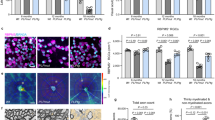
Abstract
Oligodendrocytes myelinate axons for rapid impulse conduction and contribute to normal axonal functions in the central nervous system. In multiple sclerosis, demyelination is caused by autoimmune attacks, but the role of oligodendroglial cells in disease progression and axon degeneration is unclear. Here we show that oligodendrocytes harbor peroxisomes whose function is essential for maintaining white matter tracts throughout adult life. By selectively inactivating the import factor PEX5 in myelinating glia, we generated mutant mice that developed normally, but within several months showed ataxia, tremor and premature death. Absence of functional peroxisomes from oligodendrocytes caused widespread axonal degeneration and progressive subcortical demyelination, but did not interfere with glial survival. Moreover, it caused a strong proinflammatory milieu and, unexpectedly, the infiltration of B and activated CD8+ T cells into brain lesions. We conclude that peroxisomes provide oligodendrocytes with an essential neuroprotective function against axon degeneration and neuroinflammation, which is relevant for human demyelinating diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Lazzarini, R.A. Myelin Biology and Disorders (Elsevier Academic, San Diego, California, USA, 2004).
Bjartmar, C. & Trapp, B.D. Axonal and neuronal degeneration in multiple sclerosis: mechanisms and functional consequences. Curr. Opin. Neurol. 14, 271–278 (2001).
Trapp, B.D. et al. Axonal transection in the lesions of multiple sclerosis. N. Engl. J. Med. 338, 278–285 (1998).
Griffiths, I. et al. Axonal swellings and degeneration in mice lacking the major proteolipid of myelin. Science 280, 1610–1613 (1998).
Lappe-Siefke, C. et al. Disruption of Cnp1 uncouples oligodendroglial functions in axonal support and myelination. Nat. Genet. 33, 366–374 (2003).
Suter, U. & Scherer, S.S. Disease mechanisms in inherited neuropathies. Nat. Rev. Neurosci. 4, 714–726 (2003).
Edgar, J.M. et al. Oligodendroglial modulation of fast axonal transport in a mouse model of hereditary spastic paraplegia. J. Cell Biol. 166, 121–131 (2004).
Ferreirinha, F. et al. Axonal degeneration in paraplegin-deficient mice is associated with abnormal mitochondria and impairment of axonal transport. J. Clin. Invest. 113, 231–242 (2004).
Schrader, M. & Fahimi, H.D. Mammalian peroxisomes and reactive oxygen species. Histochem. Cell Biol. 122, 383–393 (2004).
Wanders, R.J. Peroxisomes, lipid metabolism, and peroxisomal disorders. Mol. Genet. Metab. 83, 16–27 (2004).
Baes, M. et al. A mouse model for Zellweger syndrome. Nat. Genet. 17, 49–57 (1997).
Hirrlinger, J., Resch, A., Gutterer, J.M. & Dringen, R. Oligodendroglial cells in culture effectively dispose of exogenous hydrogen peroxide: comparison with cultured neurones, astroglial and microglial cells. J. Neurochem. 82, 635–644 (2002).
Gould, S.J. & Collins, C.S. Opinion: peroxisomal-protein import: is it really that complex? Nat. Rev. Mol. Cell Biol. 3, 382–389 (2002).
Dodt, G. et al. Mutations in the PTS1 receptor gene, PXR1, define complementation group 2 of the peroxisome biogenesis disorders. Nat. Genet. 9, 115–125 (1995).
Baes, M., Dewerchin, M., Janssen, A., Collen, D. & Carmeliet, P. Generation of Pex5-loxP mice allowing the conditional elimination of peroxisomes. Genesis 32, 177–178 (2002).
Chang, C.C. et al. Metabolic control of peroxisome abundance. J. Cell Sci. 112, 1579–1590 (1999).
Saher, G. et al. High cholesterol level is essential for myelin membrane growth. Nat. Neurosci. 8, 468–475 (2005).
Zoeller, R.A. & Raetz, C.R. Isolation of animal cell mutants deficient in plasmalogen biosynthesis and peroxisome assembly. Proc. Natl. Acad. Sci. USA 83, 5170–5174 (1986).
Rodemer, C. et al. Inactivation of ether lipid biosynthesis causes male infertility, defects in eye development and optic nerve hypoplasia in mice. Hum. Mol. Genet. 12, 1881–1895 (2003).
Moser, H.W., Bergin, A. & Cornblath, D. Peroxisomal disorders. Biochem. Cell Biol. 69, 463–474 (1991).
Wanders, R.J. & Waterham, H.R. Peroxisomal disorders I: biochemistry and genetics of peroxisome biogenesis disorders. Clin. Genet. 67, 107–133 (2005).
Forss-Petter, S. et al. Targeted inactivation of the X-linked adrenoleukodystrophy gene in mice. J. Neurosci. Res. 50, 829–843 (1997).
Lu, J.F. et al. A mouse model for X-linked adrenoleukodystrophy. Proc. Natl. Acad. Sci. USA 94, 9366–9371 (1997).
Kobayashi, T., Shinnoh, N., Kondo, A. & Yamada, T. Adrenoleukodystrophy protein-deficient mice represent abnormality of very long chain fatty acid metabolism. Biochem. Biophys. Res. Commun. 232, 631–636 (1997).
Simpson, J.E., Newcombe, J., Cuzner, M.L. & Woodroofe, M.N. Expression of monocyte chemoattractant protein-1 and other beta-chemokines by resident glia and inflammatory cells in multiple sclerosis lesions. J. Neuroimmunol. 84, 238–249 (1998).
McManus, C. et al. MCP-1, MCP-2 and MCP-3 expression in multiple sclerosis lesions: an immunohistochemical and in situ hybridization study. J. Neuroimmunol. 86, 20–29 (1998).
Loers, G., Aboul-Enein, F., Bartsch, U., Lassmann, H. & Schachner, M. Comparison of myelin, axon, lipid, and immunopathology in the central nervous system of differentially myelin-compromised mutant mice: a morphological and biochemical study. Mol. Cell. Neurosci. 27, 175–189 (2004).
Genoud, S. et al. Notch1 control of oligodendrocyte differentiation in the spinal cord. J. Cell Biol. 158, 709–718 (2002).
Schrader, M. & Fahimi, H.D. Peroxisomes and oxidative stress. Biochim. Biophys. Acta 1763, 1755–1766 (2006).
Loes, D.J. et al. Analysis of MRI patterns aids prediction of progression in X-linked adrenoleukodystrophy. Neurology 61, 369–374 (2003).
Wilken, B. et al. Quantitative proton magnetic resonance spectroscopy of children with adrenoleukodystrophy before and after hematopoietic stem cell transplantation. Neuropediatrics 34, 237–246 (2003).
Dubois-Dalcq, M., Feigenbaum, V. & Aubourg, P. The neurobiology of X-linked adrenoleukodystrophy, a demyelinating peroxisomal disorder. Trends Neurosci. 22, 4–12 (1999).
Mosser, J. et al. Putative X-linked adrenoleukodystrophy gene shares unexpected homology with ABC transporters. Nature 361, 726–730 (1993).
Kemp, S. & Wanders, R.J. X-linked adrenoleukodystrophy: Very long-chain fatty acid metabolism, ABC half-transporters and the complicated route to treatment. Mol. Genet. Metab. 90, 268–276 (2007).
Pujol, A. et al. Late onset neurological phenotype of the X-ALD gene inactivation in mice: a mouse model for adrenomyeloneuropathy. Hum. Mol. Genet. 11, 499–505 (2002).
Hudspeth, M.P. & Raymond, G.V. Immunopathogenesis of adrenoleukodystrophy: current understanding. J. Neuroimmunol. 182, 5–12 (2006).
Barth, P.G. et al. Late onset white matter disease in peroxisome biogenesis disorder. Neurology 57, 1949–1955 (2001).
Baumgartner, M.R. et al. Clinical approach to inherited peroxisomal disorders: a series of 27 patients. Ann. Neurol. 44, 720–730 (1998).
Goverman, J. et al. The role of CD8+ T cells in multiple sclerosis and its animal models. Curr. Drug Targets Inflamm. Allergy 4, 239–245 (2005).
Barnett, M.H. & Prineas, J.W. Relapsing and remitting multiple sclerosis: pathology of the newly forming lesion. Ann. Neurol. 55, 458–468 (2004).
Raivich, G. et al. Immune surveillance in the injured nervous system: T-lymphocytes invade the axotomized mouse facial motor nucleus and aggregate around sites of neuronal degeneration. J. Neurosci. 18, 5804–5816 (1998).
Ip, C.W. et al. Immune cells contribute to myelin degeneration and axonopathic changes in mice overexpressing proteolipid protein in oligodendrocytes. J. Neurosci. 26, 8206–8216 (2006).
Natt, O. et al. High-resolution 3D MRI of mouse brain reveals small cerebral structures in vivo. J. Neurosci. Methods 120, 203–209 (2002).
Gallyas, F. Silver staining of myelin by means of physical development. Neurol. Res. 1, 203–209 (1979).
Bielschowsky, M. Die Silberimprägnation der Neurofibrillen. J. Psychol. Neurol. 3, 169–183 (1904).
Jung, M., Sommer, I., Schachner, M. & Nave, K.A. Monoclonal antibody O10 defines a conformationally sensitive cell-surface epitope of proteolipid protein (PLP): evidence that PLP misfolding underlies dysmyelination in mutant mice. J. Neurosci. 16, 7920–7929 (1996).
Norton, W.T. & Poduslo, S.E. Myelination in rat brain: method of myelin isolation. J. Neurochem. 21, 749–757 (1973).
Bligh, E.G. & Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 37, 911–917 (1959).
Koivusalo, M., Haimi, P., Heikinheimo, L., Kostiainen, R. & Somerharju, P. Quantitative determination of phospholipid compositions by ESI-MS: effects of acyl chain length, unsaturation, and lipid concentration on instrument response. J. Lipid Res. 42, 663–672 (2001).
Gutcher, I., Urich, E., Wolter, K., Prinz, M. & Becher, B. Interleukin 18–independent engagement of interleukin 18 receptor is required for autoimmune inflammation. Nat. Immunol. 7, 946–953 (2006).
Acknowledgements
We dedicate this paper to Hugo Moser for his pioneering work on peroxisomes in childhood neurological diseases. We thank J. Barth, U. Bode, A. Fahrenholz, A. Nave, S. Relitz and S. Hühold for excellent technical assistance, and gratefully acknowledge J. Gärtner and D.H. Hunneman for clinical diagnostic service (VLCFA). This work was funded by grants from the European Union (PEX and X-ALD), the US National Multiple Sclerosis Society, the Hertie Foundation, and the generous support of the private Liley and Del Marmol Foundations.
Author information
Authors and Affiliations
Contributions
C.M.K. performed the analyses and drafted the manuscript. C.L.-S. provided Cnp-Cre and M.B. provided Pex5 floxed mice. B.B. determined myelin lipids by mass spectroscopy. A.M. and M.P. performed FACS analysis and quantified cytokines. H.B.W. participated in mouse genetics. O.N., T.M. and J.F. performed MRI. K.-A.N. designed the study and finalized the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–4 (PDF 731 kb)
Supplementary Video 1
Ataxia of Pex5flox/flox*Cnp1-Cre mice. Conditional Pex5 mutant mouse (Pex5flox/flox*Cnp1-Cre) with hind limb ataxia as found in clinical stage III-IV. Motor defects are obvious when the mouse is placed on a cage top (slow motion video). (MOV 1096 kb)
Rights and permissions
About this article
Cite this article
Kassmann, C., Lappe-Siefke, C., Baes, M. et al. Axonal loss and neuroinflammation caused by peroxisome-deficient oligodendrocytes. Nat Genet 39, 969–976 (2007). https://doi.org/10.1038/ng2070
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng2070
This article is cited by
-
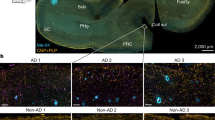
Rise and fall of peroxisomes during Alzheimer´s disease: a pilot study in human brains
Acta Neuropathologica Communications (2023)
-
Depletion of LONP2 unmasks differential requirements for peroxisomal function between cell types and in cholesterol metabolism
Biology Direct (2023)
-
Myelin insulation as a risk factor for axonal degeneration in autoimmune demyelinating disease
Nature Neuroscience (2023)
-
Four decades of chemotherapy-induced cognitive dysfunction: comprehensive review of clinical, animal and in vitro studies, and insights of key initiating events
Archives of Toxicology (2022)
-
Functionally selective activation of the dopamine receptor D2 is mirrored by the protein expression profiles
Scientific Reports (2021)