Abstract
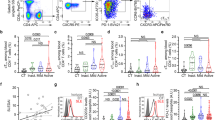
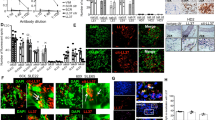
In systemic lupus erythematosus (SLE), self-reactive antibodies can target the kidney (lupus nephritis), leading to functional failure and possible mortality. We report that activation of basophils by autoreactive IgE causes their homing to lymph nodes, promoting T helper type 2 (TH2) cell differentiation and enhancing the production of self-reactive antibodies that cause lupus-like nephritis in mice lacking the Src family protein tyrosine kinase Lyn (Lyn−/− mice). Individuals with SLE also have elevated serum IgE, self-reactive IgEs and activated basophils that express CD62 ligand (CD62L) and the major histocompatibility complex (MHC) class II molecule human leukocyte antigen-DR (HLA-DR), parameters that are associated with increased disease activity and active lupus nephritis. Basophils were also present in the lymph nodes and spleen of subjects with SLE. Thus, in Lyn−/− mice, basophils and IgE autoantibodies amplify autoantibody production that leads to lupus nephritis, and in individuals with SLE IgE autoantibodies and activated basophils are factors associated with disease activity and nephritis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Rahman, A. & Isenberg, D.A. Systemic lupus erythematosus. N. Engl. J. Med. 358, 929–939 (2008).
Moser, K.L., Kelly, J.A., Lessard, C.J. & Harley, J.B. Recent insights into the genetic basis of systemic lupus erythematosus. Genes Immun. 10, 373–379 (2009).
Masutani, K. et al. Predominance of TH1 immune response in diffuse proliferative lupus nephritis. Arthritis Rheum. 44, 2097–2106 (2001).
Balomenos, D., Rumold, R. & Theofilopoulos, A.N. Interferon-γ is required for lupus-like disease and lymphoaccumulation in MRL-lpr mice. J. Clin. Invest. 101, 364–371 (1998).
Peng, S.L., Szabo, S.J. & Glimcher, L.H. T-bet regulates IgG class switching and pathogenic autoantibody production. Proc. Natl. Acad. Sci. USA 99, 5545–5550 (2002).
Zeng, D., Liu, Y., Sidobre, S., Kronenberg, M. & Strober, S. Activation of natural killer T cells in NZB/W mice induces TH1-type immune responses exacerbating lupus. J. Clin. Invest. 112, 1211–1222 (2003).
Nalbandian, A., Crispin, J.C. & Tsokos, G.C. Interleukin-17 and systemic lupus erythematosus: current concepts. Clin. Exp. Immunol. 157, 209–215 (2009).
Pernis, A.B. TH17 cells in rheumatoid arthritis and systemic lupus erythematosus. J. Intern. Med. 265, 644–652 (2009).
Valencia, X., Yarboro, C., Illei, G. & Lipsky, P.E. Deficient CD4+CD25high T regulatory cell function in patients with active systemic lupus erythematosus. J. Immunol. 178, 2579–2588 (2007).
Zhao, X.F. et al. Increased serum interleukin 17 in patients with systemic lupus erythematosus. Mol. Biol. Rep. 37, 81–85 (2010).
Akahoshi, M. et al. TH1/TH2 balance of peripheral T helper cells in systemic lupus erythematosus. Arthritis Rheum. 42, 1644–1648 (1999).
Heine, G. et al. A shift in the TH1/TH2 ratio accompanies the clinical remission of systemic lupus erythematosus in patients with end-stage renal disease. Nephrol. Dial. Transplant. 17, 1790–1794 (2002).
Shimizu, S. et al. Membranous glomerulonephritis development with TH2-type immune deviations in MRL/lpr mice deficient for IL-27 receptor (WSX-1). J. Immunol. 175, 7185–7192 (2005).
Tiller, T. et al. Autoreactivity in human IgG+ memory B cells. Immunity 26, 205–213 (2007).
Tsuiji, M. et al. A checkpoint for autoreactivity in human IgM+ memory B cell development. J. Exp. Med. 203, 393–400 (2006).
Atta, A.M., Sousa, C.P., Carvalho, E.M. & Sousa-Atta, M.L. Immunoglobulin E and systemic lupus erythematosus. Braz. J. Med. Biol. Res. 37, 1497–1501 (2004).
Odom, S. et al. Negative regulation of immunoglobulin E–dependent allergic responses by Lyn kinase. J. Exp. Med. 199, 1491–1502 (2004).
Charles, N. et al. Lyn kinase controls basophil GATA-3 transcription factor expression and induction of TH2 cell differentiation. Immunity 30, 533–543 (2009).
Beavitt, S.J. et al. Lyn-deficient mice develop severe, persistent asthma: Lyn is a critical negative regulator of TH2 immunity. J. Immunol. 175, 1867–1875 (2005).
Hibbs, M.L. et al. Multiple defects in the immune system of Lyn-deficient mice, culminating in autoimmune disease. Cell 83, 301–311 (1995).
Nishizumi, H. et al. Impaired proliferation of peripheral B cells and indication of autoimmune disease in lyn-deficient mice. Immunity 3, 549–560 (1995).
Yu, C.C., Yen, T.S., Lowell, C.A. & DeFranco, A.L. Lupus-like kidney disease in mice deficient in the src family tyrosine kinases Lyn and Fyn. Curr. Biol. 11, 34–38 (2001).
Lu, R. et al. Genetic associations of LYN with systemic lupus erythematosus. Genes Immun. 10, 397–403 (2009).
Liossis, S.N. et al. B-cell kinase lyn deficiency in patients with systemic lupus erythematosus. J. Investig. Med. 49, 157–165 (2001).
Kopf, M. et al. Disruption of the murine IL-4 gene blocks TH2 cytokine responses. Nature 362, 245–248 (1993).
Oettgen, H.C. et al. Active anaphylaxis in IgE-deficient mice. Nature 370, 367–370 (1994).
Asai, K. et al. Regulation of mast cell survival by IgE. Immunity 14, 791–800 (2001).
Kalesnikoff, J. et al. Monomeric IgE stimulates signaling pathways in mast cells that lead to cytokine production and cell survival. Immunity 14, 801–811 (2001).
Seshan, S.V. & Jennette, J.C. Renal disease in systemic lupus erythematosus with emphasis on classification of lupus glomerulonephritis: advances and implications. Arch. Pathol. Lab. Med. 133, 233–248 (2009).
Sinico, R.A. et al. Anti-C1q autoantibodies in lupus nephritis. Ann. NY Acad. Sci. 1173, 47–51 (2009).
Toran, E.J. & Lee, C.M. Isolation and analysis of nephritic-producing immune complexes in Plasmodium berghei–infected mice. J. Natl. Med. Assoc. 87, 693–699 (1995).
Perrigoue, J.G. et al. MHC class II–dependent basophil-CD4+ T cell interactions promote TH2 cytokine–dependent immunity. Nat. Immunol. 10, 697–705 (2009).
Sokol, C.L. et al. Basophils function as antigen-presenting cells for an allergen-induced T helper type 2 response. Nat. Immunol. 10, 713–720 (2009).
Yoshimoto, T. et al. Basophils contribute to TH2-IgE responses in vivo via IL-4 production and presentation of peptide–MHC class II complexes to CD4+ T cells. Nat. Immunol. 10, 706–712 (2009).
American College of Rheumatology Ad Hoc Committee on Systemic Lupus Erythematosus Response Criteria. The American College of Rheumatology response criteria for systemic lupus erythematosus clinical trials: measures of overall disease activity. Arthritis Rheum. 50, 3418–3426 (2004).
Hauswirth, A.W. et al. Recombinant allergens promote expression of CD203c on basophils in sensitized individuals. J. Allergy Clin. Immunol. 110, 102–109 (2002).
Singh, R.R., Ebling, F.M., Sercarz, E.E. & Hahn, B.H. Immune tolerance to autoantibody-derived peptides delays development of autoimmunity in murine lupus. J. Clin. Invest. 96, 2990–2996 (1995).
Kyttaris, V.C., Katsiari, C.G., Juang, Y.T. & Tsokos, G.C. New insights into the pathogenesis of systemic lupus erythematosus. Curr. Rheumatol. Rep. 7, 469–475 (2005).
Holmdahl, R., Tarkowski, A. & Jonsson, R. Involvement of macrophages and dendritic cells in synovial inflammation of collagen induced arthritis in DBA/1 mice and spontaneous arthritis in MRL/lpr mice. Autoimmunity 8, 271–280 (1991).
Levesque, M.C. Translational mini-review series on B cell–directed therapies: recent advances in B cell–directed biological therapies for autoimmune disorders. Clin. Exp. Immunol. 157, 198–208 (2009).
Schroeder, J.T. & MacGlashan, D.W. New concepts: the basophil. J. Allergy Clin. Immunol. 99, 429–433 (1997).
Mukai, K. et al. Basophils play a critical role in the development of IgE-mediated chronic allergic inflammation independently of T cells and mast cells. Immunity 23, 191–202 (2005).
Sokol, C.L., Barton, G.M., Farr, A.G. & Medzhitov, R. A mechanism for the initiation of allergen-induced T helper type 2 responses. Nat. Immunol. 9, 310–318 (2008).
Denzel, A. et al. Basophils enhance immunological memory responses. Nat. Immunol. 9, 733–742 (2008).
Chen, K. et al. Immunoglobulin D enhances immune surveillance by activating antimicrobial, proinflammatory and B cell–stimulating programs in basophils. Nat. Immunol. 10, 889–898 (2009).
Lin, H. et al. Omalizumab rapidly decreases nasal allergic response and FcɛRI on basophils. J. Allergy Clin. Immunol. 113, 297–302 (2004).
Burmeister Getz, E., Fisher, D.M. & Fuller, R. Human pharmacokinetics/pharmacodynamics of an interleukin-4 and interleukin-13 dual antagonist in asthma. J. Clin. Pharmacol. 49, 1025–1036 (2009).
De Carli, M., D'Elios, M.M., Zancuoghi, G., Romagnani, S. & Del Prete, G. Human TH1 and TH2 cells: functional properties, regulation of development and role in autoimmunity. Autoimmunity 18, 301–308 (1994).
Kono, D.H., Balomenos, D., Park, M.S. & Theofilopoulos, A.N. Development of lupus in BXSB mice is independent of IL-4. J. Immunol. 164, 38–42 (2000).
Peng, S.L., Moslehi, J. & Craft, J. Roles of interferon-γ and interleukin-4 in murine lupus. J. Clin. Invest. 99, 1936–1946 (1997).
Lee, H.Y. et al. Altered frequency and migration capacity of CD4+CD25+ regulatory T cells in systemic lupus erythematosus. Rheumatology (Oxford) 47, 789–794 (2008).
Miyajima, H. et al. IgE allotypes in sera of mice with autoimmune diseases and in mice with graft-versus-host disease after transfusion or bone marrow transplantation. Int. Arch. Allergy Immunol. 111, 152–155 (1996).
Hochberg, M.C. Updating the American College of Rheumatology revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum. 40, 1725 (1997).
Tan, E.M. et al. The 1982 revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum. 25, 1271–1277 (1982).
Liu, F.T. et al. Monoclonal dinitrophenyl-specific murine IgE antibody: preparation, isolation and characterization. J. Immunol. 124, 2728–2737 (1980).
Kepley, C.L., Craig, S.S. & Schwartz, L.B. Identification and partial characterization of a unique marker for human basophils. J. Immunol. 154, 6548–6555 (1995).
McEuen, A.R., Buckley, M.G., Compton, S.J. & Walls, A.F. Development and characterization of a monoclonal antibody specific for human basophils and the identification of a unique secretory product of basophil activation. Lab. Invest. 79, 27–38 (1999).
Acknowledgements
We thank J. Daruwalla and G. Souto-Adeva (Office of the Clinical Director, NIAMS) for human samples, data input and analysis and H.C. Oettgen (Harvard University) for providing the Igh-7−/− mice. We also thank M. Hourseau for human sample preparation and L.B. Schwartz (Virginia Commonwealth University) and A.F. Walls (University of Southampton) for the gift of human basophil–specific monoclonal antibodies. We acknowledge the support of the Laboratory Animal Care and Use Section and the Flow Cytometry Section of the Office of Science and Technology, NIAMS. This research was supported by the intramural programs of NIAMS and National Institute of Dental and Craniofacial Research, NIH.
Author information
Authors and Affiliations
Contributions
N.C. and J.R. conceived and directed the project, designed experiments and wrote the manuscript. N.C. conducted experiments. D.H., E.D. and G.G.I. provided SLE patient history, samples and analysis.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–15, Supplementary Tables 1 and 2 and Supplementary Methods (PDF 1028 kb)
Rights and permissions
About this article
Cite this article
Charles, N., Hardwick, D., Daugas, E. et al. Basophils and the T helper 2 environment can promote the development of lupus nephritis. Nat Med 16, 701–707 (2010). https://doi.org/10.1038/nm.2159
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2159
This article is cited by
-
PD-L1- and IL-4-expressing basophils promote pathogenic accumulation of T follicular helper cells in lupus
Nature Communications (2024)
-
Oxidative stress and neuroimmune proteins in a mouse model of autism
Cell Stress and Chaperones (2023)
-
High serum immunoglobulin D levels in systemic lupus erythematosus: more to be found?
Clinical Rheumatology (2023)
-
Association of peripheral basophils with tumor M2 macrophage infiltration and outcomes of the anti-PD-1 inhibitor plus chemotherapy combination in advanced gastric cancer
Journal of Translational Medicine (2022)
-
Immunoprofiling of early, untreated rheumatoid arthritis using mass cytometry reveals an activated basophil subset inversely linked to ACPA status
Arthritis Research & Therapy (2021)