Abstract
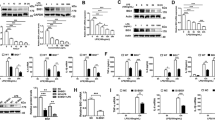
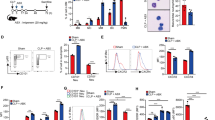
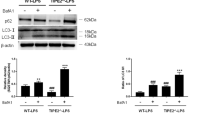
Sepsis, sepsis-induced hyperinflammation and subsequent sepsis-associated immunosuppression (SAIS) are important causes of death. Here we show in humans that the loss of the major reactive oxygen species (ROS) scavenger, glutathione (GSH), during SAIS directly correlates with an increase in the expression of activating transcription factor 3 (ATF3). In endotoxin-stimulated monocytes, ROS stress strongly superinduced NF-E2–related factor 2 (NRF2)–dependent ATF3. In vivo, this ROS-mediated superinduction of ATF3 protected against endotoxic shock by inhibiting innate cytokines, as Atf3−/− mice remained susceptible to endotoxic shock even under conditions of ROS stress. Although it protected against endotoxic shock, this ROS-mediated superinduction of ATF3 caused high susceptibility to bacterial and fungal infections through the suppression of interleukin 6 (IL-6). As a result, Atf3−/− mice were protected against bacterial and fungal infections, even under conditions of ROS stress, whereas Atf3−/−Il6−/− mice were highly susceptible to these infections. Moreover, in a model of SAIS, secondary infections caused considerably less mortality in Atf3−/− mice than in wild-type mice, indicating that ROS-induced ATF3 crucially determines susceptibility to secondary infections during SAIS.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Martin, G.S., Mannino, D.M., Eaton, S. & Moss, M. The epidemiology of sepsis in the United States from 1979 through 2000. N. Engl. J. Med. 348, 1546–1554 (2003).
Riedemann, N.C., Guo, R.F. & Ward, P.A. Novel strategies for the treatment of sepsis. Nat. Med. 9, 517–524 (2003).
Hotchkiss, R.S. & Opal, S. Immunotherapy for sepsis—a new approach against an ancient foe. N. Engl. J. Med. 363, 87–89 (2010).
Bone, R.C. et al. Definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. The ACCP/SCCM Consensus Conference Committee. American College of Chest Physicians/Society of Critical Care Medicine. 1992. Chest 136, e28 (2009).
Imai, Y. et al. Identification of oxidative stress and Toll-like receptor 4 signaling as a key pathway of acute lung injury. Cell 133, 235–249 (2008).
Bone, R.C. et al. A controlled clinical trial of high-dose methylprednisolone in the treatment of severe sepsis and septic shock. N. Engl. J. Med. 317, 653–658 (1987).
Ziegler, E.J. et al. Treatment of gram-negative bacteremia and septic shock with HA-1A human monoclonal antibody against endotoxin. A randomized, double-blind, placebo-controlled trial. The HA-1A Sepsis Study Group. N. Engl. J. Med. 324, 429–436 (1991).
Fisher, C.J. Jr. et al. Treatment of septic shock with the tumor necrosis factor receptor:Fc fusion protein. The Soluble TNF Receptor Sepsis Study Group. N. Engl. J. Med. 334, 1697–1702 (1996).
Abraham, E. et al. Efficacy and safety of monoclonal antibody to human tumor necrosis factor alpha in patients with sepsis syndrome. A randomized, controlled, double-blind, multicenter clinical trial. TNF-alpha MAb Sepsis Study Group. J. Am. Med. Assoc. 273, 934–941 (1995).
Fisher, C.J. Jr. et al. Initial evaluation of human recombinant interleukin-1 receptor antagonist in the treatment of sepsis syndrome: a randomized, open-label, placebo-controlled multicenter trial. Crit. Care Med. 22, 12–21 (1994).
Hotchkiss, R.S. & Karl, I.E. The pathophysiology and treatment of sepsis. N. Engl. J. Med. 348, 138–150 (2003).
Ward, N.S., Casserly, B. & Ayala, A. The compensatory anti-inflammatory response syndrome (CARS) in critically ill patients. Clin. Chest Med. 29, 617–625 (2008).
Muller Kobold, A.C. et al. Leukocyte activation in sepsis; correlations with disease state and mortality. Intensive Care Med. 26, 883–892 (2000).
Hotchkiss, R.S. et al. Prevention of lymphocyte cell death in sepsis improves survival in mice. Proc. Natl. Acad. Sci. USA 96, 14541–14546 (1999).
Osuchowski, M.F., Welch, K., Siddiqui, J. & Remick, D.G. Circulating cytokine/inhibitor profiles reshape the understanding of the SIRS/CARS continuum in sepsis and predict mortality. J. Immunol. 177, 1967–1974 (2006).
Gogos, C.A., Drosou, E., Bassaris, H.P. & Skoutelis, A. Pro- versus anti-inflammatory cytokine profile in patients with severe sepsis: a marker for prognosis and future therapeutic options. J. Infect. Dis. 181, 176–180 (2000).
Steinhauser, M.L. et al. IL-10 is a major mediator of sepsis-induced impairment in lung antibacterial host defense. J. Immunol. 162, 392–399 (1999).
Gilchrist, M. et al. Systems biology approaches identify ATF3 as a negative regulator of Toll-like receptor 4. Nature 441, 173–178 (2006).
Whitmore, M.M. et al. Negative regulation of TLR-signaling pathways by activating transcription factor-3. J. Immunol. 179, 3622–3630 (2007).
Rosenberger, C.M., Clark, A.E., Treuting, P.M., Johnson, C.D. & Aderem, A. ATF3 regulates MCMV infection in mice by modulating IFN-gamma expression in natural killer cells. Proc. Natl. Acad. Sci. USA 105, 2544–2549 (2008).
Gilchrist, M. et al. Activating transcription factor 3 is a negative regulator of allergic pulmonary inflammation. J. Exp. Med. 205, 2349–2357 (2008).
Hai, T., Wolfgang, C.D., Marsee, D.K., Allen, A.E. & Sivaprasad, U. ATF3 and stress responses. Gene Expr. 7, 321–335 (1999).
Biolo, G., Antonione, R. & De Cicco, M. Glutathione metabolism in sepsis. Crit. Care Med. 35, S591–S595 (2007).
Fläring, U.B., Hebert, C., Wernerman, J., Hammarqvist, F. & Rooyackers, O.E. Circulating and muscle glutathione turnover in human endotoxaemia. Clin. Sci. (Lond.) 117, 313–319 (2009).
Chan, K., Han, X.D. & Kan, Y.W. An important function of Nrf2 in combating oxidative stress: detoxification of acetaminophen. Proc. Natl. Acad. Sci. USA 98, 4611–4616 (2001).
Kim, K.H., Jeong, J.Y., Surh, Y.J. & Kim, K.W. Expression of stress-response ATF3 is mediated by Nrf2 in astrocytes. Nucleic Acids Res. 38, 48–59 (2010).
Echtenacher, B., Mannel, D.N. & Hultner, L. Critical protective role of mast cells in a model of acute septic peritonitis. Nature 381, 75–77 (1996).
Kopf, M. et al. Impaired immune and acute-phase responses in interleukin-6-deficient mice. Nature 368, 339–342 (1994).
Kneilling, M. et al. Targeted mast cell silencing protects against joint destruction and angiogenesis in experimental arthritis in mice. Arthritis Rheum. 56, 1806–1816 (2007).
Annane, D., Bellissant, E. & Cavaillon, J.M. Septic shock. Lancet 365, 63–78 (2005).
Benjamim, C.F., Hogaboam, C.M., Lukacs, N.W. & Kunkel, S.L. Septic mice are susceptible to pulmonary aspergillosis. Am. J. Pathol. 163, 2605–2617 (2003).
Benjamim, C.F., Hogaboam, C.M. & Kunkel, S.L. The chronic consequences of severe sepsis. J. Leukoc. Biol. 75, 408–412 (2004).
Akram, A. et al. Activating transcription factor 3 confers protection against ventilator induced lung injury. Am. J. Respir. Crit. Care Med. 182, 489–500 (2010).
Li, H.F., Cheng, C.F., Liao, W.J., Lin, H. & Yang, R.B. ATF3-mediated epigenetic regulation protects against acute kidney injury. J. Am. Soc. Nephrol. 21, 1003–1013 (2010).
Tsung, A. et al. HMGB1 release induced by liver ischemia involves Toll-like receptor 4 dependent reactive oxygen species production and calcium-mediated signaling. J. Exp. Med. 204, 2913–2923 (2007).
Gill, R., Tsung, A. & Billiar, T. Linking oxidative stress to inflammation: Toll-like receptors. Free Radic. Biol. Med. 48, 1121–1132 (2010).
Marino, M.W. et al. Characterization of tumor necrosis factor-deficient mice. Proc. Natl. Acad. Sci. USA 94, 8093–8098 (1997).
Beutler, B., Milsark, I.W. & Cerami, A.C. Passive immunization against cachectin/tumor necrosis factor protects mice from lethal effect of endotoxin. Science 229, 869–871 (1985).
Fattori, E. et al. Defective inflammatory response in interleukin 6-deficient mice. J. Exp. Med. 180, 1243–1250 (1994).
Villa, P., Saccani, A., Sica, A. & Ghezzi, P. Glutathione protects mice from lethal sepsis by limiting inflammation and potentiating host defense. J. Infect. Dis. 185, 1115–1120 (2002).
Ortolani, O. et al. The effect of glutathione and N-acetylcysteine on lipoperoxidative damage in patients with early septic shock. Am. J. Respir. Crit. Care Med. 161, 1907–1911 (2000).
Alves-Filho, J.C. et al. Interleukin-33 attenuates sepsis by enhancing neutrophil influx to the site of infection. Nat. Med. 16, 708–712 (2010).
Döcke, W.D. et al. Monocyte deactivation in septic patients: restoration by IFN-gamma treatment. Nat. Med. 3, 678–681 (1997).
Deng, J.C. et al. Sepsis-induced suppression of lung innate immunity is mediated by IRAK-M. J. Clin. Invest. 116, 2532–2542 (2006).
Hoogerwerf, J.J. et al. Loss of suppression of tumorigenicity 2 (ST2) gene reverses sepsis-induced inhibition of lung host defense in mice. Am. J. Respir. Crit. Care Med. 183, 932–940 (2011).
Sultzer, B.M. Genetic control of leucocyte responses to endotoxin. Nature 219, 1253–1254 (1968).
Janeway, C.A. Jr. & Medzhitov, R. Innate immune recognition. Annu. Rev. Immunol. 20, 197–216 (2002).
O'Brien, A.D. et al. Genetic control of susceptibility to Salmonella typhimurium in mice: role of the LPS gene. J. Immunol. 124, 20–24 (1980).
Poltorak, A. et al. Defective LPS signaling in C3H/HeJ and C57BL/10ScCr mice: mutations in Tlr4 gene. Science 282, 2085–2088 (1998).
Levy, M.M. et al. 2001 SCCM/ESICM/ACCP/ATS/SIS International Sepsis Definitions Conference. Intensive Care Med. 29, 530–538 (2003).
Hartman, M.G. et al. Role for activating transcription factor 3 in stress-induced beta-cell apoptosis Mol. Cell. Biol. 24, 5721–5732 (2004).
Ghoreschi, K. et al. Interleukin-4 therapy of psoriasis induces Th2 responses and improves human autoimmune disease. Nat. Med. 9, 40–46 (2003).
Biedermann, T. et al. IL-4 instructs TH1 responses and resistance to Leishmania major in susceptible BALB/c mice. Nat. Immunol. 2, 1054–1060 (2001).
Ghoreschi, K. et al. Fumarates improve psoriasis and multiple sclerosis by inducing type II dendritic cells. J. Exp. Med. 208, 2291–2303 (2011).
Acknowledgements
We thank B. Pichler, N. Suttorp, T. Welte, S. Werner and P. Dotto for helpful discussions, and for reviewing the manuscript, and M. Haberbosch and S. Hemberger for technical assistance. S. Werner (Eidgenössische Technische Hochschule Zürich) provided the Nrf2−/− (Nfe2l2tm1Ywk) mice25 with the permission of J.A. Johnson (University of California, San Francisco). This work has been supported by grants from the Sander Stiftung (2005.043.2 and 2005.043.3), Deutsche Krebshilfe (no. 109037), Deutsche Forschungsgemeinschaft (SFB 685 A6 and C1, Bi 696/3-3, Bi 696/5-1), German Federal Ministry of Education and Research (BMBF FKZ 0315079 to K.G., DLR 01GN0970 to M.R.), University of Tuebingen (W.H., f-33654-87; E.G., f-1803-0-0; K.G. and M.R., IZKF-Tuebingen, collaborative research program), European Union FP7-HEALTH-2007-2.4.4-1 200515 (M.R.), The German Dermatologic Society/Arbeitsgemeinschaft Dermatologische Forschung (W.H.), a National Research Foundation of Korea grant that was funded by the Ministry of Education, Science and Technology through the Creative Research Initiative Program (grant R16-2004-001010010,2010) and a World Class University grant (no. R31-2008-000-10103-0).
Author information
Authors and Affiliations
Contributions
W.H., B.E., E.G., N.V., A.T. and F.W. performed the experiments and analytical methods (quantitative PCR, western blots, flow cytometry, ELISA, CLP and endotoxin model) and analyzed the data. J.-H.P., K.-H.K. and K.-W.K. performed the luciferase and chromatin immunoprecipitation experiments. K.H. and C.K. collected the blood samples of subjects with sepsis. P.H. measured the bacterial and fungal loads. K.F. and M.K. performed arthritis experiments. J.B., K.G. and M.R. developed the glutathione depletion model in vitro and in vivo and the initial proof of concept. W.H., B.E., E.G., F.W., T.B. and M.R. designed the experiments. W.H., B.E., E.G., F.W., J.B., K.G., T.B. and M.R. discussed the manuscript. M.R. developed the concept. W.H., E.G., B.E. and M.R. coordinated and directed the project. T.H. provided the Atf3−/− mice. W.H., T.B. and M.R. interpreted the data and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8, Supplementary Table 1 and Supplementary Methods (PDF 3085 kb)
Rights and permissions
About this article
Cite this article
Hoetzenecker, W., Echtenacher, B., Guenova, E. et al. ROS-induced ATF3 causes susceptibility to secondary infections during sepsis-associated immunosuppression. Nat Med 18, 128–134 (2012). https://doi.org/10.1038/nm.2557
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2557
This article is cited by
-
Sevoflurane Exposure Induces Neuronal Cell Ferroptosis Initiated by Increase of Intracellular Hydrogen Peroxide in the Developing Brain via ER Stress ATF3 Activation
Molecular Neurobiology (2024)
-
Differential cardiomyocyte transcriptomic remodeling during in vitro Trypanosoma cruzi infection using laboratory strains provides implications on pathogenic host responses
Tropical Medicine and Health (2023)
-
NRF2 mediates melanoma addiction to GCDH by modulating apoptotic signalling
Nature Cell Biology (2022)
-
Decreased expression of ATF3, orchestrated by β-catenin/TCF3, miR-17-5p and HOXA11-AS, promoted gastric cancer progression via increased β-catenin and CEMIP
Experimental & Molecular Medicine (2021)
-
Genetic loss-of-function of activating transcription factor 3 but not C-type lectin member 5A prevents diabetic peripheral neuropathy
Laboratory Investigation (2021)