Abstract
Toll-like receptor 4 (TLR4) has a key role in innate immunity by activating an inflammatory signaling pathway. Free fatty acids (FFAs) stimulate adipose tissue inflammation through the TLR4 pathway, resulting in insulin resistance1,2,3,4,5,6,7. However, current evidence suggests that FFAs do not directly bind to TLR48,9, but an endogenous ligand for TLR4 remains to be identified. Here we show that fetuin-A (FetA) could be this endogenous ligand and that it has a crucial role in regulating insulin sensitivity via Tlr4 signaling in mice. FetA (officially known as Ahsg) knockdown in mice with insulin resistance caused by a high-fat diet (HFD) resulted in downregulation of Tlr4-mediated inflammatory signaling in adipose tissue, whereas selective administration of FetA induced inflammatory signaling and insulin resistance. FFA-induced proinflammatory cytokine expression in adipocytes occurred only in the presence of both FetA and Tlr4; removing either of them prevented FFA-induced insulin resistance. We further found that FetA, through its terminal galactoside moiety, directly binds the residues of Leu100–Gly123 and Thr493–Thr516 in Tlr4. FFAs did not produce insulin resistance in adipocytes with mutated Tlr4 or galactoside-cleaved FetA. Taken together, our results suggest that FetA fulfills the requirement of an endogenous ligand for TLR4 through which lipids induce insulin resistance. This may position FetA as a new therapeutic target for managing insulin resistance and type 2 diabetes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
Change history
11 January 2013
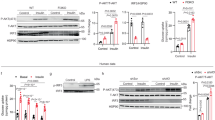
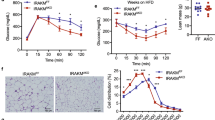
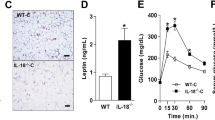
In the version of this article initially published, the groups presented in each key of the graphs for the insulin tolerance test results in Figure 1c,i were incorrectly assigned to the lines of the graphs. Also, the y-axis labels for the graphs were incorrect and misleading. The values along the graph lines for each group of animals presented represent the percentage of the initial value measured at time 0 for each group, not the percentage change. The errors have been corrected in the HTML and PDF versions of the article.
References
Lee, J.Y., Sohn, K.H., Rhee, S.H. & Hwang, D. Saturated fatty acids, but not unsaturated fatty acids, induce the expression of cyclooygenase-2 mediated through Toll-like receptor 4. J. Biol. Chem. 276, 16683–16689 (2001).
Suganami, T. et al. Role of the Toll-like receptor 4/NF-κB pathway in saturated fatty acid–induced inflammatory changes in the interaction between adipocytes and macrophages. Arterioscler. Thromb. Vasc. Biol. 27, 84–91 (2007).
Nguyen, M.T.A. et al. A subpopulation of microphages infiltrates hypertrophic adipose tissue and is activate by free fatty acids via Toll-like receptors 2 and 4 and JNK-dependent pathways. J. Biol. Chem. 282, 35279–35292 (2007).
Shi, H. et al. TLR4 links innate immunity and fatty acid-induced insulin resistance. J. Clin. Invest. 116, 3015–3025 (2006).
Kim, J.K. Fat uses a TOLL-road to connect inflammation and diabetes. Cell Metab. 4, 417–419 (2006).
Fessler, M.B., Rudel, L.L. & Brown, J.M. Toll-like receptor signaling links dietary fatty acids to the metabolic syndrome. Curr. Opin. Lipidol. 20, 379–385 (2009).
Kim, F. et al. Toll-like receptor-4 mediates vascular inflammation and insulin resistance in diet-induced obesity. Circ. Res. 100, 1589–1596 (2007).
Schaeffler, A. et al. Fatty acid–induced induction of Toll-like receptor-4/nuclear factor-κB pathway in adipocytes links nutritional signalling with innate immunity. Immunology 126, 233–245 (2009).
Erridge, C. & Samani, N.J. Saturated fatty acids do not directly stimulate Toll-like receptor signaling. Arterioscler. Thromb. Vasc. Biol. 29, 1944–1949 (2009).
Xu, H. et al. Chronic inflammation in fat plays a crucial role in the development of obesity-related insulin resistance. J. Clin. Invest. 112, 1821–1830 (2003).
Hotamisligil, G.S., Arner, P., Caro, J.F., Atkinson, R.L. & Spiegelman, B.M. Increased adipose tissue expression of tumor necrosis factor-α in human obesity and insulin resistance. J. Clin. Invest. 95, 2409–2415 (1995).
de Luca, C. & Olefsky, J.M. Stressed out about obesity and insulin resistance. Nat. Med. 12, 41–42 (2006).
Qatanani, M. & Lazar, M.A. Mechanisms of obesity-associated insulin resistance: many choices on the menu. Genes Dev. 21, 1443–1455 (2007).
Mori, K. et al. Association of serum fetuin-A with insulin resistance in type 2 diabetic and nondiabetic subjects. Diabetes Care 29, 468 (2006).
Stefan, N. et al. Plasma fetuin-A levels and the risk of type 2 diabetes. Diabetes 57, 2762–2767 (2008).
Ix, J.H. & Sharma, K. Mechanisms linking obesity, chronic kidney disease, and fatty liver disease: the roles of fetuin-A, adiponectin, and AMPK. J. Am. Soc. Nephrol. 21, 406–412 (2010).
Dasgupta, S. et al. NF-κB mediates lipid-induced fetuin-A expression in hepatocytes that impairs adipocyte function effecting insulin resistance. Biochem. J. 429, 451–462 (2010).
Hennige, A.M. et al. Fetuin-A induces cytokine expression and suppresses adiponectin production. PLoS ONE 3, e1765 (2008).
Mathews, S.T. et al. Improved insulin sensitivity and resistance to weight gain in mice null for the Ahsg gene. Diabetes 51, 2450–2458 (2002).
Tsukumo, D.M.L. et al. Loss-of-function mutation in TLR4 prevents diet-induced obesity and insulin resistance. Diabetes 56, 1986–1998 (2007).
Mathews, S.T. et al. Fetuin null mice are protected against obesity and insulin resistance associated with aging. Biochem. Biophys. Res. Commun. 350, 437–443 (2006).
Cayatte, A.J., Kumbla, L. & Ravi Subbiah, M.T. Marked acceleration of exogenous fatty acid incorporation into cellular triglycerides by fetuin. J. Biol. Chem. 265, 5883–5888 (1990).
Jiao, P. et al. Obesity-related upregulation of monocyte chemotactic factors in adipocytes involvement of nuclear factor-κB and c-Jun NH2–terminal kinase pathways. Diabetes 58, 104–115 (2009).
Zu, L. et al. Bacterial endotoxin stimulates adipose lipolysis via Toll-like receptor 4 and extracellular signal-regulated kinase pathway. J. Biol. Chem. 284, 5915–5926 (2009).
Mellgren, R.L. & Huang, X. Fetuin A stabilizes m-calpain and facilitates plasma membrane repair. J. Biol. Chem. 282, 35868–35877 (2007).
Davis, J.E., Gabler, N.K., Daniel, J.W. & Spurlock, M.E. Tlr-4 deficiency selectively protects against obesity induced by diets high in saturated fat. Obesity (Silver Spring) 16, 1248–1255 (2008).
Schwartz, E.A. et al. Nutrient modification of the innate immune response: a novel mechanism by which saturated fatty acids greatly amplify monocyte inflammation. Arterioscler. Thromb. Vasc. Biol. 30, 802–808 (2010).
Barton, G.M. & Medzhitov, R. Toll-like receptor signaling pathways. Science 300, 1524–1525 (2003).
Kawai, T. & Akira, S. TLR signaling. Cell Death Differ. 13, 816–825 (2006).
Medzhitov, R., Preston-Hurlburt, P. & Janeway, C.A.J. A human homologue of the Drosophila Toll protein signals activation of adaptive immunity. Nature 388, 394–397 (1997).
Paulson, J.C. & Colley, J.C.J. Glycosyltransferases. Structure, localization, and control of cell type-specific glycosylation. J. Biol. Chem. 264, 17615–17618 (1989).
Park, B.S. et al. The structural basis of lipopolysaccharide recognition by the TLR4–MD-2 complex. Nature 458, 1191–1195 (2009).
Barata, T.S., Teo, I., Brocchini, S., Zloh, M. & Shaunak, S. Partially glycosylated dendrimers block MD-2 and prevent TLR4–MD-2–LPS complex mediated cytokine responses. PLoS Comput. Biol. 7, e1002095 (2011).
Ohto, U., Fukase, K., Miyake, K. & Satow, Y. Crystal structures of human MD-2 and its complex with antiendotoxic lipid IVa. Science 316, 1632–1634 (2007).
Orr, J.S. et al. Toll-like receptor 4 deficiency promotes the alternative activation of adipose tissue macrophages. Diabetes published online, doi:10.2337/db11-1595 (29 June 2012).
Suganami, T. et al. Attenuation of obesity-induced adipose tissue inflammation in C3H/HeJ mice carrying a Toll-like receptor 4 mutation. Biochem. Biophys. Res. Commun. 354, 45–49 (2017).
Matthews, D.R. et al. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 28, 412–419 (1985).
Li, W. et al. A hepatic protein, fetuin-a, occupies a protective role in lethal systemic inflammation. PLoS ONE 6, e16945 (2011).
Lassenius, M.I. et al. Bacterial endotoxin activity in human serum is associated with dyslipidemia, insulin resistance, obesity, and chronic inflammation. Diabetes Care 34, 1809–1815 (2011).
Lowry, O.H., Rosebrough, N.J., Farr, A.E. & Randall, R.J. Protein measurement with Folin phenol reagent. J. Biol. Chem. 193, 265–275 (1951).
Dasgupta, S. et al. Mechanism of lipid induced insulin resistance: Activated PKCɛ is a key regulator. Biochim. Biophys. Acta 1812, 495–506 (2011).
Dowler, S. et al. Protein lipid overlay assay. Sci. STKE 2002, pl6 (2002).
Acknowledgements
This research was financially supported by a grant from the CSIR and Department of Science and Technology, Ministry of Science and Technology, New Delhi. We thank the National Centre for Cell Science, Pune, India, for providing the 3T3L1 cell line; A. Bandyopadhyay and S. Roy of the Indian Institute of Chemical Biology, Kolkata for their help in the confocal and SPR work, respectively. D.P. is thankful to CSIR, New Delhi, for the award of Senior Research Fellowship. S.D. thanks the University Grants Commission (UGC), New Delhi, for the award of a UGC-Dr. D.S. Kothari postdoctoral fellowship and CSIR-NEIST for a Quick Hire Fellowship. S.B. thanks the Indian National Science Academy for his Senior Scientist position. G.D. gratefully acknowledges the gift of Tlr4−/− mice from R. Medzhitov, Yale School of Medicine. The authors appreciate the use of facilities as extended to us by the head of the Department of Zoology, Visva-Bharati University, Santiniketan; the director of IICB, director of IPGME&R-SSKM Hospital, Kolkata; the director of the National Institute of Immunology, director of ICGEB, New Delhi and the director of NEIST, Jorhat, India.
Author information
Authors and Affiliations
Contributions
D.P. and S.D. designed and performed all the experiments, analyzed the data and wrote the manuscript; R.K. generated VMO–based FetA and Tlr4 knockdown mice; G.D. performed lipid infusion study in WT and FetAKD mice; S.R. provided nondiabetic and diabetic human blood and fat tissue; S.S.M. provided blood and tissue samples from WT and db/db mice; S. Maitra, G.D., S. Mukhopadhyay and S.S.M. wrote the manuscript; S.B. designed and supervised this study, analyzed the data and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–4 and Supplementary Tables 1 and 2 (PDF 418 kb)
Rights and permissions
About this article
Cite this article
Pal, D., Dasgupta, S., Kundu, R. et al. Fetuin-A acts as an endogenous ligand of TLR4 to promote lipid-induced insulin resistance. Nat Med 18, 1279–1285 (2012). https://doi.org/10.1038/nm.2851
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2851
This article is cited by
-
A secondary analysis of indices of hepatic and beta cell function following 12 weeks of carbohydrate and energy restriction vs. free-living control in adults with type 2 diabetes
Nutrition & Metabolism (2024)
-
Inter-organ crosstalk during development and progression of type 2 diabetes mellitus
Nature Reviews Endocrinology (2024)
-
Effect of Intracerebroventricular Administration of Galanin-Like Peptide on Hepatokines in C57BL/6 J Mice
Journal of Molecular Neuroscience (2024)
-
Novel targets for potential therapeutic use in Diabetes mellitus
Diabetology & Metabolic Syndrome (2023)
-
In vitro, in vivo, and in silico analysis of synbiotics as preventive interventions for lipid metabolism in ethanol-induced adipose tissue injury
Lipids in Health and Disease (2023)