Key Points
-
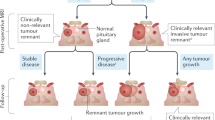
Pituitary tumours are common and cause unrestrained hormone secretion or compression of intracranial structures. Treatment aims to suppress hormone hypersecretion, ablate tumours or prevent their growth without compromising endogenous pituitary function.
-
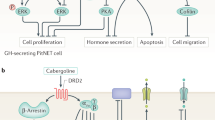
Hypothalamic and pituitary growth factors and their receptors regulate normal and abnormal pituitary-tumour hormone secretion and cell growth.
-
Pituitary tumours express various hormone receptors and retain inhibitory responses to agonistic and antagonistic ligands. Recent efforts have enabled the development of safe and potent anti-hormonal and anti-proliferative compounds aimed at specific pituitary-tumour-cell molecular targets and, potentially, other cancers.
-
First-generation dopamine-receptor agonists have been superceded by potent compounds with long-lasting effects and improved side-effect profiles. Treatment with somatostatin analogues is poised to broaden in application with the development of somatostatin-receptor subtype-specific and universal somatostatin analogues. Newer chimeric agonists that encompass dopaminergic actions as well as somatostatin-like actions are in development.
-
Peripheral growth-hormone-receptor antagonists (for example, pegvisomant) specifically block insulin-like growth factor 1 production. Retinoic-acid receptor and peroxisome-proliferator-activated receptor-γ also represent new approaches for treating pituitary tumours.
Abstract
Pituitary tumours are associated with unrestrained secretion and subsequent action of trophic hormones. One approach to therapy involves suppressing pituitary-hormone hypersecretion without compromising endogenous pituitary function. Identification of novel neuroendocrine-receptor targets has enabled the development of safe and effective receptor ligands that can be used to treat pituitary tumours and associated hormonal excess. Some of these agents, such as somatostatin analogues and a growth-hormone-receptor antagonist, will also have broader applications in treating other cancers and metabolic disorders.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Melmed, S. & Kleinberg, D. in Williams Textbook of Endocrinology 10th edn Ch. 8 (eds Larson, P. R., Kronenberg, H. M., Melmed, S. & Polonsky, K. S.) 177–279 (W. B. Saunders, Philadelphia, 2003). Comprehensive and up-to-date background information on pituitary structure and function.
Melmed, S. Mechanisms for pituitary tumorigenesis: the plastic pituitary. J. Clin. Invest. 112, 1603–1618 (2003).
Sano, K. Incidence of primary tumors (1969–1983) in Brain Tumor Registry of Japan. Neurol. Med. Chir. 37, 391–441 (1992).
Schlechte, J. A. Clinical impact of hyperprolactinaemia. Baillieres Clin. Endocrinol. Metab. 9, 359–366 (1995).
Molitch, M. E. Medical management of prolactin-secreting pituitary adenomas. Pituitary 5, 55–65 (2002).
Molitch, M. E. in The Pituitary 2nd edn (ed. Malden, M. A.) 455–495 (Blackwell Publishing, 2002)
Nabarro, J. D. Acromegaly. Clin. Endocrinol. 26, 481–512 (1987).
Miller, G. M. et al. Somatostatin receptor subtype gene expression in pituitary adenomas. J. Clin. Endocrinol. 80, 1386–1392 (1995).
Larsson, C., Skogseid, B., Oberg, K., Nakamura, Y. & Nordenskjold, M. Multiple endocrine neoplasia type 1 gene maps to chromosome 11 and is lost in insulinoma. Nature 332, 85–87 (1988).
Chandrasekharappak, S. C. et al. Positional cloning of the gene for multiple endocrine neoplasia-type 1. Science 276, 404–406 (1997). Describes the menin gene, which is one of the few characterized genes associated with familial pituitary-tumour syndromes.
Casey, M. et al. Mutations in the protein kinase A R1α regulatory subunit cause familial cardiac myxomas and Carney complex. J. Clin. Invest. 106, R31–R38 (2000).
Kirschner, L. S. et al. Mutations of the gene encoding the protein kinase A type I-α regulatory subunit in patients with Carney complex. Nature Genet. 26, 89–92 (2000).
Dotsch, J. et al. Gs α mutation at codon 201 in pituitary adenoma causing gigantism in a 6-year old boy with McCune–Albright syndrome. J. Clin. Endocrinol. Metab. 81, 3839–3842 (1996).
Asa, S. L. & Ezzat, S. The pathogenesis of pituitary tumors. Nature Rev. Cancer 2, 836–849 (2002).
Alexander, J. M. et al. Clinically non-functioning pituitary tumors are monoclonal in origin. J. Clin. Invest. 86, 336–340 (1990).
Herman, V., Fagin, J., Gonsky, R., Kovacs, K. & Melmed, S. Clonal origins of pituitary adenomas. J. Clin. Endocrinol. Metab. 71, 1427–1433 (1990). Seminal paper demonstrating that pituitary tumours are monoclonal expansions.
Thorner, M. O. et al. Somatotroph hyperplasia: successful treatment of acromegaly by removal of a pancreatic islet tumor secreting a growth-hormone-releasing factor. J. Clin. Invest. 70, 965–977 (1982).
Sano, T., Asa, S. L. & Kovacs, K. Growth hormone-releasing hormone-producing tumors: clinical, biochemical, and morphological manifestations. Endocr. Rev. 9, 357–373 (1988).
Lyons, J. et al. Two G protein oncogenes in human endocrine tumors. Science 249, 655–659 (1990). Describes the most common genetic mutation that is detected in sporadic pituitary tumours.
Spada, A. & Vallar, L. G-protein oncogenes in acromegaly. Horm. Res. 38, 90–93 (1992).
Zhang, X. et al. Pituitary tumor transforming gene (PTTG) expression in pituitary adenomas. J. Clin. Endocrinol. Metab. 84, 761–767 (1999).
Heaney, A. P. et al. Early involvement of estrogen-induced pituitary tumor transforming gene (PTTG) and fibroblast growth factor expression in prolactinoma pathogenesis. Nature Med. 5, 1317–1321 (1999).
Shimon, I. & Melmed, S. Management of pituitary tumors. Ann. Intern. Med. 129, 472–483 (1998).
Greenman, Y. & Melmed, S. Diagnosis and management of non-functioning pituitary tumors. Ann. Rev. Med. 47, 95–106 (1996).
Mampalam, T. J., Tyrrell, J. B. & Wilson, C. B. Transsphenoidal microsurgery for Cushing's disease: a report of 216 cases. Ann. Intern. Med. 109, 487–493 (1988).
Simmons, N. E., Alden, T. D., Thorner, M. O. & Laws, E. R. Jr. Serum cortisol response to transphenoidal surgery for Cushing disease. J. Neurosurg. 95, 1–8 (2001).
Hoybye, C. et al. Adrenocorticotrophic hormone-producing pituitary tumors: 12 to 22-year follow-up after treatment with sterotactic radiosurgery. Neurosurgery 49, 284–291 (2001).
Swearingen, B. et al. Long-term mortality after transsphenoidal surgery and adjunctive therapy for acromegaly. J. Clin. Endocrinol. Metab. 83, 3419–3426 (1998).
Freda, P. U., Wardlaw, S. L. & Post, K. D. Long-term endocrinologic follow-up after transsphenoidal surgery for acromegaly. J. Neurosurg. 89, 353–358 (1998).
Ebersold, M. J., Quast, L. M., Laws, E. R., Scheithauer, B. & Randall, R. V. Long-term results in transsphenoidal removal of nonfunctioning pituitary adenomas. J. Neurosurg. 64, 713–719 (1986).
Brada, M. et al. The long-term efficacy of conservative surgery and radiotherapy in the control of pituitary adenomas. Clin. Endocrinol. 38, 571–578 (1993).
Barrande, G. et al. Hormonal and metabolic effects of radiotherapy in acromegaly: long-term results in 128 patients followed in a single center. J. Clin. Endocrinol. Metab. 85, 3779–3785 (2000).
Erfurth, E. M., Bulow, B., Mikoczy, Z. & Hagmar, L. Incidence of a second tumor in hypopituitary patients operated for pituitary tumors. J. Clin. Endocrinol. Metab. 86, 659–662 (2001).
Freda, P. U. & Wardlaw, S. L. Primary medical therapy for acromegaly. J. Clin. Endocrinol. Metab. 83, 3031–3033 (1998).
Orth, D. N. Cushing's syndrome. N. Engl. J. Med. 332,791–803 (1995).
Reubi, J. C. et al. Somatostatin receptors in human endocrine tumours. Cancer Res. 47, 551–558 (1987).
Casini-Raggi, C. et al. Somatostatin receptors in non-endocrine tumors. Minerva Endocrinol. 26, 149–158 (2001).
Shimon, I. et al. Somatostatin receptor (SSTR) subtype-selective analogues differentially suppress in vitro growth hormone and prolactin in human pituitary adenomas. J. Clin. Invest. 100, 2386–2392 (1997).
Greenman, Y. & Melmed, S. Heterogenous expression of two somatostatin receptor subtypes in pituitary tumors. J. Clin. Endocrinol. Metab. 78, 398–403 (1994).
Ballare, E. et al. Mutation of somatostatin receptor type 5 in an acromegalic patient resistant to somatostatin analog treatment. J. Clin. Endocrinol. Metab. 86, 3809–3814 (2001).
Melmed, S. Acromegaly. N. Engl. J. Med. 322, 966–977 (1990).
Lamberts, S. W., van Der Lely, A. J., de Herder, W. W. & Hofland, L. J. Octreotide. N. Engl. J. Med. 334, 246–254 (1994).
Lamberts, S. W. J. et al. The sensitivity of growth hormone and prolactin secretion to the somatostatin analogue 201–995 in patients with prolactinomas and acromegaly. Clin. Endocrinol. 25, 201–212 (1986). Classical description of the developmental history and early use of somatostatin analogues.
Ezzat, S. et al. Octreotide treatment of acromegaly. A randomized multicenter study. Ann. Intern. Med. 117, 711–718 (1992).
Newman, C. B. et al. Safety and efficacy of long term octreotide therapy of acromegaly: results of a multicenter trial in 103 patients — a clinical research center study. J. Clin. Endocrinol. Metab. 80, 2768–2775 (1995).
Vance, M. L. & Harris, A. G. Long term treatment of 189 acromegalic patients with the somatostatin analog octreotide. Results of a multicenter acromegaly study group. Arch. Int. Med. 151, 1573–1578 (1991).
Legovini, P. et al. 111Indium-pentetreotide pituitary scintigraphy and hormonal responses to octreotide in acromegalic patients. J. Endocrinol. Invest. 20, 424–428 (1997).
Colao, A. et al. Long-term effects of depot long-acting somatostatin analog octreotide on hormone levels and tumor mass in acromegaly. J. Clin. Endocrinol. Metab. 86, 2779–2786 (2001).
Baldelli, R. et al. Two-year follow-up of acromegalic patients treated with slow release lanreotide (30 mg). J. Clin. Endocrinol. Metab. 85, 4099–4103 (2000).
Freda, P. U. Somatostatin analogs in acromegaly. J. Clin. Endocrinol. Metab. 87, 3013–3018 (2002).
Hofland, L. J. & Lamberts, S. W. The pathophysiological consequences of somatostatin receptor internalisation and resistance. Endocr. Rev. 24, 28–47 (2003).
Melmed, S. et al. Consensus: guidelines for acromegaly management. J. Clin. Endocrinol. Metab. 87, 4054–4058 (2001).
Patel, Y. et al. Molecular biology of somatostain receptor subtypes. Metabolism 45, 31–38 (1996).
Culler, M. D., Taylor, J. E. & Moreau, J. P. Somatostatin receptor subtypes: targeting functional and therapeutic specificity. Ann. Endocrinol. 63, 2S5–12 (2002).
Ren, S. G. et al. Functional association of somatostatin receptor subtypes 2 and 5 in inhibiting human growth hormone secretion. J. Clin. Endo. Metab. 88, 4239–4245 (2003).
Bruns, C., Lewis, I., Briner, U., Meno-Tetang, G. & Weckbecker, G. SOM230: a novel somatostatin peptidomimentic with broad somatotropin release inhibiting factor (SRIF) receptor binding and a unique antisecretory profile. Eur. J. Endocrinol. 146, 707–716 (2002).
Hofland, L. J. et al. The novel somatostatin analog SOM230 has a broad spectrum of inhibitory action on hormone release by human somatotroph, corticotroph and PRL-secreting pituitary adenomas in vitro. Program 85th Annual Meeting of The Endocrine Society P2-449 (2003).
van der Hoek, J. et al. A single dose comparison of the acute effects of the new somatostatin analog SOM230 and octreotide in acromegalic patients. Program 85th Annual Meeting of The Endocrine Society P1-625 (2003).
Murray, R. D. et al. The universal somatostatin ligand (SOM230) regulates human growth hormone secretion: novel peptide therapy for acromegaly. Program 84th Annual Meeting of The Endocrine Society 58–56 (2002).
Weckbecker, G., Briner, U., Lewis, I. & Bruns, C. SOM230: a new somatostatin peptidomimetic with potent inhibitory effects on the growth hormone/insulin-like growth factor-I axis in rats, primates, and dogs. Endocrinology 143, 4123–4130 (2002).
Sharma, K. & Srikant, C. B. Int. J. Cancer. 76, 259–266 (1998).
Pakes, D. Drug therapy: bromocriptine. N. Engl. J. Med. 301, 873–878 (1979).
Besser, G. M., Parke, L., Edwards, C. R., Forsyth, I. A. & McNeilly, A. S. Galactorrhoea: successful treatment with reduction of prolactin levels by brom-ergocryptine. Br. Med. J. 3, 669–672 (1972). First paper to describe the use of D2-agonist therapy for prolactin-secreting tumours.
Vance, M. L., Evans, W. S. & Thorner, M. O. Drugs five years later. Bromocriptine. Ann. Intern. Med. 100, 78–91 (1984).
Klibanski, A. & Zervas, N. T. Diagnosis and management of hormone-secreting pituitary adenomas. N. Engl. J. Med. 324, 822–831 (1991).
Bevan, J. S., Webster, J., Burke, C. W. & Scanlon, M. F. Dopamine agonists and pituitary tumor shrinkage. Endocr. Rev. 13, 220–240 (1992).
Kleinberg, D. L. et al. Pergolide for the treatment of pituitary tumors secreting prolactin or growth hormone. N. Engl. J. Med. 309, 704–709 (1983).
Vance, M. L. et al. Treatment of prolactin-secreting pituitary macroadenomas with the long-acting non-ergot dopamine agonist CV 205-502. Ann. Intern. Med. 112, 668–673 (1990).
Bevan, J. S. & Davis, J. R. Cabergoline: an advance in dopaminergic therapy. Clin. Endocrinol. 41, 709–712 (1994).
Colao, A. et al. Prolactinomas resistant to standard dopamine agonists respond to chronic cabergoline treatment. J. Clin. Endocrinol. Metab. 82, 876–883 (1997).
Webster, J. et al. A comparison of cabergoline and bromocriptine in the treatment of hyperprolactinemic amenorrhea. Cabergoline Comparative Study Group. N. Engl. J. Med. 331, 904–909 (1994).
Cunningham, B. C. et al. Dimerization of the extracellular domain of the human growth hormone receptor by a single hormone molecule. Science 254, 821–825 (1991).
Kopchick, J. J., Parkinson, C., Stevens, E. C. & Trainer, P. J. Growth hormone receptor antagonists: discovery, development, and use in patients with acromegaly. Endocr. Rev. 23, 623–646 (2002). Describes the developmental molecular rationale and mechanism of action of growth-hormone-receptor antagonists.
Kohn, D. T. & Kopchick, J. J. Growth hormone receptor antagonists. Minerva Endocrinol. 27, 287–298 (2002).
Frank, S. J. Receptor dimerization in GH and erythropoietin action: it takes two to tango. Endocinology 143, 2–10 (2002)
Trainer, P. J. et al. Treatment of acromegaly with the growth hormone receptor antagonist pegvisomant. N. Eng. J. Med. 342, 1171–1177 (2000).
Bonert, V. S., Zib, K., Scarlett, J. A. & Melmed, S. Growth hormone receptor antagonist therapy in acromegalic patients resistant to somatostatin analogs. J. Clin. Endocrinol. Metab. 85, 2958–2961 (2000).
van der Lely, A. J. et al. Long-term treatment of acromegaly with pegvisomant, a growth hormone receptor antagonist. Lancet. 358, 1754–1759 (2001).
Sheppard, M. C. Primary medical therapy for acromegaly. Clin. Endocrinol. 58, 387–399 (2003).
Flogstad, A. K. et al. A comparison of octreotide, bromocriptine, or a combination of both drugs in acromegaly. J. Clin. Endocrinol. Metab. 79, 461–465 (1994).
Saveanu, A. et al. Demonstration of enhanced potency of a chimeric somatostain-dopamine molecule, BIM-23A387, in suppressing growth hormone and prolactin secretion from human pituitary somatotroph adenoma cells. J. Clin. Endocrinol. Metab. 87, 5545–5552 (2002).
Ren, S. G. et al. Suppression of rat and human growth hormone and prolactin secretion by a novel somatostatin/dopaminergic chimeric ligand. J. Clin. Endocrinol. Metab. 11, 5414–5421 (2003).
Musset, F., Bertrand, P., Kordon, C. & Enjalbert, A. Differential coupling with pertussis toxin-sensitive G proteins of dopamine and somatostatin receptors involved in regulation of adenohypophyseal secretion. Mol. Cell Endocrinol. 73, 1–10 (1990).
Rocheville, M. et al. Receptors for dopamine and somatostatin: formation of hetero-oligomers with enhanced functional activity. Science 288, 154–157 (2000). First paper to demonstrate heterodimerization of the G-protein-coupled dopamine and somatostatin receptors.
Forman, B. M. et al. 15-Deoxy-δ 12,14-prostaglandin J1 is a ligand for the adipocyte determination factor PPAR γ. Cell 83, 803–812 (1995).
Elstner, E. et al. Ligands for peroxisome proliferator-activated receptor-γ and retinoic acid receptor inhibit growth and induce apoptosis of human breast cancer cells in vitro and in BNX mice. Proc. Natl Acad. Sci. USA 95, 8806–8811 (1988).
Kubota, T. et al. Ligand for peroxisome proliferator-activated receptor γ (troglitazone) has potent anti-tumor effects against prostate cancer both in vitro and in vivo. Cancer Res. 58, 3344–3352 (1998).
Heaney, A. P., Fernando, M., Yong, W. & Melmed, S. Functional PPAR-γ receptor represents a novel therapeutic target in Cushing's disease. Nature Med. 11, 1281–1287 (2002).
Heaney, A. P., Fernando, M. & Melmed, S. PPAR-γ receptor ligands: a novel therapy for pituitary tumors. J. Clin. Invest. 111, 1381–1388 (2003).
Nieman, L. Medical therapy of Cushing's disease. Pituitary 5, 77–82 (2003).
Gudas, L. J., Sporn, M. B. & Roberts, A. B. in The Retinoids: Biology, Chemistry, and Medicine. (eds Sporn, M. B., Roberts, A. B. & Goodman, D. S.) 443–520 (Raven, New York, 1994).
Pitha-Rowe, I., Petty, W. J., Kitareewan, S. & Dmitrovsky, E. Retinoid target genes in acute promyelocytic leukemia. Leukemia 17, 1723–1730 (2003).
Kurie, J. M. The biologic basis for the use of retinoids in cancer prevention and treatment. Curr. Opin. Oncol. 11, 497–502 (1999). Good review of rationale and potential therapeutic use of retinoids in cancer.
Rizvi, N. A. et al. A phase I study of LGD1069 in adults with advanced cancer. Clin. Cancer Res. 5, 1658–1664 (1999).
Koeffler, H. P. Peroxisome proliferator-activated receptor γ and cancers. Clin. Cancer Res. 9, 1–9 (2003).
Paez-Pereda, M. et al. Retinoic acid prevents experimental Cushing syndrome. J. Clin. Invest. 108, 1123–1131 (2001).
Colombo, P. et al. Effects of long-term treatment with the gonadotrophin-releasing analog nafarelin in patients with non-functioning pituitary adenomas. Eur. J. Endocrinol. 130, 339–345 (1994).
Schally, A. V. & Nagy, A. New approaches to treatment of various cancers based on cytotoxic analogs of LHRH, somatostatin and bombesin. Life. Sci. 11, 2305–2320 (2003).
Horwitz, G. A., Miklovsky, I., Heaney, A. P., Ren, S. G. & Melmed, S. Human pituitary tumor-transforming gene (PTTG1) motif suppresses prolactin expression. Mol. Endocrinol. 17, 600–609 (2003)
Acknowledgements
Work in the authors' laboratories is supported by grants from the National Institutes of Health (S.M.), the Doris Factor Molecular Endocrinology Laboratory (S.M.), the Annenberg Foundation (S.M. & A.P.H.) and the Margaret Early Trust (A.P.H.).
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- THYROTOXICOSIS
-
A clinical condition in which there is pathologically excessive production of thyroid hormones. The most common form is Grave's disease. Others include subacute thyroiditis and Hashimoto's disease.
- MULTIPLE ENDOCRINE NEOPLASIA TYPE 1 SYNDROME
-
An autosomal dominant disorder, features of which include pituitary and parathyroid adenomas, and/or tumours of the endocrine pancreas.
- CARNEY COMPLEX
-
An autosomal dominant disorder that is characterized by cutaneous pigmentation, atrial and other myxomas, schwannomas, and endocrine overactivity, most commonly adrenocorticotropin-hormone- and/or growth-hormone-secreting pituitary adenomas or thyroid adenomas, in association with spotty skin pigmentation.
- McCUNE–ALBRIGHT SYNDROME
-
Also known as precocious puberty, Cushing's syndrome, thyrotoxicosis and gigantism, this syndrome is characterized by polyostotic fibrous dysplasia, cutaneous hyperpigmentation and endocrine overactivity. Patients carry mutations in the GSP gene that lead to overactivation of adenyl cyclase, resulting in somatotroph hyperplasia and growth-hormone hypersecretion.
- TRANSSPHENOIDAL
-
A microsurgical approach that represents the most accessible and minimally traumatic corridor for removing a tumour of the pituitary gland. In this approach, the anterior face of the sphenoid bone is reached by tunnelling under the submucosa of the nose or under the lip.
- NON-FUNCTIONING PITUITARY TUMOURS
-
Pituitary tumours that are not associated with increased serum hormone levels in patients.
- ERGOT
-
Ergot is caused by the fungus Claviceps purpurea that infects various plants, including rye, cereals and grass. Ergot derivatives have had several uses in the field of medicine. Ergotamine was a former treatment for migraine headaches, but can cause gangrene with chronic use. Other derivatives include the psychotomimetic drug lysergic acid and bromocriptine, which binds the dopamine D2 receptor to lower serum prolactin levels.
- CHIMERIC LIGAND
-
A hybrid compound that combines characteristics of ligands that interact with two different receptors.
- PERTUSSIS-SENSITIVE G-PROTEINS
-
Pertussis toxin covalently modifies the α subunits of numerous G proteins (Gαi,Gαo) by ADP-ribosylation, uncoupling the G protein from its receptor.
- ALTERNATIVE PROMOTER USAGE
-
Differential gene regulation exercised through use of an alternative, often tissue-specific regulatory site, located at variable distances upstream of the known promoter.
Rights and permissions
About this article
Cite this article
Heaney, A., Melmed, S. Molecular targets in pituitary tumours. Nat Rev Cancer 4, 285–295 (2004). https://doi.org/10.1038/nrc1320
Issue Date:
DOI: https://doi.org/10.1038/nrc1320
This article is cited by
-
The tendency of reduced periodontal destruction in acromegalic patients showing similar inflammatory status with periodontitis patients
Endocrine (2019)
-
Malignant transformation in non-functioning pituitary adenomas (pituitary carcinoma)
Pituitary (2018)
-
Gastrointestinal neuroendocrine tumors (NETs): new diagnostic and therapeutic challenges
Cancer and Metastasis Reviews (2014)