Abstract
Current therapies for asthma and allergy are relatively safe and effective at controlling symptoms but do not change the chronic course of disease. There is no established method to prevent asthma and allergy, and major unmet needs in this area include the better control of the severe forms of these diseases and the developments of curative therapies. Two major therapeutic strategies for asthma and allergy are currently being developed, and I here discuss the advances and challenges for future therapeutic development in these two areas. The first approach, allergen-specific immunotherapy, aims to induce specific immune tolerance and has a long-term disease-modifying effect. The second approach is the use of biological immune response modifiers to decrease pathological immune responses. Combination strategies using both of these approaches may also provide a route for addressing the unmet clinical needs in allergic diseases.
Similar content being viewed by others
Main
Allergic inflammation is caused by the development of an allergen-induced immune response and can lead to several diseases, including asthma, allergic rhinoconjunctivitis, anaphylaxis, urticaria and atopic dermatitis1. It is now becoming clear that allergic diseases are complex disorders and that there are several disease variants caused by different underlying cellular and molecular mechanisms. A limited knowledge of the pathophysiology of these disease subgroups has been the greatest obstacle to identifying consistent correlations between genes, the environment and the different disease subgroups. Although there are several clinically relevant phenotypes for asthma, atopic dermatitis and urticaria, these phenotypes might not provide any insights into the mechanisms that underpin the diseases. It is now thought that some clinical trials may have previously been unsuccessful because they were performed without attempting to classify patients into subgroups defined by distinct pathophysiologies, namely 'endotypes'2. In asthma, phenotypes describe clinically relevant characteristics but do not necessarily give insights into the underlying pathological mechanisms of the disease. In contrast, asthma endotypes describe disease subtypes on the basis of cellular and molecular pathogenic mechanisms3. In addition, the results of large-scale genetic studies, particularly studies of asthma and atopic dermatitis, may have been inconclusive because of the bulk patient selection approaches used in many of these studies (Box 1).
Despite substantial improvements in treatments for asthma and allergy, approximately 5–10% of all people with these disorders have inadequately controlled severe persistent asthma or severe atopic dermatitis. These individuals use a large proportion of public health resources devoted to the treatment of asthma and allergy, and new, more effective therapies are urgently required. The possibility of curing allergic diseases is an essential issue for research because the medications currently used to treat these diseases, such as antihistamines, leukotriene receptor antagonists and glucocorticoids, only temporarily relieve symptoms by suppressing inflammation4. However, a long-term cure for allergic diseases can be achieved through the use of allergen-specific immunotherapy (allergen SIT), which has a disease-modifying effect and might also lead to decreased requirements for anti-inflammatory and symptomatic medications5,6. The basic principle of allergen SIT is to induce immune tolerance to allergens through multiple cellular and molecular mechanisms by administering repeated, increased doses of the causative allergen. Almost all of the immune tolerance mechanisms involved in responses to SIT are currently being targeted by drug discovery programs.
Unmet needs in treatment and advances in molecular biology and immunology have also spurred the development of new biological immune response modifiers to treat allergy and asthma. Such biological modifiers (biologicals) include therapeutic antibodies, soluble receptors, cytokines and small molecules, as well as combinations of these agents that can target effector molecules at various points in the immune and inflammatory pathways on different immune cells. So far, over 30 monoclonal antibodies have been approved for various clinical indications, particularly for autoimmune disorders, organ transplantation, infectious diseases and cancer. More than 300 biologicals are currently in clinical trials, and some researchers have suggested that there will be a switch from the use of chemicals to the use of biologicals in drug development within the next 10 years. Although this review will focus on the results from human clinical trials of such biological therapeutics, I also highlight promising therapeutic candidates from preclinical and mouse studies, which will need to be validated and tested in humans to see whether they fulfill their initial promise.
As there is evidence that allergen SIT might offer the possibility of a cure for asthma and allergic diseases, many research efforts are now concentrating on combining allergen SIT with new biological immune modifiers (Table 1). Such combination strategies could be advantageous over individual therapies in reducing side effects, providing synergistic effects that may allow for the treatment of persistent disease and providing more flexibility for treatments to be tailored according to different disease endotypes. Devising the separate treatment arms of the combination of allergen SIT and a biological immune modifier will pose substantial challenges, and we still have a long way to go before we can bring such combinations to the clinic.
Mechanistic insights into immune tolerance to allergens
The concept of inducing immune tolerance has become crucial in the development of prevention and treatment strategies for many diseases in which dysregulation of the immune system has a key role, such as allergy, asthma, autoimmunity, organ transplantation and infertility. Immune tolerance to allergens is characterized by the establishment of long-term clinical tolerance5,6. In addition to the immune responses induced by various modes of allergen SIT, the development of healthy immune responses in beekeepers and cat owners who are exposed to high doses of allergens has been intensively studied to understand the mechanisms of allergen tolerance in humans7,8. Although several details of the tolerance process have yet not been elucidated, mechanisms involved include changes in the profiles of allergen-specific memory T and B cell responses and the production of specific antibody isotypes to skew the immune response in a noninflammatory direction, as well as decreased activation, tissue migration and degranulation of mast cells, basophils and eosinophils (Box 2).
Rapid desensitization of mast cells and basophils by allergens.
Several mechanisms have been proposed to operate to ensure that mast cells and basophils are unresponsive to environmental proteins, even in the presence of specific immunoglobulin E (IgE). Notably, in patients, even after the first injection of allergen SIT, very early decreases in the susceptibility of mast cells and basophils to degranulation and decreases in systemic anaphylaxis are observed, although all of the treated individuals have high quantities of specific IgE. Although the underlying molecular pathways of this response are not yet elucidated, this mechanism seems similar to the one observed when these two immune-cell types are rapidly desensitized in anaphylactic reactions to drugs9. Anaphylaxis is associated with the release of inflammatory mediators from both mast cells and basophils, and successful hyposensitization alters the magnitude of the mediator release10. The release of these inflammatory mediators in low quantities (below the required dose for systemic anaphylaxis) may affect the threshold of activation of the mast cells and basophils10,11.
Regulatory T (Treg) cells and peripheral T cell tolerance to allergens.
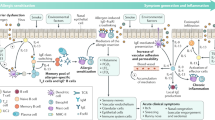
The induction of peripheral T cell tolerance, which is characterized primarily by the generation of allergen-specific Treg cells, is an essential step in allergen SIT12,13 (Fig. 1). Peripheral tolerance is initiated by the cytokines interleukin-10 (IL-10) and transforming growth factor-β (TGF-β), which are produced in increasing amounts by the allergen-specific Treg cells as allergen SIT proceeds12,13. Roles have been shown for both IL-10–secreting type 1 regulatory T (Treg1) cells or forkhead box P3 (FOXP3)-positive Treg cell subsets, suggesting that there is an overlap in the functions and characteristics of the cytokines and surface molecules of these inducible subsets of Treg cells in humans14. CD4+CD25+ Treg cells from atopic donors showed a reduced potential to inhibit CD4+CD25− T cell proliferation, which is an indicator of peripheral allergen tolerance15. Allergen SIT has been shown to increase the number of FOXP3+CD25+CD3+ cells in the nasal mucosa, and this increase associates with clinical efficacy and the suppression of seasonal allergic inflammation, supporting a putative role for Treg cells in the induction of allergen-specific tolerance16. A study investigating a wasp venom immunotherapy used human major histocompatibility complex (MHC) class II tetramers to monitor clinical allergen tolerance and showed that this therapy led to a switch in the frequencies of the antigen-specific T cells that produced certain cytokines. In response to the wasp venom immunotherapy, there was a marked decrease in the number of IL-4–producing T cells and an increase in the number of FOXP3+ and IL-10–producing antigen-specific CD4+ T cells17. Similarly, peptide immunotherapy in allergic asthma leads to decreased T helper type 2 (TH2) cell responses as a result of IL-10–dependent peripheral T cell tolerance. Therapy with peptides of selected T cell epitopes from the Fel d 1 major cat allergen resulted in a suppression of T cell proliferation in response to other 'linked' T cell epitopes within the same allergen18. As has been shown in other models, such as in suppression of germinal center reactions and in intestinal lymphoid tissues, this T cell suppression can generally take place in both secondary lymphoid organs19,20 and in the affected tissues21.
After the first administration of the SIT vaccine, there is an early decrease in mast cell and basophil degranulation and a decreased tendency for systemic anaphylaxis resulting from early desensitization. Allergen-specific Treg cells are then generated, and there is a suppression of allergen-specific TH2 cells and other effector cells. Because of the immune tolerance of TH2 cells, they can no longer contribute to IgE production, endothelial cell activation, TH2 cell homing to tissues, mucus production by the epithelium or tissue migration, priming and survival of mast cells, eosinophils and basophils. IL-10 and TGF-β directly and indirectly regulate B cells and effector cells. Other T cell subsets, such as TH1, TH9, TH17 and TH22 cells, are suppressed by Treg cells. Within the spectrum of changes in the immune system after allergen SIT, there is a relatively early increase in the amount of IgG4 and a late decrease in the amount of IgE. A substantial decrease in the allergen-specific IgE-to-IgG4 ratio occurs several months after allergen SIT. A decrease in the number of tissue mast cells and eosinophils and in the release of their mediators and a decrease in late-phase response is observed in the affected tissues (red lines, suppression; black arrows, induction). DC, dendritic cell.
The investigation of human high-dose allergen exposure models has also provided key insights into the nature of Treg cell responses in allergen tolerance. In nonallergic beekeepers and cat owners7,8, Treg cells specific for the major allergens in bee venom and cat saliva represent the major T cell subset present in these healthy individuals. These Treg cells use numerous suppressive mechanisms, including the involvement of IL-10, TGF-β, cytotoxic T lymphocyte antigen 4 (CTLA4) and programmed death 1 (PD1)7,22. Supporting these findings, FOXP3 expression negatively correlates with the amount of IgE, eosinophilia and interferon-γ (IFN-γ), and the ratio of FOXP3+ T cells to total CD4+ T cells is significantly lower in individuals with asthma or atopic dermatitis compared to healthy individuals23. In addition, the expression of FOXP3 correlates with the suppressive capacity of Treg cells24.
Studies of Treg cells in allergic individuals from other populations have also highlighted the key role of these cells in allergen tolerance. The induction of mucosal tolerance against dietary antigens is associated with the development of CD4+CD25+ Treg cells. Children who develop clinical tolerance to milk have decreased peripheral blood mononuclear cell proliferation in response to bovine β-lactoglobulin as a consequence of an increased number of circulating CD4+CD25+ Treg cells25. In allergic children, the number of Treg cells increases during the pollen season, and these Treg cells may have a role in the control of other T cell subsets that are activated by pollen allergens26. Both healthy and allergic individuals produce all three types of allergen-specific subsets of T cells, namely TH1, TH2 and Treg1 cells, but in different proportions. Accordingly, a change in the balance between TH2 and Treg cells, particularly in the dominant allergen-specific T cell subset, may lead to either the development of allergy or the recovery from allergic disease7,22.
Allergen-specific IgE and IgG responses.
Instead of becoming fully tolerant to allergen SIT as T cells do, B cells show a decisive skew from producing IgE to IgG4 in response to allergen SIT27. Allergen SIT induces a transient increase in serum-specific IgE concentrations followed by a gradual decrease in these concentrations over years of treatment28. In contrast, the specific concentration of IgG4 in serum has a relatively early and rapid increase in response to allergen SIT, and this concentration continues to increase during the whole duration that SIT is administered. IL-10 produced by Treg cells potently suppresses allergen-specific IgE while increasing IgG4 production12,29. Therefore, in addition to inducing T cell tolerance, IL-10 regulates the production of specific Ig isotypes, directing the immune response toward a noninflammatory phenotype. However, this effect of IL-10 cannot explain the observed therapeutic effect of allergen SIT because the decrease in serum IgE concentration in response to SIT, occurs relatively late in the process and does not correlate with clinical improvement after treatment. The reason for the long time gap between the generation of Treg cells after SIT and any changes in IgE concentrations is thought to be related to the continued production of IgE by plasma cells residing in the bone marrow that have a very long lifespan30.
An analysis of the regulation of IgG subtypes by allergen SIT indicated that there are increases in the plasma concentrations of allergen-specific IgG1 and, particularly, IgG4 in response to SIT, and that these increases are in the range of 10- to 100-fold31. IgG4 competes with allergens for binding to the IgE on the Fcɛ receptors of mast cells and basophils and thus acts as a blocking antibody that prevents the activation and degranulation of effector cells32. In addition, some distinct features of IgG4 suggest that it may have an anti-inflammatory role. IgG4 antibodies are dynamic molecules that exchange fragment, antigen binding (Fab) arms by swapping heavy-light chain pairs between IgG4 molecules with different specificities. This process results in the production of bi-specific antibodies with a substantially decreased capacity for crosslinking because they are functionally monomeric33. In addition, the IgG4 hinge region has specialized structural features that result in a lower affinity for certain Fcγ receptors, and IgG4 does not fix complement and can inhibit immune-complex formation by other antibody isotypes34.
Suppression of mast cells, basophils and eosinophils.
The thresholds for mast cell and basophil activation and decreased IgE-mediated histamine release are efficiently modulated by allergen SIT35. Several molecular mechanisms have been proposed for how Treg cells and anti-inflammatory cytokines affect mast cells and basophils. IL-10 suppresses IL-5 production through human TH2 cells, reduces proinflammatory cytokine release from mast cells and downregulates eosinophil function and activity36. Furthermore, through direct contact between Treg cells and mast cells, Treg cells inhibit Fcɛ receptor I (FcɛRI)-dependent mast cell degranulation37. Mast cells also seem to have a role in immune tolerance and are not simply enhancers of allergic inflammation; for example, mast cells downregulate allergic inflammation in ultraviolet light–induced skin injury and venom-induced tissue damage models in which IL-10 has a key role38,39. Treg cells have been reported to be suppressive in various models of eosinophilic inflammation, including inflammation induced by schistosome infection and asthma-like lung inflammation in mice40. In addition, decreased numbers of eosinophils and decreased concentrations of eosinophil chemoattractants and eosinophil mediators have been observed in the nasal mucosa as a long-lasting effect of allergen SIT41.
Vaccine development for allergen SIT and immunomodulation
Although it has been performed in the clinic for the past 100 years, allergen SIT faces several problems related to its limited efficacy, side effects, low patient adherence and the high costs resulting from the long duration (3–5 years) of treatment required. The main approaches to improve the efficacy and safety of vaccine-based allergen SIT can be classified into six groups (Table 2).
The first approach is targeting T cells to induce T cell tolerance and bypassing IgE binding to avoid IgE-mediated side effects42. The conformation dependence of B cell epitopes and the linearity of the amino acid sequence of T cell epitopes in the three-dimensional structure of an allergen have been targeted using allergen fragments, fusions, hybrids and chimeras42,43,44,45. The prototype of this approach is peptide immunotherapy that uses linear T cell epitope peptides46,47,48,49. All of these T cell–targeting approaches enable higher doses of allergens to be sequentially administered, which is required to induce T cell tolerance without risk of anaphylaxis42,50.
The second approach is the use of recombinant allergens or their mixtures to partially reconstitute an allergen extract. A study that tried to reconstruct a native grass pollen allergen extract using a mixture of five recombinant allergens was effective in reducing symptoms and the need for symptomatic medication in patients with grass pollen allergy31. All treated subjects developed high allergen-specific IgG1 and IgG4 antibody responses. During the study, seven systemic reactions were observed in patients from a total of 1,479 injections. Relatively mild local side effects related to treatment were observed in 10.7% of the patients that were actively treated with the recombinant allergens and in 5.9% of the patients receiving placebo. There have been 15 clinical trials that used recombinant allergens and showed greater clinical efficacy of the allergens compared to placebo over the last decade. However, licensing all these products may not be cost effective, particularly for minor allergens that do not cause allergic reactions in a substantial number of people. Recombinant vaccines for grass pollen, birch pollen and house dust mites have been the major focus in these trials.
The third approach is to physically couple allergens to stimulators of the innate immune response. This area is open to the possibility of future developments, as there are infinite numbers of possible combinations owing to the existence of multiple immune stimulators and methods for coupling51,52,53,54,55. Notably, the time, intensity and tissue location of stimulation of the innate immune response by the allergen are key to the induction of tolerance or immune activation. For example, house dust mites can activate Toll-like receptor 4 (TLR4) in airway structural cells to induce asthma-like inflammation in mice56. However, it should also be noted that natural exposure to house dust mites and other aeroallergens involves exposure to innate immune response–stimulating substances that activate TLR4.
The fourth approach is varying the routes of vaccine administration. A meta-analysis of double-blind, placebo-controlled trials of sublingual immunotherapy (SLIT) indicated that SLIT showed clinical efficacy and had a treatment benefit of approximately half of that achieved by subcutaneous allergen SIT57. Sustained disease-modifying effects of this type of therapy have been shown in large-scale randomized, double-blind, placebo-controlled trials in adults and in children5,58. Although the magnitudes of the changes in most clinical and immunological parameters have been modest or no changes have been observed in response to SLIT, the immunological mechanisms of SLIT seem to be similar to those of subcutaneous allergen SIT. The reduced treatment benefits and modest changes in immunological markers observed with SLIT suggest that there is still room to improve this method of vaccine delivery. Multiple mechanisms of immune tolerance are induced by SLIT that involve Treg cells, IL-10 production and increased numbers of sublingual FOXP3-expressing T cells. In addition, increased concentrations of blocking IgG4 and of IgA antibodies that show inhibitory activity for IgE-facilitated binding of allergens to B cells have been observed after SLIT, similar to what is seen after subcutaneous allergen SIT59,60. Recently, allergen-specific FOXP3+ Treg cells have been found in human lingual and palatine tonsils in humans, and these cells may participate in oral allergen tolerance and SLIT61. It is possible that sugar-modified antigens can be used to induce oral tolerance, and a C-type lectin receptor, SIGNR1 (also known as CD209b), has recently been shown to condition dendritic cells to induce tolerance in the gastrointestinal lamina propria in a model of food-induced anaphylaxis62.
Recently the intralymph node and epicutaneous routes of vaccine delivery have been tested. Both these routes showed a similar efficacy as subcutaneous immunotherapy injections in the treatment of grass pollen allergy, but fewer applications and lower total doses of allergen were required using these two routes63,64. Intralymphatic vaccines have been shown to induce T cell responses that are associated with strong cytotoxic activity and IFN-γ production, which are key in long-term protection against viral infections and tumors.
The fifth strategy is the fusion of allergens to immune modifiers. FcγRIIb is an immune tyrosine-based inhibitory motif-containing receptor65. The coaggregation of FcɛRI and FcγRIIb inhibits FcɛRI signaling, so one strategy that has been tested in the treatment of allergy is the fusion of FcγRIIb to allergens to downregulate downstream allergen-specific immune responses. In a similar approach, the fusion of allergens to human Fcγ suppressed allergen-induced degranulation of basophils and mast cells by crosslinking Fcγ and FcɛRI65,66. Recently, the major cat allergen Fel d 1 was cloned and expressed together with a human immunodeficiency virus protein, TAT-derived membrane translocation domain, and a truncated peptide of the invariant chain (modular antigen translocation (MAT)-Fel d 1)67. This MAT–Fel d 1 vaccine is efficiently internalized and potently presented to T cells by antigen-presenting cells, and the vaccine induces T cell responses at doses that are approximately 100 times lower than those at which the native allergens are present. In a double-blind, placebo-controlled clinical trial, the MAT–Fel d 1 vaccine in alum adjuvant was administered in three increasing doses (1 μg, 3 μg and 10 μg) into the inguinal lymph nodes of patients with cat allergies at 4-week intervals. The vaccine showed a good safety profile, and after treatment, individuals who were allergic to cats became clinically tolerant to nasal challenge of cat dander extract, which was observed in parallel with increased concentrations of serum IgG4 in these individuals68.
In addition to physical fusion, conventional and new methods of allergen SIT may also be combined with immune-response modifiers. For example, a monoclonal antibody (mAb) to IgE combined with allergen SIT has been evaluated in several studies69, and this combination treatment resulted in a significant decrease in the risk of anaphylaxis caused by rush immunotherapy (which uses rapid dose increases to reach the maintenance therapeutic dose as quickly as possible) and improved rescue medication scores (therefore decreasing the need for a rescue medication to suppress the symptoms) of allergen SIT with a good safety profile69,70. Combination strategies with biological immune-response modifiers are expected to substantially expand the treatment scope of allergen SIT. Although there has been some progress in improving the efficacy and safety of allergen SIT, which is the only approach currently being investigated that could cure allergic diseases, further modifications could still be made to this mode of therapy, which are hoped to improve the application of SIT to allergic diseases as well as to other diseases related to immune dysregulation.
Biological immunotherapeutics in allergy and asthma
Developments in understanding the immunological mechanisms of allergy and asthma have enabled the identification of many potential therapeutic targets (Box 3). Because of the complexity and redundancy of the immune mechanisms that are involved in asthma and allergy, it is possible that the use of a single biological might be effective in specific subgroups of asthma and atopic dermatitis, such as for well-defined endotypes that can identify those individuals who will benefit most from a particular treatment. Therefore, many new treatments that target a single mediator or receptor are currently in clinical development or preclinical investigation (Table 3). However, given the complex clinical spectrum of these diseases, it is improbable that any of these biologicals alone will have a major effect in a clinical trial testing a population of individuals that are heterogeneous for the allergic disease being studied or for asthma who have not been classified into specific phenotypes or endotypes. When a new drug is developed to target a very specific molecular mechanism, it is expected that it will function only in a specific asthma endotype. It is anticipated that endotype-tailored treatment (known as 'stratified medicine') could lead to measurable improvements in the health economics of asthma, but more importantly, it may enable disease management to be optimized for individual patients. Currently, combinations of biologicals or the sequential use of two or more biologicals are being considered as viable therapeutic strategies, as reported recently in a study that used a treatment combining mAb to IgE and mAb to B cell CD20 (ref. 71). However, the combination of two biologicals is prohibitively expensive, and it is practically impossible to combine biologicals in a clinical trial setting before one of them gets full approval. This problem needs to be solved before combination strategies of two unapproved biologicals can be developed, which may take several years.
Treatment strategies focused on IgE
Several clinical studies to test different humanized mAbs that bind to the Fc portion of the IgE molecule have been performed, and one of the mAbs tested, omalizumab, was shown to be an effective treatment in patients with poorly controlled, moderate to severe allergic asthma or allergic rhinitis72,73,74. Omalizumab decreases the serum concentration of free (unbound) IgE and the expression of the high-affinity IgE receptor FcɛRI on several cell types72. Soluble immune complexes of IgE and mAb to IgE are subsequently cleared by the reticuloendothelial system. Omalizumab binds to the same site that IgE molecules use to attach to FcɛRI and cannot crosslink cell-surface–expressed IgE75.
Clinical studies of mAb to IgE have improved our understanding of the multiple roles of IgE in the development of the allergen-specific immune response and inflammation. Omalizumab reduces the expression of FcɛRI on basophils, dendritic cells and monocytes within 7 d of administration72. Omalizumab treatment reduces the number of eosinophils in the blood of individuals with seasonal allergic rhinitis and asthma, as well as the number of sputum eosinophils in patients with asthma. In addition to its effects on mast cells and basophils that are related to decreasing the concentrations of free serum IgE, therapy with mAb to IgE causes a rapid decrease in FcɛRI expression on the surface of dendritic cells, suggesting that circulating IgE regulates the expression of this molecule on dendritic cells76. A significant number of patients treated with omalizumab who had allergic asthma and rhinitis had a positive response to treatment and were able to decrease or discontinue their use of inhaled corticosteroids73. Studies of therapies using mAb to IgE in children have been limited, but a recent study showed that children, adolescents and young adults with atopic asthma living in the inner city who were treated with omalizumab had reductions in the number of asthmatic episodes, particularly if they were first sensitized to cockroach antigens74. However, two current caveats to the use of treatment with mAb to IgE are its unexpected efficacy in some cases without any reasonable explanation of the underlying molecular mechanisms, as well as considerations of cost effectiveness. In a recent study, treatment with omalizumab reduced histamine release from peripheral blood leukocytes stimulated with cat allergen in vitro; however, paradoxically, histamine release increased twofold after omalizumab-mediated basophil stimulation77. The cost effectiveness of omalizumab has been evaluated and has been shown to be low in patients with moderate to severe allergic asthma compared to the clinical benefits of the drug. Given this evaluation, one possible approach to use omalizumab is a relatively late treatment choice as an add-on therapy78.
Although there have been no detailed double-blind, placebo-controlled studies on the subject, several reports have suggested that treatment with mAb to IgE is also beneficial for patients suffering from other IgE-related conditions, such as atopic dermatitis, peanut allergy, latex sensitivity, chronic urticaria or allergic bronchopulmonary aspergillosis. In allergic bronchopulmonary aspergillosis, omalizumab has been shown to eliminate the need for treatment with steroids79. Treatment with mAb to IgE has been reported to be effective in several cases of recurrent idiopathic angioedema80 as well as in unprovoked anaphylaxis in patients with systemic mastocytosis81. In most patients with Churg-Strauss syndrome, a type of autoimmune vasculitis, severe asthma and upper respiratory symptoms persist, requiring continuous therapy. Treatment with mAb to IgE was shown to improve asthma and decrease the eosinophil blood count in individuals with this syndrome82.
Cytokine inhibitors
The inflammatory processes that underlie asthma and allergy are coordinated by a cytokine network. Modulating this network using new biological molecules has continuously been attempted in clinical trials for almost two decades. Some of these studies have been unsuccessful, and some trials have had to be discontinued, even for agents that have been approved for other indications, suggesting that there are substantial hurdles to be overcome for the clinical use of cytokine inhibitors to treat asthma and allergy. Some cytokines show redundancy in their ability to generate or suppress TH cell subsets and TReg cells in addition to their roles in disease pathogenesis, which may indicate that agents targeting a single cytokine may not be effective. Despite this redundancy, therapies using cytokine inhibitors may provide a way to elucidate the role of individual cytokines in the pathogenesis of human disease, although it should also be acknowledged that the complexity of the inflammatory networks might make these individual roles difficult to decipher. In addition, some of these biological modifiers could be possible candidates to augment the effects of allergen SIT in combination strategies.
Strategies using mAb to IL-2.
It is generally thought that strategies that block IL-2 pathways could adversely affect CD25+FOXP3+ TReg cell populations, which also rely on IL-2 signaling for their expansion and survival. In addition to promoting the proliferation and survival of recently activated effector T cells, IL-2 is also key in TReg cell homeostasis, thymic development and suppressive function of TReg cells. Daclizumab is a humanized monoclonal antibody that binds to the IL-2 receptor α chain CD25 on activated lymphocytes, inhibits IL-2–induced proliferation and reduces the production of TH2 and TH1 cell cytokines by activated T cells. A phase 2 study in patients with moderate to severe asthma showed that daclizumab improved pulmonary function, reduced asthma symptoms and the need for 'rescue' medication, increased the intervals between asthma exacerbations and reduced blood eosinophilia concentrations and the amount of serum eosinophil cationic protein83,84. The therapeutic benefit of daclizumab treatment in patients with more refractory asthma was more pronounced. The issue of whether blocking IL-2 signaling adversely affects Treg cell development and function was assessed in a heart transplantation study, which showed that daclizumab treatment did not interfere with Treg cell generation and had a beneficial effect on heart allograft survival85.
Strategies targeting IL-4.
IL-4 induces IgE isotype switching and promotes the differentiation of naive CD4+ T lymphocytes into TH2 cells, as well as the subsequent release of increased amounts IL-4, IL-5 and IL-13 by TH2 cells. IL-4 may have a role in the expression of adhesion molecules and chemokines that promote the migration of TH2 cells and eosinophils into tissues, as well as in the development of myeloid dendritic cells. Several clinical trials have evaluated the efficacy and safety of a recombinant human soluble IL-4 receptor (IL-4R) and mAbs to IL-4, but no significant improvements were noted in symptoms or asthma exacerbations using treatments with either of these two agents. These findings suggested that targeting IL-4 on its own does not seem to have clinical benefit and led to the discontinuation of the further exploration of therapies that target IL-4. One of the primary reasons why this approach failed could be the high redundancy of IL-4 and IL-13 signaling. Therefore, it may be necessary to block both IL-4 and IL-13 to see any efficacy for this type of therapy. The receptor of IL-4 and IL-13 consists of two chains, IL-4Rα (CD124) and the common γ chain. IL-13R consists of two subunits, IL-13Rα1 and IL-13Rα2, and IL-4 and IL-13 signaling occurs through the receptor complex type II that consists of the IL-4Rα and IL-13Rα (ref. 86). A mutated IL-4 protein that inhibits the effects of both IL-4 and IL-13 through its ability to block IL-4Rα has been generated, and clinical studies testing therapies using this agent are currently under way87. A mAb to IL-4Rα that blocks both the IL-4 and IL-13 receptors may have the same potential as the mutant IL-4 protein for blocking the effects of both of these cytokines88.
Strategies using mAb to IL-13.
IL-13 is as crucial as IL-4 in the production of IgE, and studies have indicated that this cytokine induces airway hyperresponsiveness, epithelial activation, mucous production, chemokine secretion and changes in airway remodeling. Studies in mouse and monkey models of asthma have shown that treatment with mAb to IL-13 leads to a reduction in lung inflammation. Various humanized mAbs specific for human IL-13 have been, and are currently, in development. A phase 1 clinical trial showed that increasing single doses of intravenously administered mAbs to IL-13 in mild asthma were well tolerated at all doses, and there were no safety concerns associated with this treatment89. A recently reported phase 2 trial of a mAb to IL-13 (lebrikizumab) provided convincing evidence that this biological could be effective in treating certain disease subgroups that have been selected on the basis of a biomarker. Notably, this mAb to IL13 showed significant efficacy in a patient group that expressed high amounts of the protein periostin but did not have any effect in patients with low amounts of periostin, which is what would be predicted based on knowledge of the functions of periostin. IL-13 induces periostin, and periostin has a role in type I collagen synthesis, fibrosis and remodeling in the lungs90. Therefore, reducing the amount of free serum IL-13 or blocking IL-13 signaling in individuals with low amounts of periostin would not be expected to have any beneficial effect, as alternative pathogenic pathways that are not downstream of periostin are probably operating in these individuals.
Strategies using mAb to IL-5.
Eosinophilia in the lung and circulation are hallmarks of asthma, and IL-5 is a key cytokine for eosinophil differentiation and survival. For this reason, there has been intensive focus on strategies using mAb to IL-5 for the treatment of asthma. A decrease in the number of serum eosinophils was noted in subjects with severe persistent asthma treated for 90 d with reslizumab, a humanized mAb to IL-5 (ref. 91). No significant sustained changes in forced expiratory volume in 1 s (FEV1), asthma symptom scores or the percentage of sputum eosinophils were noted with any dose of reslizumab. Studies in patients with mild atopic asthma showed that the humanized mAb to IL-5 mepolizumab decreased extracellular matrix protein remodeling and the percentage of airway eosinophils. Treatment with mepolizumab decreased serum eosinophilia by 100%, whereas it decreased airway eosinophils to a lesser extent, by 55%92. Similar to reslizumab, mepolizumab treatment did not affect the clinical measures of asthma, including airway hyperresponsiveness, FEV1 and peak flow measures. Recent trials of mAb to IL-5 in patients with prominent sputum eosinophilia and severe asthma that is refractory to existing therapies showed that the treatment significantly reduced asthma exacerbations93. Notably, in these trials, only 5% of the patients showed this clinical improvement and fit into a group that had eosinophils comprising greater than 3% of their total sputum cells, suggesting that it may be possible to define an asthma endotype that is responsive to mAb to IL-5, similar to the endotypes observed for periostin and lebrikizumab. If, similar to mAb to IL-5 and IL-13, a drug is only successful for a small percentage of affected individuals, it is now a matter of debate whether this factor would influence the likelihood of approval of the drug. In addition, treatment with mAb to IL-5 has shown promising results in patients with hypereosinophilic syndrome94, nasal polyps95 and eosinophilic esophagitis96.
Strategies to target tumor necrosis factor-α.
Tumor necrosis factor-α (TNF-α) is a proinflammatory cytokine that has been implicated in many aspects of the airway pathology in asthma, particularly in refractory and severe cases. Clinical trials have shown improvement in lung function, airway hyperresponsiveness and quality-of-life symptom scores and reduction in the exacerbation frequency in patients with asthma who were treated with mAb to TNF97,98. Treatment with etanercept, a soluble TNF receptor (TNFR) fusion protein, also improved airway hyperresponsiveness, FEV1 values and overall asthma symptom scores in a phase 2 study for moderate to severe and refractory asthma98. Although promising results have been obtained using these agents, because of their association with an increased risk of infection, particularly tuberculosis and malignancy, strategies that target TNF will probably not be enthusiastically pursued for general treatment of asthma.
The TNF superfamily consists of many membrane-bound and soluble proteins with proinflammatory effects in the innate and adaptive immune responses. A recent preclinical study showed that using a fusion protein to block the TNF ligand superfamily member 14, LIGHT, which is expressed on many immune cells such as activated T cells, monocytes and macrophages, reduced airway hyperresponsiveness, lung fibrosis and smooth muscle hyperplasia in mouse models of chronic asthma, despite showing little effect on airway eosinophilia in these models. LIGHT may be therapeutically targeted to prevent asthma-related airway remodeling, as LIGHT-deficient mice also showed an impairment in fibrosis and in smooth muscle accumulation99. However, given the safety concerns that already exist with therapies targeting TNF-α, it is probable that similar issues also exist for targeting LIGHT and other TNF-family molecules.
Other approaches using cytokine inhibitors.
A number of other therapeutic approaches to target cytokines are in various stages of preclinical and clinical development and testing. A newly described population of T cells called TH9 cells, which produce IL-9 and IL-10, have been proposed to have a role in allergic inflammation100. IL-9 is secreted by CD4+ TH2 cells, eosinophils, mast cells and neutrophils. It inhibits cytokine production by TH1 cells, induces chemokine and mucous secretion by bronchial epithelial cells and promotes the proliferation of mast cells. In a recent clinical trial, an mAb to IL-9 did not to meet the clinical endpoints; however, it did show an acceptable safety profile, and findings from the trial were suggestive of clinical activity in subjects with mild to moderate asthma101. Approaches that target IL-17, IL-25, IL-31 or IL-33 may be useful in some endotypes of asthma and are currently in preclinical development102,103,104,105. However, positive results from preclinical studies do not necessarily mean that these approaches will work in the setting of human asthma, and in addition, specific endotypes that might be targeted by these cytokines have not been elucidated in humans.
Thymic stromal lymphopoietin (TSLP) is an essential cytokine for the initiation and development of allergic inflammation. Treatment with TSLP diverted airway tolerance against ovalbumin to TH2 sensitization and inhibited the generation of ovalbumin-specific inducible Treg cells in an asthma-like mouse model106. A soluble TSLP antagonist, TSLPR immunoglobulin, was shown to reduce the severity of allergic disease by blocking TSLP signaling and by regulating pulmonary dendritic cells. The tumor necrosis factor receptor superfamily member 4 (OX40) ligand (OX40L) is a key regulator of TSLP-mediated TH2 responses. Preclinical studies in a mouse model of asthma with an mAb that blocked OX40 signaling showed promising results, with substantial inhibition of the immune responses induced by TSLP in the lung and skin, including decreased TH2 cell infiltration, cytokine secretion and IgE production. OX40L-blocking antibodies have also been shown to inhibit antigen-driven TH2 inflammation in mouse and nonhuman primate models of asthma107.
Targeting cell adhesion, co-stimulation and chemotaxis
Chemokine receptors, which are G-protein–coupled receptors, can be therapeutically targeted by small molecules. In contrast, approaches to target cytokines and cytokine receptors using humanized blocking mAbs, soluble receptors and mutant cytokines are currently being investigated. As chemokines are involved in stimulating the migration of TH2 cells and eosinophils into inflamed tissues, targeting these molecules and their receptors may be a useful strategy for the treatment of tissue inflammation in asthma and allergy. It is probable that a subset of chemokines are the key targets in asthma and are selective for eosinophils108. Selective eosinophil recruitment can be induced by chemokines that bind to chemokine (C-C motif) receptor 3 (CCR3) on eosinophils, such as eotaxin-1 (chemokine (C-C motif) ligand 1 (CCL11)), eotaxin-2 (CCL24), eotaxin-3 (CCL26), RANTES (CCL5) and MCP-4 (CCL13). Mice deficient in one of these chemokines have impaired eosinophil trafficking to the skin, airway and/or gut109.
A key role for chemokine (C-X-C motif) receptor 2 (CXCR2) has been identified in asthma, chronic obstructive pulmonary disease and fibrotic pulmonary disorders. After CXCR2 inhibition, the angiogenesis and collagen deposition caused by lung injury is still observed, but the pulmonary damage induced by neutrophils, antigen or irritant-induced goblet cell hyperplasia is inhibited110. These features are common in inflammatory and fibrotic disorders of the lung. Clinical trials to evaluate small-molecule CXCR2 antagonists in chronic obstructive pulmonary disease, asthma and cystic fibrosis have been completed and have suggested that these molecules may be promising in the treatment of these conditions110.
The prostaglandin and chemotactic receptors DP1 and CRTH2 (also known as DP2) have key roles in the development and maintenance of allergy. Studies with DP1 and CRTH2 antagonists have suggested that they could have a crucial role in modulating aspects of allergic diseases that are resistant to current therapies111,112. Prostaglandin D2 (PGD2) has a dominant role in mediating mast-cell–dependent activation of TH2 lymphocytes, which is also mediated by CRTH2 (ref. 112). PGD2 produced by mast cells might link the early and late phase allergic responses, and the antagonism of PGD2 is an attractive target for therapeutic intervention111.
In people with asthma, a fraction of CD4+ T cells express chemokine (C-X3-C motif) receptor 1 (CX3CR1), the receptor for CX3CL1. After allergen challenge, CX3CL1 expression is increased in the airway smooth muscle, lung endothelium and epithelium, and therefore this chemokine might be a target for immune modulation113. Consistent with this, wild-type mice treated with CX3CR1-blocking reagents and CXCR1-deficient mice show reduced inflammation in the lungs after allergen sensitization and challenge113.
Biologicals in atopic dermatitis
A subgroup of patients with severe atopic dermatitis requires systemic immunomodulatory treatment. Clinical studies of omalizumab, efalizumab (a humanized antibody to CD11a), infliximab, adalimumab and etanercept (agents that target TNF-α signaling) and rituximab have been performed with varying results and safety profiles. Efalizumab inhibits T cell activation and thereby impairs the recruitment of T cells into the skin. A retrospective analysis of 11 of the patients in the efalizumab study showed that only 2 had a positive outcome. Nine patients stopped treatment because of a progression of atopic dermatitis or a lack of any clinical effect114. In the study, in the individuals who showed disease progression after treatment with efalizumab, it was not clear whether this progression was a result of side effects of the drug or was a natural progression of the disease.
TNF-α plays a part in inflammation and keratinocyte apoptosis, leading to eczema in atopic dermatitis115. Infliximab, a chimeric mAb to TNF-α, has been used to treat sporadic cases of atopic dermatitis and has also been tested in an open, prospective pilot study of nine patients with atopic dermatitis that was refractory to conventional therapy97. The results from the treated patients and the pilot study suggest that infliximab can be used as an additional therapeutic option for refractory severe atopic dermatitis. Etanercept, which is a soluble TNF receptor, has been used as a treatment option in two patients with severe, chronic atopic dermatitis and led to clinically significant improvements in skin symptoms. These preliminary results suggest that etanercept therapy may be beneficial for atopic dermatitis, particularly in chronic variants associated with lichenification, which is characterized by a thickening and hardening of the skin as a result of the disease116. Similar to asthma, safety concerns for treatments that target TNF-α may be a caveat in their use for atopic dermatitis.
Omalizumab has been used in selected patients suffering from both atopic eczema and allergic asthma or from atopic eczema alone117. Improvements seen after omalizumab treatment in these patients seem to be a result of more immune regulatory mechanisms rather than of simple neutralization of IgE by omalizumab. Recently, a randomized, placebo-controlled, double-blind pilot study of omalizumab was performed in 20 patients, which showed that the drug did not improve the clinical course in patients with chronic atopic dermatitis118; however, it did improve the results of skin prick tests and atopy patch tests.
Rituximab is a chimeric mAb to CD20 that efficiently eliminates circulating B cells. B cells that have class switched to produce IgE have long been suggested to have a role in atopic dermatitis because highly elevated concentrations of serum IgE are commonly observed in patients with generalized extrinsic atopic dermatitis. A pilot study with six patients was conducted in which rituximab was intravenously administered in two doses of 1,000 mg given 2 weeks apart119. All six patients showed an improvement of their skin symptoms within 4–8 weeks of the start of treatment, suggesting that rituximab may be a promising treatment option for patients with severe atopic dermatitis. However, these initial results have not been confirmed in two patients treated with rituximab who did not show amelioration of disease symptoms after treatment120. Therefore, larger randomized studies will be required to more rigorously test the effects and mechanisms of immune regulation induced by omalizumab and rituximab in the treatment of atopic dermatitis.
Conclusions and future perspectives
A better understanding of the molecular mechanisms involved in asthma and allergy is hoped to spur the development of new treatment modalities for these conditions. Currently, allergen SIT is the only available curative treatment for allergic diseases, as it can induce long-term allergen-specific immune tolerance using multiple mechanisms. Despite the benefits of this therapy for most treated individuals, not everyone improves; life-threatening side effects can occur, recovery may not be permanent and the duration of the treatment is long. Therefore, new vaccines, as well as reliable biomarkers to select patients who may have a good clinical response, are crucial. Notably, allergen-SIT–based curative approaches may also be promising for the prevention of allergic disease, although the challenges accompanying the prevention approaches might differ to those for treatment. The major challenges for prevention include the requirement for very early intervention, safety problems for pediatric usage of vaccines and the lack of early biomarkers to predict who will develop allergies and to which particular allergen.
There is also a strong rationale for the development of biological immune response modifiers. The future of this area of research should be exciting, as advances in immunology and bioengineering are being applied, allowing for the optimized design of biologicals to improve the clinical efficacy and the feasibility of clinical grade production of these agents. Patient-specific treatments that depend on the development of new diagnostic biomarkers that can discriminate between distinct disease endotypes are also on the horizon. The analysis of large datasets of adults and children with severe allergic diseases and asthma is now beginning to allow for the identification of distinct disease phenotypes. It will be crucial to identify biomarkers and use systems biology to predict clinical responses for various subphenotypes of the diseases. This area requires more research for personalized therapy to become feasible because only a small proportion of patients with severe allergic diseases (for example, asthma) are likely to respond to a single biological. In particular, the combination of immune response modifiers with allergen SIT might provide a way for efficient immunomodulation of allergic diseases. Moreover, a greater understanding of the underlying disease mechanisms in allergic diseases could bring the possibility of cure in a larger patient population closer to reality.
References
Galli, S.J., Tsai, M. & Piliponsky, A.M. The development of allergic inflammation. Nature 454, 445–454 (2008).
Lötvall, J. et al. Asthma endotypes: a new approach to classification of disease entities within the asthma syndrome. J. Allergy Clin. Immunol. 127, 355–360 (2011).
Wenzel, S. Asthma phenotypes: the evolution from clinical to molecular approaches. Nat. Med. 18, aaa–bbb (2012).
Bousquet, J. et al. Unmet needs in severe chronic upper airway disease (SCUAD). J. Allergy Clin. Immunol. 124, 428–433 (2009).
Durham, S.R. et al. Long-term clinical efficacy in grass pollen-induced rhinoconjunctivitis after treatment with SQ-standardized grass allergy immunotherapy tablet. J. Allergy Clin. Immunol. 125, 131–138 (2010).
Akdis, M. & Akdis, C.A. Therapeutic manipulation of immune tolerance in allergic disease. Nat. Rev. Drug Discov. 8, 645–660 (2009).
Meiler, F. et al. In vivo switch to IL-10-secreting T regulatory cells in high dose allergen exposure. J. Exp. Med. 205, 2887–2898 (2008).
Platts-Mills, T.A. & Woodfolk, J.A. Allergens and their role in the allergic immune response. Immunol. Rev. 242, 51–68 (2011).
Romano, A., Torres, M.J., Castells, M., Sanz, M.L. & Blanca, M. Diagnosis and management of drug hypersensitivity reactions. J. Allergy Clin. Immunol. 127, S67–S73 (2011).
Eberlein-König, B., Ullmann, S., Thomas, P. & Przybilla, B. Tryptase and histamine release due to a sting challenge in bee venom allergic patients treated successfully or unsuccessfully with hyposensitization. Clin. Exp. Allergy 25, 704–712 (1995).
Plewako, H. et al. Basophil interleukin 4 and interleukin 13 production is suppressed during the early phase of rush immunotherapy. Int. Arch. Allergy Immunol. 141, 346–353 (2006).
Akdis, C.A., Blesken, T., Akdis, M., Wuthrich, B. & Blaser, K. Role of interleukin 10 in specific immunotherapy. J. Clin. Invest. 102, 98–106 (1998).
Jutel, M. et al. IL-10 and TGF-β cooperate in the regulatory T cell response to mucosal allergens in normal immunity and specific immunotherapy. Eur. J. Immunol. 33, 1205–1214 (2003).
Pepper, M. & Jenkins, M.K. Origins of CD4+ effector and central memory T cells. Nat. Immunol. 12, 467–471 (2011).
Ling, E.M. et al. Relation of CD4+CD25+ regulatory T-cell suppression of allergen-driven T-cell activation to atopic status and expression of allergic disease. Lancet 363, 608–615 (2004).
Radulovic, S., Jacobson, M.R., Durham, S.R. & Nouri-Aria, K.T. Grass pollen immunotherapy induces Foxp3-expressing CD4+ CD25+ cells in the nasal mucosa. J. Allergy Clin. Immunol. 121, 1467–1472 (2008).
Aslam, A., Chan, H., Warrell, D.A., Misbah, S. & Ogg, G.S. Tracking antigen-specific T-cells during clinical tolerance induction in humans. PLoS ONE 5, e11028 (2010).
Campbell, J.D. et al. Peptide immunotherapy in allergic asthma generates IL-10–dependent immunological tolerance associated with linked epitope suppression. J. Exp. Med. 1535–1547 (2009).
Chung, Y. et al. Follicular regulatory T cells expressing Foxp3 and Bcl-6 suppress germinal center reactions. Nat. Med. 17, 983–988 (2011).
Linterman, M.A. et al. Foxp3+ follicular regulatory T cells control the germinal center response. Nat. Med. 17, 975–982 (2011).
Balachandran, V.P. et al. Imatinib potentiates antitumor T cell responses in gastrointestinal stromal tumor through the inhibition of IDO. Nat. Med. 17, 1094–1100 (2011).
Akdis, M. et al. Immune responses in healthy and allergic Individuals are characterized by a fine balance between allergen-specific T regulatory 1 and T helper 2 cells. J. Exp. Med. 199, 1567–1575 (2004).
Orihara, K. et al. Circulating Foxp3+CD4+ cell numbers in atopic patients and healthy control subjects. J. Allergy Clin. Immunol. 120, 960–962 (2007).
Klunker, S. et al. Transcription factors RUNX1 and RUNX3 in the induction and suppressive function of Foxp3+ inducible regulatory T cells. J. Exp. Med. 206, 2701–2715 (2009).
Karlsson, M.R., Rugtveit, J. & Brandtzaeg, P. Allergen-responsive CD4+CD25+ regulatory T cells in children who have outgrown cow's milk allergy. J. Exp. Med. 199, 1679–1688 (2004).
Jartti, T. et al. Association between CD4+CD25high T cells and atopy in children. J. Allergy Clin. Immunol. 120, 177–183 (2007).
Akdis, C.A. et al. Epitope specific T cell tolerance to phospholipase A2 in bee venom immunotherapy and recovery by IL-2 and IL-15 in vitro. J. Clin. Invest. 98, 1676–1683 (1996).
Van Ree, R. et al. Measurement of IgE antibodies against purified grass pollen allergens (Lol p 1, 2, 3 and 5) during immunotherapy. Clin. Exp. Allergy 27, 68–74 (1997).
Meiler, F., Klunker, S., Zimmermann, M., Akdis, C.A. & Akdis, M. Distinct regulation of IgE, IgG4 and IgA by T regulatory cells and toll-like receptors. Allergy 63, 1455–1463 (2008).
Radbruch, A. et al. Competence and competition: the challenge of becoming a long-lived plasma cell. Nat. Rev. Immunol. 6, 741–750 (2006).
Jutel, M. et al. Allergen-specific immunotherapy with recombinant grass pollen allergens. J. Allergy Clin. Immunol. 116, 608–613 (2005).
James, L.K. et al. Long-term tolerance after allergen immunotherapy is accompanied by selective persistence of blocking antibodies. J. Allergy Clin. Immunol. 127, 509–516 (2011).
van der Neut Kolfschoten, M. et al. Anti-inflammatory activity of human IgG4 antibodies by dynamic Fab arm exchange. Science 317, 1554–1557 (2007).
van der Zee, J.S., van Swieten, P. & Aalberse, R.C. Inhibition of complement activation by IgG4 antibodies. Clin. Exp. Immunol. 64, 415–422 (1986).
Pierkes, M. et al. Decreased release of histamine and sulfidoleukotrienes by human peripheral blood leukocytes after wasp venom immunotherapy is partially due to induction of IL-10 and IFN-γ production of T cells. J. Allergy Clin. Immunol. 103, 326–332 (1999).
Marshall, J.S., Leal-Berumen, I., Nielsen, L., Glibetic, M. & Jordana, M. Interleukin (IL)-10 inhibits long-term IL-6 production but not preformed mediator release from rat peritoneal mast cells. J. Clin. Invest. 97, 1122–1128 (1996).
Gri, G. et al. CD4+CD25+ regulatory T cells suppress mast cell degranulation and allergic responses through OX40–OX40L interaction. Immunity 29, 771–781 (2008).
Biggs, L. et al. Evidence that vitamin D(3) promotes mast cell-dependent reduction of chronic UVB-induced skin pathology in mice. J. Exp. Med. 207, 455–463 (2010).
Galli, S.J., Grimbaldeston, M. & Tsai, M. Immunomodulatory mast cells: negative, as well as positive, regulators of immunity. Nat. Rev. Immunol. 8, 478–486 (2008).
Turner, J.D. et al. CD4+CD25+ regulatory cells contribute to the regulation of colonic Th2 granulomatous pathology caused by schistosome infection. PLoS Negl. Trop. Dis. 5, e1269 (2011).
Plewako, H. et al. A follow-up study of immunotherapy-treated birch-allergic patients: effect on the expression of chemokines in the nasal mucosa. Clin. Exp. Allergy 38, 1124–1131 (2008).
Akdis, C.A. & Blaser, K. Bypassing IgE and targeting T cells for specific immunotherapy of allergy. Trends Immunol. 22, 175–178 (2001).
Kussebi, F. et al. A major allergen gene-fusion protein for potential usage in allergen-specific immunotherapy. J. Allergy Clin. Immunol. 115, 323–329 (2005).
Karamloo, F. et al. Prevention of allergy by a recombinant multi-allergen vaccine with reduced IgE binding and preserved T cell epitopes. Eur. J. Immunol. 35, 3268–3276 (2005).
Niederberger, V. et al. Vaccination with genetically engineered allergens prevents progression of allergic disease. Proc. Natl. Acad. Sci. USA 101 (suppl. 2), 14677–14682 (2004).
Norman, P.S. et al. Treatment of cat allergy with T cell reactive peptides. Am. J. Respir. Crit. Care Med. 154, 1623–1628 (1996).
Müller, U. et al. Successful immunotherapy with T cell epitope peptides of bee venom phospholipase A2 induces specific T cell anergy in bee sting allergic patients. J. Allergy Clin. Immunol. 101, 747–754 (1998).
Oldfield, W.L., Larche, M. & Kay, A.B. Effect of T-cell peptides derived from Fel d 1 on allergic reactions and cytokine production in patients sensitive to cats: a randomised controlled trial. Lancet 360, 47–53 (2002).
Worm, M. et al. Development and preliminary clinical evaluation of a peptide immunotherapy vaccine for cat allergy. J. Allergy Clin. Immunol. 127, 89–97 (2011).
Pree, I. et al. Analysis of epitope-specific immune responses induced by vaccination with structurally folded and unfolded recombinant Bet v 1 allergen derivatives in man. J. Immunol. 179, 5309–5316 (2007).
Creticos, P.S. et al. Immunotherapy with a ragweed-toll-like receptor 9 agonist vaccine for allergic rhinitis. N. Engl. J. Med. 355, 1445–1455 (2006).
Kündig, T.M. et al. Der p 1 peptide on virus-like particles is safe and highly immunogenic in healthy adults. J. Allergy Clin. Immunol. 117, 1470–1476 (2006).
Thunberg, S. et al. Prolonged antigen-exposure with carbohydrate particle based vaccination prevents allergic immune responses in sensitized mice. Allergy 64, 919–926 (2009).
Niespodziana, K. et al. A hypoallergenic cat vaccine based on Fel d 1–derived peptides fused to hepatitis B PreS. J. Allergy Clin. Immunol. 127, 1562–1570 (2011).
Rosewich, M. et al. Tolerance induction after specific immunotherapy with pollen allergoids adjuvanted by monophosphoryl lipid A in children. Clin. Exp. Immunol. 160, 403–410 (2010).
Hammad, H. et al. House dust mite allergen induces asthma via Toll-like receptor 4 triggering of airway structural cells. Nat. Med. 15, 410–416 (2009).
Wilson, D.R., Lima, M.T. & Durham, S.R. Sublingual immunotherapy for allergic rhinitis: systematic review and meta-analysis. Allergy 60, 4–12 (2005).
Bufe, A. et al. Safety and efficacy in children of an SQ-standardized grass allergen tablet for sublingual immunotherapy. J. Allergy Clin. Immunol. 123, 167–173 (2009).
Scadding, G.W. et al. Sublingual grass pollen immunotherapy is associated with increases in sublingual Foxp3-expressing cells and elevated allergen-specific immunoglobulin G4, immunoglobulin A and serum inhibitory activity for immunoglobulin E-facilitated allergen binding to B cells. Clin. Exp. Allergy 40, 598–606 (2010).
Allam, J.P. et al. Toll-like receptor 4 ligation enforces tolerogenic properties of oral mucosal Langerhans cells. J. Allergy Clin. Immunol. 121, 368–374 (2008).
Palomares, O. et al. Induction and maintenance of allergen-specific FOXP3+ Treg cells in human tonsils as potential first-line organs of oral tolerance. J. Allergy Clin. Immunol. 129, 510–520 (2012).
Zhou, Y. et al. Oral tolerance to food-induced systemic anaphylaxis mediated by the C-type lectin SIGNR1. Nat. Med. 16, 1128–1133 (2010).
Senti, G. et al. Intralymphatic allergen administration renders specific immunotherapy faster and safer: a randomized controlled trial. Proc. Natl. Acad. Sci. USA 105, 17908–17912 (2008).
Senti, G. et al. Epicutaneous allergen-specific immunotherapy ameliorates grass pollen-induced rhinoconjunctivitis: a double-blind, placebo-controlled dose escalation study. J. Allergy Clin. Immunol. 129, 128–135 (2012).
Zhu, D., Kepley, C.L., Zhang, M., Zhang, K. & Saxon, A. A novel human immunoglobulin Fc γ Fc ɛ bifunctional fusion protein inhibits Fc ɛ RI-mediated degranulation. Nat. Med. 8, 518–521 (2002).
Zhu, D. et al. A chimeric human-cat fusion protein blocks cat-induced allergy. Nat. Med. 11, 446–449 (2005).
Crameri, R., Kundig, T.M. & Akdis, C.A. Modular antigen-translocation as a novel vaccine strategy for allergen-specific immunotherapy. Curr. Opin. Allergy Clin. Immunol. 9, 568–573 (2009).
Senti, G. et al. Intralymphatic immunotherapy with a modular antigen transporter vaccine induces tolerance after only three injections. J. Allergy Clin. Immunol. (in the press).
Massanari, M. et al. Effect of pretreatment with omalizumab on the tolerability of specific immunotherapy in allergic asthma. J. Allergy Clin. Immunol. 125, 383–389 (2010).
Klunker, S. et al. Combination treatment with omalizumab and rush immunotherapy for ragweed-induced allergic rhinitis: Inhibition of IgE-facilitated allergen binding. J. Allergy Clin. Immunol. 120, 688–695 (2007).
Yong, P.F. et al. Rituximab and omalizumab in severe, refractory insulin allergy. N. Engl. J. Med. 360, 1045–1047 (2009).
Lin, H. et al. Omalizumab rapidly decreases nasal allergic response and FcɛRI on basophils. J. Allergy Clin. Immunol. 113, 297–302 (2004).
Casale, T.B. et al. Effect of omalizumab on symptoms of seasonal allergic rhinitis: a randomized controlled trial. J. Am. Med. Assoc. 286, 2956–2967 (2001).
Busse, W.W. et al. Randomized trial of omalizumab (anti-IgE) for asthma in inner-city children. N. Engl. J. Med. 364, 1005–1015 (2011).
Beck, L.A., Marcotte, G.V., MacGlashan, D., Togias, A. & Saini, S. Omalizumab-induced reductions in mast cell Fce psilon RI expression and function. J. Allergy Clin. Immunol. 114, 527–530 (2004).
Prussin, C. et al. Omalizumab treatment downregulates dendritic cell FcɛRI expression. J. Allergy Clin. Immunol. 112, 1147–1154 (2003).
Zaidi, A.K., Saini, S.S. & MacGlashan, D.W. Jr. Regulation of Syk kinase and FcRβ expression in human basophils during treatment with omalizumab. J. Allergy Clin. Immunol. 125, 902–908 (2010).
Wu, A.C., Paltiel, A.D., Kuntz, K.M., Weiss, S.T. & Fuhlbrigge, A.L. Cost-effectiveness of omalizumab in adults with severe asthma: results from the Asthma Policy Model. J. Allergy Clin. Immunol. 120, 1146–1152 (2007).
Zirbes, J.M. & Milla, C.E. Steroid-sparing effect of omalizumab for allergic bronchopulmonary aspergillosis and cystic fibrosis. Pediatr. Pulmonol. 43, 607–610 (2008).
Sands, M.F., Blume, J.W. & Schwartz, S.A. Successful treatment of 3 patients with recurrent idiopathic angioedema with omalizumab. J. Allergy Clin. Immunol. 120, 979–981 (2007).
Carter, M.C. et al. Omalizumab for the treatment of unprovoked anaphylaxis in patients with systemic mastocytosis. J. Allergy Clin. Immunol. 119, 1550–1551 (2007).
Bargagli, E., Madioni, C., Olivieri, C., Penza, F. & Rottoli, P. Churg-Strauss vasculitis in a patient treated with omalizumab. J. Asthma 45, 115–116 (2008).
Nelson, H.S. et al. Daclizumab improves asthma control in patients with refractory asthma. J. Allergy Clin. Immunol. 115 (suppl.), S134 (2005).
Casale, T.B. & Stokes, J.R. Immunomodulators for allergic respiratory disorders. J. Allergy Clin. Immunol. 121, 288–296 (2008).
Vlad, G. et al. Anti-CD25 treatment and FOXP3-positive regulatory T cells in heart transplantation. Transpl. Immunol. 18, 13–21 (2007).
Akdis, M. et al. Interleukins, from 1 to 37, and interferon-γ: receptors, functions, and roles in diseases. J. Allergy Clin. Immunol. 127, 701–721 (2011).
Andrews, A.L., Holloway, J.W., Holgate, S.T. & Davies, D.E. IL-4 receptor alpha is an important modulator of IL-4 and IL-13 receptor binding: implications for the development of therapeutic targets. J. Immunol. 176, 7456–7461 (2006).
Corren, J. et al. A randomized, controlled, phase 2 study of AMG 317, an IL-4Ralpha antagonist, in patients with asthma. Am. J. Respir. Crit. Care Med. 181, 788–796 (2010).
Blanchard, C. et al. Inhibition of human interleukin-13-induced respiratory and oesophageal inflammation by anti-human-interleukin-13 antibody (CAT-354). Clin. Exp. Allergy 35, 1096–1103 (2005).
Corren, J. et al. Lebrikizumab treatment in adults with asthma. N. Engl. J. Med. 365, 1088–1098 (2011).
Kips, J.C. et al. Effect of SCH55700, a humanized anti-human interleukin-5 antibody, in severe persistent asthma: a pilot study. Am. J. Respir. Crit. Care Med. 167, 1655–1659 (2003).
Flood-Page, P.T., Manzies-Gow, A.N., Kay, A.B. & Robinson, D.S. Eosinophil's role remains uncertain as anti-interleukin-5 only partially depletes numbers in asthmatic airway. Am. J. Respir. Crit. Care Med. 167, 199–204 (2003).
Nair, P. et al. Mepolizumab for prednisone-dependent asthma with sputum eosinophilia. N. Engl. J. Med. 360, 985–993 (2009).
Rothenberg, M.E. et al. Treatment of patients with the hypereosinophilic syndrome with mepolizumab. N. Engl. J. Med. 358, 1215–1228 (2008).
Gevaert, P. et al. Mepolizumab, a humanized anti-IL-5 mAb, as a treatment option for severe nasal polyposis. J. Allergy Clin. Immunol. 128, 989–995 (2011).
Straumann, A. et al. Anti-interleukin-5 antibody treatment (mepolizumab) in active eosinophilic oesophagitis: a randomised, placebo-controlled, double-blind trial. Gut 59, 21–30 (2010).
Jacobi, A., Antoni, C., Manger, B., Schuler, G. & Hertl, M. Infliximab in the treatment of moderate to severe atopic dermatitis. J. Am. Acad. Dermatol. 52, 522–526 (2005).
Berry, M.A. et al. Evidence of a role of tumor necrosis factor α in refractory asthma. N. Engl. J. Med. 354, 697–708 (2006).
Doherty, T.A. et al. The tumor necrosis factor family member LIGHT is a target for asthmatic airway remodeling. Nat. Med. 17, 596–603 (2011).
Veldhoen, M. et al. Transforming growth factor-β 'reprograms' the differentiation of T helper 2 cells and promotes an interleukin 9-producing subset. Nat. Immunol. 9, 1341–1346 (2008).
Parker, J.M. et al. Safety profile and clinical activity of multiple subcutaneous doses of MEDI-528, a humanized anti–interleukin-9 monoclonal antibody, in two randomized phase 2a studies in subjects with asthma. BMC Pulm. Med. 11, 14 (2011).
Hellings, P.W. et al. Interleukin-17 orchestrates the granulocyte influx into airways after allergen inhalation in a mouse model of allergic asthma. Am. J. Respir. Cell Mol. Biol. 28, 42–50 (2003).
Rickel, E.A. et al. Identification of functional roles for both IL-17RB and IL-17RA in mediating IL-25-induced activities. J. Immunol. 181, 4299–4310 (2008).
Dillon, S.R. et al. Interleukin 31, a cytokine produced by activated T cells, induces dermatitis in mice. Nat. Immunol. 5, 752–760 (2004).
Kearley, J., Buckland, K.F., Mathie, S.A. & Lloyd, C.M. Resolution of allergic inflammation and airway hyperreactivity is dependent upon disruption of the T1/ST2-IL-33 pathway. Am. J. Respir. Crit. Care Med. 179, 772–781 (2009).
Lei, L., Zhang, Y., Yao, W., Kaplan, M.H. & Zhou, B. Thymic stromal lymphopoietin interferes with airway tolerance by suppressing the generation of antigen-specific regulatory T cells. J. Immunol. 186, 2254–2261 (2011).
Seshasayee, D. et al. In vivo blockade of OX40 ligand inhibits thymic stromal lymphopoietin driven atopic inflammation. J. Clin. Invest. 117, 3868–3878 (2007).
Bromley, S.K., Mempel, T.R. & Luster, A.D. Orchestrating the orchestrators: chemokines in control of T cell traffic. Nat. Immunol. 9, 970–980 (2008).
Bochner, B.S. & Gleich, G.J. What targeting eosinophils has taught us about their role in diseases. J. Allergy Clin. Immunol. 126, 16–25 (2010).
Holz, O. et al. SCH527123, a novel CXCR2 antagonist, inhibits ozone-induced neutrophilia in healthy subjects. Eur. Respir. J. 35, 564–570 (2010).
Angeli, V. et al. Activation of the D prostanoid receptor 1 regulates immune and skin allergic responses. J. Immunol. 172, 3822–3829 (2004).
Barnes, N. et al. A randomized, double-blind, placebo-controlled study of the CRTH2 antagonist OC000459 in moderate persistent asthma. Clin. Exp. Allergy 42, 38–48 (2012).
Mionnet, C. et al. CX3CR1 is required for airway inflammation by promoting T helper cell survival and maintenance in inflamed lung. Nat. Med. 16, 1305–1312 (2010).
Ibler, K. et al. Efalizumab for severe refractory atopic eczema: retrospective study on 11 cases. J. Eur. Acad. Dermatol. Venereol. 24, 837–839 (2010).
Zimmermann, M. et al. TWEAK and TNF-α cooperate in the induction of keratinocyte-apoptosis. J. Allergy Clin. Immunol. 127, 200–207 (2011).
Rullan, P. & Murase, J. Two cases of chronic atopic dermatitis treated with soluble tumor necrosis factor receptor therapy. J. Drugs Dermatol. 8, 873–876 (2009).
Belloni, B. et al. Low-dose anti-IgE therapy in patients with atopic eczema with high serum IgE levels. J. Allergy Clin. Immunol. 120, 1223–1225 (2007).
Heil, P.M., Maurer, D., Klein, B., Hultsch, T. & Stingl, G. Omalizumab therapy in atopic dermatitis: depletion of IgE does not improve the clinical course—a randomized, placebo-controlled and double blind pilot study. J. Dtsch. Dermatol. Ges. 8, 990–998 (2010).
Simon, D., Hosli, S., Kostylina, G., Yawalkar, N. & Simon, H.U. Anti-CD20 (rituximab) treatment improves atopic eczema. J. Allergy Clin. Immunol. 121, 122–128 (2008).
Sedivá, A. et al. Anti-CD20 (rituximab) treatment for atopic eczema. J. Allergy Clin. Immunol. 121, 1515–1516, author reply 1516–1517 (2008).
Asturias, J.A. et al. Engineering of major house dust mite allergens Der p 1 and Der p 2 for allergen-specific immunotherapy. Clin. Exp. Allergy 39, 1088–1098 (2009).
Busse, W.W. et al. Daclizumab improves asthma control in patients with moderate to severe persistent asthma: a randomized, controlled trial. Am. J. Respir. Crit. Care Med. 178, 1002–1008 (2008).
Hart, T.K. et al. Preclinical efficacy and safety of pascolizumab (SB 240683): a humanized anti–interleukin-4 antibody with therapeutic potential in asthma. Clin. Exp. Immunol. 130, 93–100 (2002).
Wenzel, S., Wilbraham, D., Fuller, R., Getz, E.B. & Longphre, M. Effect of an interleukin-4 variant on late phase asthmatic response to allergen challenge in asthmatic patients: results of two phase 2a studies. Lancet 370, 1422–1431 (2007).
Catley, M.C. Asthma & COPD–IQPC's Second Conference. IDrugs 13, 601–604 (2010).
Borish, L.C. et al. Interleukin-4 receptor in moderate atopic asthma. A phase I/II randomized, placebo-controlled trial. Am. J. Respir. Crit. Care Med. 160, 1816–1823 (1999).
Haldar, P. et al. Mepolizumab and exacerbations of refractory eosinophilic asthma. N. Engl. J. Med. 360, 973–984 (2009).
Busse, W.W. et al. Safety profile, pharmacokinetics, and biologic activity of MEDI-563, an anti-IL-5 receptor α antibody, in a phase I study of subjects with mild asthma. J. Allergy Clin. Immunol. 125, 1237–1244 (2010).
Castro, M. et al. Reslizumab for poorly controlled, eosinophilic asthma: a randomized, placebo-controlled study. Am. J. Respir. Crit. Care Med. 184, 1125–1132 (2011).
Spergel, J.M. et al. Reslizumab in children and adolescents with eosinophilic esophagitis: results of a double-blind, randomized, placebo-controlled trial. J. Allergy Clin. Immunol. 129, 456–463 (2012).
Wilson, M.S. et al. IL-13Rα2 and IL-10 coordinately suppress airway inflammation, airway-hyperreactivity, and fibrosis in mice. J. Clin. Invest. 117, 2941–2951 (2007).
Nicholson, G.C. et al. The effects of an anti–IL-13 mAb on cytokine levels and nasal symptoms following nasal allergen challenge. J. Allergy Clin. Immunol. 128, 800–807 (2011).
Ballantyne, S.J. et al. Blocking IL-25 prevents airway hyperresponsiveness in allergic asthma. J. Allergy Clin. Immunol. 120, 1324–1331 (2007).
Grimstad, O. et al. Anti–interleukin-31-antibodies ameliorate scratching behaviour in NC/Nga mice: a model of atopic dermatitis. Exp. Dermatol. 18, 35–43 (2009).
Zhang, F., Huang, G., Hu, B., Song, Y. & Shi, Y. A soluble thymic stromal lymphopoietin (TSLP) antagonist, TSLPR-immunoglobulin, reduces the severity of allergic disease by regulating pulmonary dendritic cells. Clin. Exp. Immunol. 164, 256–264 (2011).
Lin, J. et al. TNFα blockade in human diseases: an overview of efficacy and safety. Clin. Immunol. 126, 13–30 (2008).
Wenzel, S.E. et al. A randomized, double-blind, placebo-controlled study of tumor necrosis factor-α blockade in severe persistent asthma. Am. J. Respir. Crit. Care Med. 179, 549–558 (2009).
Harper, E.G. et al. Efalizumab therapy for atopic dermatitis causes marked increases in circulating effector memory CD4+ T cells that express cutaneous lymphocyte antigen. J. Invest. Dermatol. 128, 1173–1181 (2008).
Allakhverdi, Z. et al. Multitargeted approach using antisense oligonucleotides for the treatment of asthma. Ann. NY Acad. Sci. 1082, 62–73 (2006).
Antoniu, S.A. Mogamulizumab, a humanized mAb against C–C chemokine receptor 4 for the potential treatment of T-cell lymphomas and asthma. Curr. Opin. Mol. Ther. 12, 770–779 (2010).
Sato, T. et al. Inhibitory effect of the new orally active CCR4 antagonist K327 on CCR4+CD4+ T cell migration into the lung of mice with ovalbumin-induced lung allergic inflammation. Pharmacology 84, 171–182 (2009).
Barnes, N. et al. A randomized, double-blind, placebo-controlled study of the CRTH2 antagonist OC000459 in moderate persistent asthma. Clin. Exp. Allergy 42, 38–48 (2012).
Basinski, T.M. et al. Dual nature of T cell-epithelium interaction in chronic rhinosinusitis. J. Allergy Clin. Immunol. 124, 74–80 (2009).
Akdis, C.A. Allergy and hypersensitivity: mechanisms of allergic disease. Curr. Opin. Immunol. 18, 718–726 (2006).
Rodrigo, G.J., Neffen, H. & Castro-Rodriguez, J.A. Efficacy and safety of subcutaneous omalizumab vs placebo as add-on therapy to corticosteroids for children and adults with asthma: a systematic review. Chest 139, 28–35 (2011).
Melendez, A.J. et al. Inhibition of Fc ɛ RI-mediated mast cell responses by ES-62, a product of parasitic filarial nematodes. Nat. Med. 13, 1375–1381 (2007).
Hitomi, K. et al. An immunoglobulin-like receptor, Allergin-1, inhibits immunoglobulin E-mediated immediate hypersensitivity reactions. Nat. Immunol. 11, 601–607 (2010).
Wilhelm, C. et al. An IL-9 fate reporter demonstrates the induction of an innate IL-9 response in lung inflammation. Nat. Immunol. 12, 1071–1077 (2011).
Burgler, S. et al. Differentiation and functional analysis of human T(H)17 cells. J. Allergy Clin. Immunol. 123, 588–595 (2009).
Eyerich, S. et al. Th22 cells represent a distinct human T cell subset involved in epidermal immunity and remodeling. J. Clin. Invest. 119, 3573–3585 (2009).
Acknowledgements
The author's laboratory is supported by the Swiss National Science Foundation grant 320030-132899 and the Christine Kühne-Center for Allergy Research and Education (CK-CARE) European 7th frame work projects MeDALL (Mechanisms of the Development of Allergy) (261357) and PREDICTA (Post-Infectious Immune Reprogramming and Its Association with Persistence and Chronicity of Respiratory Allergic Diseases) (260895).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
C.A.A. received research grants from the Swiss National Science Foundation, European 7th Framework projects (264357); PREDICTA (260895); AllergoPharma Joachim-Ganzer KG, Reinbek, Germany; Stallergenes, Paris, France; Novartis; and GlaxoSmithKline.
Rights and permissions
About this article
Cite this article
Akdis, C. Therapies for allergic inflammation: refining strategies to induce tolerance. Nat Med 18, 736–749 (2012). https://doi.org/10.1038/nm.2754
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2754
This article is cited by
-
Paedoksan ameliorates allergic disease through inhibition of the phosphorylation of STAT6 in DNCB-induced atopic dermatitis like mice
Applied Biological Chemistry (2023)
-
The unique properties of IgG4 and its roles in health and disease
Nature Reviews Immunology (2023)
-
Old Friends with Unexploited Perspectives: Current Advances in Mesenchymal Stem Cell-Based Therapies in Asthma
Stem Cell Reviews and Reports (2021)
-
Immunogenicity of Milk Protein-Containing Hydrophilic Gel Patch for Epicutaneous Immunotherapy for Milk Allergy
Pharmaceutical Research (2020)
-
A20-OVA Nanoparticles Inhibit Allergic Asthma in a Murine Model
Inflammation (2020)