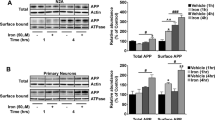
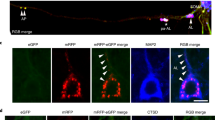
Abstract
Alzheimer's disease (AD) is a neurodegenerative disorder involving the florid deposition of vascular and cerebral plaques composed chiefly of amyloid β-peptide (Aβ) derived from cleavage of the amyloid precursor protein (APP)1–2. Varying in length from 39 to 43 amino acids, Aβ, particularly the longer Aβ(42), is thought to play a significant role in AD pathogenesis3–4. To better understand AD it is important to identify the subcellular organelles generating Aβ. Studies using agents that disrupt endosomal/lysosomal function suggest that Aβ is generated late in the secretory and endocytic pathways5. However, much of what is known about Aβ biosynthesis has been inferred by monitoring extracellular Aβ levels since intracellular Aβ is undetectable in most cell types. Consequently, the precise site or sites that generate Aβ, or whether Aβ(1–40) and Aβ(1–42) are generated at the same point in the biosynthetic pathway, is not known. Using human NT2N neurons, we found that retention of APP in the endoplasmic reticulum/intermediate compartment (ER/IC) by three independent approaches eliminated production of intracellular Aβ(1–40), but did not alter intracellular Aβ(1–42) synthesis. These findings suggest that the ER/IC may be an important site for generating this highly amyloidogenic species of Aβ.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Masters, C.L. et al. Amyloid plaque core protein in Alzheimer disease and Down syndrome. Proc. Natl. Acad. Sci. USA 82, 4245–4249 (1985).
Kang, J. et al. The precursor of Alzheimer's disease amyloid A4 protein resembles a cell-surface receptor. Nature 325, 733–736 (1987).
Suzuki, N. et al. An increased percentage of long amyloid (5 protein is secreted by familial amyloid β protein precursor (βAPP717) mutants. Science 264, 1336–1340 (1994).
Iwatsubo, T. et al. Visualization of Aβ 42 (43) and Aβ 40 in senile plaques with end-specific Aβ monoclonals: Evidence that an initially deposited species is Aβ 42 (43). Neuron 13, 45–53 (1994).
Golde, T.E., Estus, S., Younkin, L.H., Selkoe, D.J. & Younkin, S.C. Processing of the amyloid protein precursor to potentially amyloidogenic derivatives. Science 255, 728–730 (1992).
Turner, R.S., Suzuki, N., Chyung, A.S.C., Younkin, S.G. & Lee, V.M-Y. Amyloids β40 and β42 are generated intracellularly in cultured human neurons and their secretion increases with maturation. J. Biol. Chem. 271, 8966–8970 (1996).
Simons, M. et al. Amyloidogenic processing of the human amyloid precursor protein in primary cultures of rat hippocampal neurons. J. Neurosci. 16, 899–908 (1996).
Jackson, M.R., Nilsson, T. & Peterson, P.A. Identification of a consensus motif for retention of transmembrane proteins in the endoplasmic reticulum. EMBO J. 9, 3153–3162 (1990).
Saraste, J. & Kuismanen, E. Pre-and post-Golgi vacuoles operate in the transport of Semliki forest virus glycoproteins to the cell surface. Cell 38, 535–549 (1984).
Higaki, J., Quon, D., Zhong, Z. & Cordell, B. Inhibition of β-amyloid formation identifies proteolytic precursors and subcellular site of catabolism. Neuron 14, 651–659 (1995).
Gabuzda, D., Busciglio, J., Chen, L.B., Masudaira, P. & Yankner, B.A. Inhibition of energy metabolism alters the processing of amyloid precursor protein and induces a potentially amyloidogenic derivative. J. Biol. Chem. 269 13623–13628 (1994).
Scheuner, D. et al. Secreted amyloid β-protein similar to that in the senile plaques of Alzheimer's disease is increased in vivo by the presenilin 1 and 2 and APP mutations linked to familial Alzheimer's disease. Nature Med. 2 864–870 (1996).
Borchelt, D.R. et al. Familial Alzheimers-disease-linked presenilin-1 variants elevate Abetal–42/1–40 ratio in-vitro and in-vivo. Neuron 17, 1005–1013 (1996).
Duff, K. et al. Increased amyloid-β42 (43) in brains of mice expressing mutant presenilin 1. Nature 383, 710–713 (1996).
Lemere, C.A. et al. The E280A presenilin 1 Alzheimer mutation produces increased Aβ42 deposition and severe cerebellar pathology. Nature Med. 2 1146–1150 (1996).
Citron, M. et al. Mutant presenilins of Alzheimer's disease increase production of 42-residue amyloid β-protein in both transfected cells and transgenic mice. Nature Med. 3, 67–72 (1997).
Lah, J. et al. Light and electron microscope localization of presenilin-1 in primate brain. J. Neurosci. 17, 1971–1980 (1997).
Cook, D.G. et al. Expression and analysis of presenilin 1 in a human neuronal system: Localization in cell bodies and dendrites. Proc. Natl. Acad. Sci. USA 93, 9223–9228 (1996).
Kovacs, D.M. et al. Alzheimer-associated presenilins 1 and 2: Neuronal expression in brain and localization to intracellular membranes in mammalian cells. Nature Med. 2 2247ndash;229 (1996).
Cook, D.G., Lee, V.M.-Y. & Doms, R.W. Expression of foreign proteins in a human neuronal system. Methods Cell Biol. 43, 289–303 (1994).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Cook, D., Forman, M., Sung, J. et al. Alzheimer's Aβ(1–42) is generated in the endoplasmic reticulum/intermediate compartment of NT2N cells. Nat Med 3, 1021–1023 (1997). https://doi.org/10.1038/nm0997-1021
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nm0997-1021
This article is cited by
-
Physiological Roles of β-amyloid in Regulating Synaptic Function: Implications for AD Pathophysiology
Neuroscience Bulletin (2023)
-
Purification and characterization of β-secretase inhibitory peptide from sea hare (Aplysia kurodai) by enzymatic hydrolysis
Fisheries and Aquatic Sciences (2018)
-
Neuronal response in Alzheimer’s and Parkinson’s disease: the effect of toxic proteins on intracellular pathways
BMC Neuroscience (2015)
-
Conformational targeting of intracellular Aβ oligomers demonstrates their pathological oligomerization inside the endoplasmic reticulum
Nature Communications (2014)
-
Nilotinib-induced autophagic changes increase endogenous parkin level and ubiquitination, leading to amyloid clearance
Journal of Molecular Medicine (2014)