilation is associated with rapid activation of circulating phagocytes in preterm infants. Pediatrics 2006; 117: 448–454.
Jobe AH . Commentary on surfactant treatment of neonates with respiratory failure and group B streptococcal infection. Pediatrics 2000; 106: 1135.
Speer CP, Ruess D, Harms K, Herting E, Gefeller O . Neutrophil elastase and acute pulmonary damage in neonates with severe respiratory distress syndrome. Pediatrics 1993; 91: 794–799.
Baur FM, Brenner B, Goetze-Speer B, Neu S, Speer CP . Natural porcine surfactant (Curosurf) down-regulates mRNA of tumor necrosis factor-alpha (TNF-alpha) and TNF-alpha type II receptor in lipopolysaccharide-stimulated monocytes. Pediatr Res 1998; 44: 32–36.
Speer CP, Robertson B, Halliday HL . Randomized trial comparing natural and synthetic surfactant: increased infection rate after natural surfactant? Acta Paediatr 2000; 89: 510–512.
Rauprich P, Möller O, Walter G, Herting E, Robertson B . Influence of modified natural or synthetic surfactant preparations on growth of bacteria causing infections in the neonatal period. Clin Diagn Lab Immunol 2000; 7: 817–822.
Speer CP, Harms K, Herting E, Neumann N, Curstedt T, Robertson B . Early versus late surfactant replacement therapy in severe respiratory distress syndrome. Lung 1990; 168 (Suppl): 870–876.
Pandit PB, Dunn MS, Colucci EA . Surfactant therapy in neonates with respiratory deterioration due to pulmonary hemorrhage. Pediatrics 1995; 95: 32–36.
Pandit PB, Dunn MS, Kelly EN, Perlman M . Surfactant replacement in neonates with early chronic lung disease. Pediatrics 1995; 95: 851–854.
Dargaville PA, South M, McDougall PN . Surfactant and surfactant inhibitors in meconium aspiration syndrome. J Pediatr 2001; 138: 113–115.
Dargaville PA, Copnell B, For the Australian and New Zealand neonatal network. The epidemiology of meconium aspiration syndrome: incidence, risk factors, therapies and outcome. Pediatrics 2006; 117: 1712–1721.
Sun B, Curstedt T, Robertson B . Surfactant inhibition in experimental meconium aspiration. Acta Paediatr 1993; 82: 182–189.
Sun B, Herting E, Curstedt T, Robertson B . Exogenous surfactant improves lung compliance and oxygenation in adult rats with meconium aspiration. J Appl Physiol 1994; 77: 1961–1971.
Herting E, Rauprich P, Stichtenoth G, Walter G, Johansson J, Robertson B . Resistance of different surfactant preparations to inactivation by meconium. Pediatr Res 2001; 50: 44–49.
Findlay R, Taeusch HW, Walther FJ . Surfactant replacement therapy for meconium aspiration syndrome. Pediatrics 1996; 97: 48–52.
Lotze A, Mitchell BR, Bulas DI, Zola EM, Shalwitz RA, Gunkel JH . Multicenter study of surfactant (beractant) use in the treatment of term infants with severe respiratory failure. J Pediatr 1998; 132: 40–47.
Chinese Collaborative Study Group for Neonatal Respiratory Disease. Treatment of severe meconium aspiration syndrome with porcine surfactant: a multicentre, randomized, controlled trial. Acta Paediatr 2005; 94: 896–902.
El Shahed AI, Dargaville P, Ohlsson A, Soll RF . Surfactant for meconium aspiration syndrome in full term/near term infants. The Cochrane Library 2007; 3, Art. No. CD002054.
Auten RL, Notter RH, Kendig JW, Davis JM, Shapiro DL . Surfactant treatment of full-term newborns with respiratory failure. Pediatrics 1991; 87: 101–107.
Khammash H, Perlman M, Wojtulewicz J, Dunn M . Surfactant therapy in full-term neonates with severe respiratory failure. Pediatrics 1993; 92: 135–139.
Ogawa Y, Ohama Y, Itakura Y . Bonchial lavage with surfactant solution for the treatment of meconium aspiration syndrome. J Jpn Med Soc Biol Interface 1996; 26: 179–184.
Lam BCC, Yeung CY . Surfactant lavage for meconium aspiration syndrome: a pilot study. Pediatrics 1999; 103: 1014–1018.
Lista G, Bianchi S, Castoldi F, Fontana P, Cavigioli F . Bronchoalveolar lavage with diluted porcine surfactant in mechanically ventilated term infants with meconium aspiration syndrome. Clin Drug Invest 2006; 26: 13–19.
Wiswell TE, Knight GR, Finer NN, Donn SM, Desai H, Walsh WF et al. A multicenter, randomized, controlled trial comparing surfaxin (Lucinactant) lavage with standard care for treatment of meconium aspiration syndrome. Pediatrics 2002; 109: 1081–1087.
Kattwinkel J . Surfactant lavage for meconium aspiration: a word of caution. Pediatrics 2002; 109: 1167–1168.
Author information
Authors and Affiliations
Corresponding author
Additional information
Presented at the Fifth Annual Evidence vs Experience in Neonatal Practices meeting, June 2008, Chicago, Illinois.
Rights and permissions
About this article
Cite this article
Wirbelauer, J., Speer, C. The role of surfactant treatment in preterm infants and term newborns with acute respiratory distress syndrome. J Perinatol 29 (Suppl 2), S18–S22 (2009). https://doi.org/10.1038/jp.2009.30
Published:
Issue Date:
DOI: https://doi.org/10.1038/jp.2009.30
Keywords
This article is cited by
-
Construction and evaluation of neonatal respiratory failure risk prediction model for neonatal respiratory distress syndrome
BMC Pulmonary Medicine (2024)
-
Comparison of the Montreux definition with the Berlin definition for neonatal acute respiratory distress syndrome
European Journal of Pediatrics (2023)
-
Surfactant therapies for pediatric and neonatal ARDS: ESPNIC expert consensus opinion for future research steps
Critical Care (2021)
-
Nebulization of Poractant alfa via a vibrating membrane nebulizer in spontaneously breathing preterm lambs with binasal continuous positive pressure ventilation
Pediatric Research (2015)