Key Points
-
NEDD8 (neural precursor cell expressed developmentally downregulated protein 8) and ubiquitin have the highest sequence and structural similarity among all ubiquitin-like proteins.
-
NEDD8-specific conjugation and de-conjugation pathways exist in all studied eukaryotes, which can discriminate between NEDD8 and other ubiquitin-like proteins through NEDD8-specific interaction domains.
-
Nevertheless, a perturbed ratio of free NEDD8 and ubiquitin or cellular stress can result in the conjugation of NEDD8 through the ubiquitylation machinery onto ubiquitylation substrates. This can lead to mis-assignments of neddylation targets, and most published reports lack sufficient evidence to substantiate the discovery of genuine neddylation substrates.
-
We propose a list of necessary criteria for bona fide neddylation substrates and re-evaluate published studies in the light of these criteria. Cullins are the best-studied and only neddylation targets to date that fulfill all of these criteria.
-
We discuss potential examples of neddylation regulating non-cullin ubiquitin E3 ligases, transcription, ribosomal stress and various signalling pathways.
-
Pharmacological inhibition of neddylation is a promising new direction for cancer therapy. We discuss the potential effects of inhibiting non-cullin, as well as cullin, neddylation.
Abstract
NEDD8 (neural precursor cell expressed developmentally downregulated protein 8) is a ubiquitin-like protein that activates the largest ubiquitin E3 ligase family, the cullin–RING ligases. Many non-cullin neddylation targets have been proposed in recent years. However, overexpression of exogenous NEDD8 can trigger NEDD8 conjugation through the ubiquitylation machinery, which makes validating potential NEDD8 targets challenging. Here, we re-evaluate studies of non-cullin targets of NEDD8 in light of the current understanding of the neddylation pathway, and suggest criteria for identifying genuine neddylation substrates under homeostatic conditions. We describe the biological processes that might be regulated by non-cullin neddylation, and the utility of neddylation inhibitors for research and as potential therapies. Understanding the biological significance of non-cullin neddylation is an exciting research prospect primed to reveal fundamental insights.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
van der Veen, A. G. & Ploegh, H. L. Ubiquitin-like proteins. Annu. Rev. Biochem. 81, 323–357 (2012).
Ravid, T. & Hochstrasser, M. Diversity of degradation signals in the ubiquitin–proteasome system. Nature Rev. Mol. Cell Biol. 9, 679–689 (2008).
Gareau, J. R. & Lima, C. D. The SUMO pathway: emerging mechanisms that shape specificity, conjugation and recognition. Nature Rev. Mol. Cell Biol. 11, 861–871 (2010).
Schreiber, A. & Peter, M. Substrate recognition in selective autophagy and the ubiquitin-proteasome system. Biochim. Biophys. Acta 1843, 163–181 (2014).
Welchman, R. L., Gordon, C. & Mayer, R. J. Ubiquitin and ubiquitin-like proteins as multifunctional signals. Nature Rev. Mol. Cell Biol. 6, 599–609 (2005).
Kamitani, T., Kito, K., Nguyen, H. P. & Yeh, E. T. Characterization of NEDD8, a developmentally down-regulated ubiquitin-like protein. J. Biol. Chem. 272, 28557–28562 (1997).
Kim, D. Y. et al. CBFβ stabilizes HIV Vif to counteract APOBEC3 at the expense of RUNX1 target gene expression. Mol. Cell 49, 632–644 (2013).
Hori, T. et al. Covalent modification of all members of human cullin family proteins by NEDD8. Oncogene 18, 6829–6834 (1999).
Li, T., Chen, X., Garbutt, K. C., Zhou, P. & Zheng, N. Structure of DDB1 in complex with a paramyxovirus V protein: viral hijack of a propeller cluster in ubiquitin ligase. Cell 124, 105–117 (2006).
Kumar, S., Yoshida, Y. & Noda, M. Cloning of a cDNA which encodes a novel ubiquitin-like protein. Biochem. Biophys. Res. Commun. 195, 393–399 (1993).
Li, T., Robert, E. I., van Breugel, P. C., Strubin, M. & Zheng, N. A promiscuous α-helical motif anchors viral hijackers and substrate receptors to the CUL4-DDB1 ubiquitin ligase machinery. Nature Struct. Mol. Biol. 17, 105–111 (2010).
Kumar, S., Tomooka, Y. & Noda, M. Identification of a set of genes with developmentally down-regulated expression in the mouse brain. Biochem. Biophys. Res. Commun. 185, 1155–1161 (1992).
Carrabino, S., Carminati, E., Talarico, D., Pardi, R. & Bianchi, E. Expression pattern of the JAB1/CSN5 gene during murine embryogenesis: colocalization with NEDD8. Gene Expr. Patterns 4, 423–431 (2004).
Noh, E. H. et al. Covalent NEDD8 conjugation increases RCAN1 protein stability and potentiates its inhibitory action on calcineurin. PLoS ONE 7, e48315 (2012).
Salon, C. et al. Altered pattern of Cul-1 protein expression and neddylation in human lung tumours: relationships with CAND1 and cyclin E protein levels. J. Pathol. 213, 303–310 (2007).
Chairatvit, K. & Ngamkitidechakul, C. Control of cell proliferation via elevated NEDD8 conjugation in oral squamous cell carcinoma. Mol. Cell. Biochem. 306, 163–169 (2007).
Tateishi, K., Omata, M., Tanaka, K. & Chiba, T. The NEDD8 system is essential for cell cycle progression and morphogenetic pathway in mice. J. Cell Biol. 155, 571–579 (2001).
Dharmasiri, S., Dharmasiri, N., Hellmann, H. & Estelle, M. The RUB/Nedd8 conjugation pathway is required for early development in Arabidopsis. EMBO J. 22, 1762–1770 (2003).
Ou, C.-Y., Lin, Y.-F., Chen, Y.-J. & Chien, C.-T. Distinct protein degradation mechanisms mediated by Cul1 and Cul3 controlling Ci stability in Drosophila eye development. Genes Dev. 16, 2403–2414 (2002).
Jones, D. & Candido, E. P. The NED-8 conjugating system in Caenorhabditis elegans is required for embryogenesis and terminal differentiation of the hypodermis. Dev. Biol. 226, 152–165 (2000).
Osaka, F. et al. Covalent modifier NEDD8 is essential for SCF ubiquitin-ligase in fission yeast. EMBO J. 19, 3475–3484 (2000).
Liakopoulos, D., Doenges, G., Matuschewski, K. & Jentsch, S. A novel protein modification pathway related to the ubiquitin system. EMBO J. 17, 2208–2214 (1998).
Lammer, D. et al. Modification of yeast Cdc53p by the ubiquitin-related protein rub1p affects function of the SCFCdc4 complex. Genes Dev. 12, 914–926 (1998).
Whitby, F. G., Xia, G., Pickart, C. M. & Hill, C. P. Crystal structure of the human ubiquitin-like protein NEDD8 and interactions with ubiquitin pathway enzymes. J. Biol. Chem. 273, 34983–34991 (1998). Structural and biochemical characterization of NEDD8, defining specificity-determining residues.
Rao-Naik, C. et al. The rub family of ubiquitin-like proteins. Crystal structure of Arabidopsis rub1 and expression of multiple rubs in Arabidopsis. J. Biol. Chem. 273, 34976–34982 (1998).
Choi, Y.-S., Jeon, Y. H., Ryu, K.-S. & Cheong, C. 60th residues of ubiquitin and Nedd8 are located out of E2-binding surfaces, but are important for K48 ubiquitin-linkage. FEBS Lett. 583, 3323–3328 (2009).
Walden, H. et al. The structure of the APPBP1-UBA3-NEDD8-ATP complex reveals the basis for selective ubiquitin-like protein activation by an E1. Mol. Cell 12, 1427–1437 (2003). Structural and biochemical characterization of NAE, defining the residues that mediate specific interactions with NEDD8.
Reverter, D. et al. Structure of a complex between Nedd8 and the Ulp/Senp protease family member Den1. J. Mol. Biol. 345, 141–151 (2005).
Shen, L.-N. et al. Structural basis of NEDD8 ubiquitin discrimination by the deNEDDylating enzyme NEDP1. EMBO J. 24, 1341–1351 (2005).
Komander, D. & Rape, M. The ubiquitin code. Annu. Rev. Biochem. 81, 203–229 (2012).
Jin, L., Williamson, A., Banerjee, S., Philipp, I. & Rape, M. Mechanism of ubiquitin-chain formation by the human anaphase-promoting complex. Cell 133, 653–665 (2008).
Linghu, B., Callis, J. & Goebl, M. G. Rub1p processing by Yuh1p is required for wild-type levels of Rub1p conjugation to Cdc53p. Eukaryot. Cell 1, 491–494 (2002).
Wada, H., Kito, K., Caskey, L. S., Yeh, E. T. & Kamitani, T. Cleavage of the C-terminus of NEDD8 by UCH-L3. Biochem. Biophys. Res. Commun. 251, 688–692 (1998).
Gan-Erdene, T. et al. Identification and characterization of DEN1, a deneddylase of the ULP family. J. Biol. Chem. 278, 28892–28900 (2003).
Mendoza, H. M. et al. NEDP1, a highly conserved cysteine protease that deNEDDylates cullins. J. Biol. Chem. 278, 25637–25643 (2003).
Schulman, B. A. & Harper, J. W. Ubiquitin-like protein activation by E1 enzymes: the apex for downstream signalling pathways. Nature Rev. Mol. Cell Biol. 10, 319–331 (2009).
Wu, K. et al. DEN1 is a dual function protease capable of processing the C terminus of Nedd8 and deconjugating hyper-neddylated CUL1. J. Biol. Chem. 278, 28882–28891 (2003).
Chan, Y. et al. DEN1 deneddylates non-cullin proteins in vivo. J. Cell. Sci. 121, 3218–3223 (2008).
Kurihara, L. J., Semenova, E., Levorse, J. M. & Tilghman, S. M. Expression and functional analysis of Uch-L3 during mouse development. Mol. Cell. Biol. 20, 2498–2504 (2000).
Bohnsack, R. N. & Haas, A. L. Conservation in the mechanism of Nedd8 activation by the human AppBp1-Uba3 heterodimer. J. Biol. Chem. 278, 26823–26830 (2003).
Olsen, S. K., Capili, A. D., Lu, X., Tan, D. S. & Lima, C. D. Active site remodelling accompanies thioester bond formation in the SUMO E1. Nature 463, 906–912 (2010).
Huang, D. T. et al. Basis for a ubiquitin-like protein thioester switch toggling E1–E2 affinity. Nature 445, 394–398 (2007).
Huang, D. T. et al. Structural basis for recruitment of Ubc12 by an E2 binding domain in NEDD8's E1. Mol. Cell 17, 341–350 (2005).
Huang, D. T. et al. A unique E1-E2 interaction required for optimal conjugation of the ubiquitin-like protein NEDD8. Nature Struct. Mol. Biol. 11, 927–935 (2004).
Deshaies, R. J. & Joazeiro, C. A. P. RING domain E3 ubiquitin ligases. Annu. Rev. Biochem. 78, 399–434 (2009).
Huang, D. T. et al. E2-RING expansion of the NEDD8 cascade confers specificity to cullin modification. Mol. Cell 33, 483–495 (2009).
Kamura, T., Conrad, M. N., Yan, Q., Conaway, R. C. & Conaway, J. W. The Rbx1 subunit of SCF and VHL E3 ubiquitin ligase activates Rub1 modification of cullins Cdc53 and Cul2. Genes Dev. 13, 2928–2933 (1999).
Monda, J. K. et al. Structural conservation of distinctive N-terminal acetylation-dependent interactions across a family of mammalian NEDD8 ligation enzymes. Structure 21, 42–53 (2013).
Eletr, Z. M., Huang, D. T., Duda, D. M., Schulman, B. A. & Kuhlman, B. E2 conjugating enzymes must disengage from their E1 enzymes before E3-dependent ubiquitin and ubiquitin-like transfer. Nature Struct. Mol. Biol. 12, 933–934 (2005).
Pruneda, J. N., Stoll, K. E., Bolton, L. J., Brzovic, P. S. & Klevit, R. E. Ubiquitin in motion: structural studies of the ubiquitin-conjugating enzyme∼ubiquitin conjugate. Biochemistry 50, 1624–1633 (2011).
Dou, H., Buetow, L., Sibbet, G. J., Cameron, K. & Huang, D. T. BIRC7-E2 ubiquitin conjugate structure reveals the mechanism of ubiquitin transfer by a RING dimer. Nature Struct. Mol. Biol. 19, 876–883 (2012).
Plechanovová, A., Jaffray, E. G., Tatham, M. H., Naismith, J. H. & Hay, R. T. Structure of a RING E3 ligase and ubiquitin-loaded E2 primed for catalysis. Nature 489, 115–120 (2012).
Yunus, A. A. & Lima, C. D. Lysine activation and functional analysis of E2-mediated conjugation in the SUMO pathway. Nature Struct. Mol. Biol. 13, 491–499 (2006).
Berndsen, C. E., Wiener, R., Yu, I. W., Ringel, A. E. & Wolberger, C. A conserved asparagine has a structural role in ubiquitin-conjugating enzymes. Nature Chem. Biol. 9, 154–156 (2013).
Scott, D. C. et al. Structure of a RING E3 trapped in action reveals ligation mechanism for the ubiquitin-like protein NEDD8. Cell 157, 1671–1684 (2014).
Enchev, R. I., Schreiber, A., Beuron, F. & Morris, E. P. Structural insights into the COP9 signalosome and its common architecture with the 26S proteasome lid and eIF3. Structure 18, 518–527 (2010).
Gazdoiu, S. et al. Proximity-induced activation of human Cdc34 through heterologous dimerization. Proc. Natl Acad. Sci. USA 102, 15053–15058 (2005).
Ohki, Y., Funatsu, N., Konishi, N. & Chiba, T. The mechanism of poly-NEDD8 chain formation in vitro. Biochem. Biophys. Res. Commun. 381, 443–447 (2009).
Wu, P. Y. A conserved catalytic residue in the ubiquitin-conjugating enzyme family. EMBO J. 22, 5241–5250 (2003).
Jones, J. et al. A targeted proteomic analysis of the ubiquitin-like modifier NEDD8 and associated proteins. J. Proteome Res. 7, 1274–1287 (2008).
Jeram, S. M. et al. An improved SUMmOn-based methodology for the identification of ubiquitin and ubiquitin-like protein conjugation sites identifies novel ubiquitin-like protein chain linkages. Proteomics 10, 254–265 (2010).
Xirodimas, D. P. et al. Ribosomal proteins are targets for the NEDD8 pathway. EMBO Rep. 9, 280–286 (2008).
Leidecker, O., Matic, I., Mahata, B., Pion, E. & Xirodimas, D. P. The ubiquitin E1 enzyme Ube1 mediates NEDD8 activation under diverse stress conditions. Cell Cycle 11, 1142–1150 (2012).
Girdwood, D., Xirodimas, D. P. & Gordon, C. The essential functions of NEDD8 are mediated via distinct surface regions, and not by polyneddylation in Schizosaccharomyces pombe. PLoS ONE 6, e20089 (2011).
Ma, T. et al. RNF111-dependent neddylation activates DNA damage-induced ubiquitination. Mol. Cell 49, 897–907 (2013).
Singh, R. K. et al. Recognition and cleavage of related to ubiquitin 1 (Rub1) and Rub1-ubiquitin chains by components of the ubiquitin-proteasome system. Mol. Cell. Proteom. 11, 1595–1611 (2012).
Kurz, T. et al. The conserved protein DCN-1/Dcn1p is required for cullin neddylation in C. elegans and S. cerevisiae. Nature 435, 1257–1261 (2005).
Kurz, T. et al. Dcn1 functions as a scaffold-type E3 ligase for cullin neddylation. Mol. Cell 29, 23–35 (2008).
Scott, D. C. et al. A Dual E3 mechanism for Rub1 ligation to Cdc53. Mol. Cell 39, 784–796 (2010).
Scott, D. C., Monda, J. K., Bennett, E. J., Harper, J. W. & Schulman, B. A. N-terminal acetylation acts as an avidity enhancer within an interconnected multiprotein complex. Science 334, 674–678 (2011).
Rabut, G. et al. The TFIIH subunit Tfb3 regulates cullin neddylation. Mol. Cell 43, 488–495 (2011).
Huang, G., Kaufman, A. J., Ramanathan, Y. & Singh, B. SCCRO (DCUN1D1) promotes nuclear translocation and assembly of the neddylation E3 complex. J. Biol. Chem. 286, 10297–10304 (2011).
Wu, K. et al. Mono-ubiquitination drives nuclear export of the human DCN1-like protein hDCNL1. J. Biol. Chem. 286, 34060–34070 (2011).
Meyer-Schaller, N. et al. The human Dcn1-like protein DCNL3 promotes Cul3 neddylation at membranes. Proc. Natl Acad. Sci. USA 106, 12365–12370 (2009).
Compe, E. & Egly, J.-M. TFIIH: when transcription met DNA repair. Nature Rev. Mol. Cell Biol. 13, 343–354 (2012).
Cui, J. et al. Glutamine deamidation and dysfunction of ubiquitin/NEDD8 induced by a bacterial effector family. Science 329, 1215–1218 (2010).
Yao, Q. et al. Structural mechanism of ubiquitin and NEDD8 deamidation catalyzed by bacterial effectors that induce macrophage-specific apoptosis. Proc. Natl Acad. Sci. USA 109, 20395–20400 (2012).
Cope, G. A. et al. Role of predicted metalloprotease motif of Jab1/Csn5 in cleavage of Nedd8 from Cul1. Science 298, 608–611 (2002).
Echalier, A. et al. Insights into the regulation of the human COP9 signalosome catalytic subunit, CSN5/Jab1. Proc. Natl Acad. Sci. USA 110, 1273–1278 (2013).
Sharon, M. et al. Symmetrical modularity of the COP9 signalosome complex suggests its multifunctionality. Structure 17, 31–40 (2009).
Birol, M. et al. Structural and biochemical characterization of the Cop9 signalosome CSN5/CSN6 heterodimer. PLoS ONE 9, e105688 (2014).
Lingaraju, G. M. et al. Crystal structure of the human COP9 signalosome. Nature 512, 161–165 (2014).
Enchev, R. I. et al. Structural basis for a reciprocal regulation between SCF and CSN. Cell Rep. 2, 616–627 (2012). References 81 and 82 describe the structural and biochemical characterization of CSN and CSN–SCF complexes, respectively, indicating the interaction interfaces.
Emberley, E. D., Mosadeghi, R. & Deshaies, R. J. Deconjugation of Nedd8 from Cul1 is directly regulated by Skp1-Fbox and substrate, and CSN inhibits deneddylated SCF by a non-catalytic mechanism. J. Biol. Chem. 287, 29679–29689 (2012).
Fischer, E. S. et al. The molecular basis of CRL4DDB2/CSA ubiquitin ligase architecture, targeting, and activation. Cell 147, 1024–1039 (2011).
Duda, D. M. et al. Structural insights into NEDD8 activation of cullin-RING ligases: conformational control of conjugation. Cell 134, 995–1006 (2008). Structural and biochemical characterization of neddylated CRLs, rationalizing the activating effects of neddylation.
Broemer, M. et al. Systematic in vivo RNAi analysis identifies IAPs as NEDD8-E3 ligases. Mol. Cell 40, 810–822 (2010).
Hemelaar, J. et al. Specific and covalent targeting of conjugating and deconjugating enzymes of ubiquitin-like proteins. Mol. Cell. Biol. 24, 84–95 (2004).
Ye, Y. et al. Polyubiquitin binding and cross-reactivity in the USP domain deubiquitinase USP21. EMBO Rep. 12, 350–357 (2011).
Kamitani, T., Kito, K., Fukuda-Kamitani, T. & Yeh, E. T. Targeting of NEDD8 and its conjugates for proteasomal degradation by NUB1. J. Biol. Chem. 276, 46655–46660 (2001).
Oved, S. et al. Conjugation to Nedd8 instigates ubiquitylation and down-regulation of activated receptor tyrosine kinases. J. Biol. Chem. 281, 21640–21651 (2006).
Besten, den, W., Verma, R., Kleiger, G., Oania, R. S. & Deshaies, R. J. NEDD8 links cullin-RING ubiquitin ligase function to the p97 pathway. Nature Struct. Mol. Biol. 19, (Suppl. 1) 511–516 (2012).
Bandau, S., Knebel, A., Gage, Z. O., Wood, N. T. & Alexandru, G. UBXN7 docks on neddylated cullin complexes using its UIM motif and causes HIF1α accumulation. BMC Biol. 10, 36 (2012).
Shamay, M., Greenway, M., Liao, G., Ambinder, R. F. & Hayward, S. D. De novo DNA methyltransferase DNMT3b interacts with NEDD8-modified proteins. J. Biol. Chem. 285, 36377–36386 (2010).
Antenos, M., Casper, R. F. & Brown, T. J. Interaction with Nedd8, a ubiquitin-like protein, enhances the transcriptional activity of the aryl hydrocarbon receptor. J. Biol. Chem. 277, 44028–44034 (2002).
Kelsall, I. R. et al. TRIAD1 and HHARI bind to and are activated by distinct neddylated cullin-RING ligase complexes. EMBO J. 32, 2848–2860 (2013).
Tanaka, T., Kawashima, H., Yeh, E. T. H. & Kamitani, T. Regulation of the NEDD8 conjugation system by a splicing variant, NUB1L. J. Biol. Chem. 278, 32905–32913 (2003).
Burch, T. J. & Haas, A. L. Site-directed mutagenesis of ubiquitin. Differential roles for arginine in the interaction with ubiquitin-activating enzyme. Biochemistry 33, 7300–7308 (1994).
Walden, H., Podgorski, M. S. & Schulman, B. A. Insights into the ubiquitin transfer cascade from the structure of the activating enzyme for NEDD8. Nature 422, 330–334 (2003).
Hjerpe, R. et al. Changes in the ratio of free NEDD8 to ubiquitin triggers NEDDylation by ubiquitin enzymes. Biochem. J. 441, 927–936 (2012). Together with reference 62, this study describes a stress-induced neddylation pathway through the ubiquitylation machinery, which can lead to artefacts in the search for neddylation substrates.
Kim, W. et al. Systematic and quantitative assessment of the ubiquitin-modified proteome. Mol. Cell 44, 325–340 (2011).
Soucy, T. A. et al. An inhibitor of NEDD8-activating enzyme as a new approach to treat cancer. Nature 458, 732–736 (2009). Describes the development and characterization of a specific NAE inhibitor.
Saha, A. & Deshaies, R. J. Multimodal activation of the ubiquitin ligase SCF by Nedd8 conjugation. Mol. Cell 32, 21–31 (2008).
Yamoah, K. et al. Autoinhibitory regulation of SCF-mediated ubiquitination by human cullin 1's C-terminal tail. Proc. Natl Acad. Sci. USA 105, 12230–12235 (2008).
Boh, B. K., Smith, P. G. & Hagen, T. Neddylation-induced conformational control regulates cullin RING ligase activity in vivo. J. Mol. Biol. 409, 136–145 (2011).
Pierce, N. W. et al. Cand1 promotes assembly of new SCF complexes through dynamic exchange of F box proteins. Cell 153, 206–215 (2013).
Zemla, A. et al. CSN- and CAND1-dependent remodelling of the budding yeast SCF complex. Nature Commun. 4, 1641 (2013).
Wu, S. et al. CAND1 controls in vivo dynamics of the cullin 1-RING ubiquitin ligase repertoire. Nature Commun. 4, 1642 (2013).
Lydeard, J. R., Schulman, B. A. & Harper, J. W. Building and remodelling cullin-RING E3 ubiquitin ligases. EMBO Rep. 14, 1050–1061 (2013).
Scheffner, M. & Kumar, S. Mammalian HECT ubiquitin-protein ligases: biological and pathophysiological aspects. Biochim. Biophys. Acta 1843, 61–74 (2014).
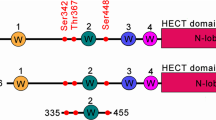
Xie, P. et al. The covalent modifier Nedd8 is critical for the activation of Smurf1 ubiquitin ligase in tumorigenesis. Nature Commun. 5, 3733 (2014). Functional characterization of the neddylation of a HECT E3 ligase.
Kaelin, W. G. Jr. von Hippel-Lindau disease. Annu. Rev. Pathol. Mech. Dis. 2, 145–173 (2007).
Stickle, N. H. et al. pVHL modification by NEDD8 is required for fibronectin matrix assembly and suppression of tumor development. Mol. Cell. Biol. 24, 3251–3261 (2004).
Russell, R. C. & Ohh, M. NEDD8 acts as a 'molecular switch' defining the functional selectivity of VHL. EMBO Rep. 9, 486–491 (2008).
Trempe, J.-F. et al. Structure of parkin reveals mechanisms for ubiquitin ligase activation. Science 340, 1451–1455 (2013).
Chaugule, V. K. et al. Autoregulation of Parkin activity through its ubiquitin-like domain. EMBO J. 30, 2853–2867 (2011).
Wauer, T, & Komander, D. Structure of the human Parkin ligase domain in an autoinhibited state. EMBO J. 32, 2099–2112 (2013).
Riley, B. E. et al. Structure and function of Parkin E3 ubiquitin ligase reveals aspects of RING and HECT ligases. Nature Commun. 4, 1982 (2013).
Deas, E., Wood, N. W. & Plun-Favreau, H. Mitophagy and Parkinson's disease: The PINK1–parkin link. Biochim. Biophys. Acta (BBA) - Mol. Cell Res. 1813, 623–633 (2011).
Choo, Y. S. et al. Regulation of parkin and PINK1 by neddylation. Hum. Mol. Genet. 21, 2514–2523 (2012).
Um, J. W. et al. Neddylation positively regulates the ubiquitin E3 ligase activity of parkin. J. Neurosci. Res. 90, 1030–1042 (2012).
Dil Kuazi, A. et al. NEDD8 protein is involved in ubiquitinated inclusion bodies. J. Pathol. 199, 259–266 (2003).
Mori, F. et al. Accumulation of NEDD8 in neuronal and glial inclusions of neurodegenerative disorders. Neuropathol. Appl. Neurobiol. 31, 53–61 (2005).
Mori, F. et al. Ubiquitin-related proteins in neuronal and glial intranuclear inclusions in intranuclear inclusion body disease. Pathol. Int. 62, 407–411 (2012).
Odagiri, S. et al. Immunohistochemical analysis of Marinesco bodies, using antibodies against proteins implicated in the ubiquitin-proteasome system, autophagy and aggresome formation. Neuropathology 32, 261–266 (2012).
Chen, Y., Liu, W., McPhie, D. L., Hassinger, L. & Neve, R. L. APP-BP1 mediates APP-induced apoptosis and DNA synthesis and is increased in Alzheimer's disease brain. J. Cell Biol. 163, 27–33 (2003).
Vousden, K. H. & Lane, D. P. p53 in health and disease. Nature Rev. Mol. Cell Biol. 8, 275–283 (2007).
Xirodimas, D. P., Saville, M. K., Bourdon, J.-C., Hay, R. T. & Lane, D. P. Mdm2-mediated NEDD8 conjugation of p53 inhibits its transcriptional activity. Cell 118, 83–97 (2004). Describes how p53 transcriptional activity can be regulated by neddylation.
Dohmesen, C., Koeppel, M. & Dobbelstein, M. Specific inhibition of Mdm2-mediated neddylation by Tip60. Cell Cycle 7, 222–231 (2008).
Liu, G. & Xirodimas, D. P. NUB1 promotes cytoplasmic localization of p53 through cooperation of the NEDD8 and ubiquitin pathways. Oncogene 29, 2252–2261 (2010).
Carter, S., Bischof, O., Dejean, A. & Vousden, K. H. C-terminal modifications regulate MDM2 dissociation and nuclear export of p53. Nature Cell Biol. 9, 428–435 (2007).
Watson, I. R., Blanch, A., Lin, D. C. C., Ohh, M. & Irwin, M. S. Mdm2-mediated NEDD8 modification of TAp73 regulates its transactivation function. J. Biol. Chem. 281, 34096–34103 (2006).
Abida, W. M., Nikolaev, A., Zhao, W., Zhang, W. & Gu, W. FBXO11 promotes the neddylation of p53 and inhibits its transcriptional activity. J. Biol. Chem. 282, 1797–1804 (2007).
van den Heuvel, S. & Dyson, N. J. Conserved functions of the pRB and E2F families. Nature Rev. Mol. Cell Biol. 9, 713–724 (2008).
Aoki, I., Higuchi, M. & Gotoh, Y. NEDDylation controls the target specificity of E2F1 and apoptosis induction. Oncogene 32, 3954–3964 (2013).
Loftus, S. J., Liu, G., Carr, S. M., Munro, S. & La Thangue, N. B. NEDDylation regulates E2F-1-dependent transcription. EMBO Rep. 13, 811–818 (2012). Describes the negative regulation of E2F transcription by neddylation.
Watson, I. R. et al. Chemotherapy induces NEDP1-mediated destabilization of MDM2. Oncogene 29, 297–304 (2010).
Kontaki, H. & Talianidis, I. Lysine methylation regulates E2F1-induced cell death. Mol. Cell 39, 152–160 (2010).
Hayden, M. S. & Ghosh, S. NF-κB, the first quarter-century: remarkable progress and outstanding questions. Genes Dev. 26, 203–234 (2012).
Amir, R. E., Iwai, K. & Ciechanover, A. The NEDD8 pathway is essential for SCF(β -TrCP)-mediated ubiquitination and processing of the NF-κ B precursor p105. J. Biol. Chem. 277, 23253–23259 (2002).
Noguchi, K. et al. TRIM40 promotes neddylation of IKKγ and is downregulated in gastrointestinal cancers. Carcinogenesis 32, 995–1004 (2011).
Gao, F., Cheng, J., Shi, T. & Yeh, E. T. H. Neddylation of a breast cancer-associated protein recruits a class III histone deacetylase that represses NFκB-dependent transcription. Nature Cell Biol. 8, 1171–1177 (2006).
Gao, N., Asamitsu, K., Hibi, Y., Ueno, T. & Okamoto, T. AKIP1 enhances NF-κB-dependent gene expression by promoting the nuclear retention and phosphorylation of p65. J. Biol. Chem. 283, 7834–7843 (2008).
Takashima, O. et al. Brap2 regulates temporal control of NF-κB localization mediated by inflammatory response. PLoS ONE 8, e58911 (2013).
Cao, X. & Südhof, T. C. A transcriptionally [correction of transcriptively] active complex of APP with Fe65 and histone acetyltransferase Tip60. Science 293, 115–120 (2001).
Lee, M.-R., Lee, D., Shin, S. K., Kim, Y. H. & Choi, C. Y. Inhibition of APP intracellular domain (AICD) transcriptional activity via covalent conjugation with Nedd8. Biochem. Biophys. Res. Commun. 366, 976–981 (2008).
Chen, Y., Neve, R. L. & Liu, H. Neddylation dysfunction in Alzheimer's disease. J. Cell. Mol. Med. 16, 2583–2591 (2012).
Ikushima, H. & Miyazono, K. TGFβ signalling: a complex web in cancer progression. Nature Rev. Cancer 10, 415–424 (2010).
Zuo, W. et al. c-Cbl-mediated neddylation antagonizes ubiquitination and degradation of the TGF-β type II receptor. Mol. Cell 49, 499–510 (2013). Describes how neddylation could regulate protein stability through the endocytosis–lysosomal pathway.
Marmor, M. D. & Yarden, Y. Role of protein ubiquitylation in regulating endocytosis of receptor tyrosine kinases. Oncogene 23, 2057–2070 (2004).
Nagano, T., Hashimoto, T., Nakashima, A., Kikkawa, U. & Kamada, S. X-linked inhibitor of apoptosis protein mediates neddylation by itself but does not function as a NEDD8-E3 ligase for caspase-7. FEBS Lett. 586, 1612–1616 (2012).
Teng, T., Thomas, G. & Mercer, C. A. Growth control and ribosomopathies. Curr. Opin. Genet. Dev. 23, 63–71 (2013).
Sundqvist, A., Liu, G., Mirsaliotis, A. & Xirodimas, D. P. Regulation of nucleolar signalling to p53 through NEDDylation of L11. EMBO Rep. 10, 1132–1139 (2009).
Mahata, B., Sundqvist, A. & Xirodimas, D. P. Recruitment of RPL11 at promoter sites of p53-regulated genes upon nucleolar stress through NEDD8 and in an Mdm2-dependent manner. Oncogene 31, 3060–3071 (2012).
Zhang, J., Bai, D., Ma, X., Guan, J. & Zheng, X. hCINAP is a novel regulator of ribosomal protein-HDM2-p53 pathway by controlling NEDDylation of ribosomal protein S14. Oncogene 33, 246–254 (2014).
Ran, F. A. et al. Genome engineering using the CRISPR-Cas9 system. Nature Protoc. 8, 2281–2308 (2013).
Bian, Y. et al. An enzyme assisted RP-RPLC approach for in-depth analysis of human liver phosphoproteome. J. Proteom. 96, 253–262 (2014).
Kettenbach, A. N. et al. Quantitative phosphoproteomics identifies substrates and functional modules of Aurora and Polo-like kinase activities in mitotic cells. Sci. Signal 4, rs5 (2011).
Weinert, B. T. et al. Lysine succinylation is a frequently occurring modification in prokaryotes and eukaryotes and extensively overlaps with acetylation. Cell Rep. 4, 842–851 (2013).
Rigbolt, K. T. G. et al. System-wide temporal characterization of the proteome and phosphoproteome of human embryonic stem cell differentiation. Sci. Signal 4, rs3 (2011).
Rabut, G. & Peter, M. Function and regulation of protein neddylation. 'Protein modifications: beyond the usual suspects' review series. EMBO Rep. 9, 969–976 (2008).
Gaj, T., Gersbach, C. A. & Barbas, C. F. ZFN, TALEN, and CRISPR/Cas-based methods for genome engineering. Trends Biotechnol. 31, 397–405 (2013).
Frescas, D. & Pagano, M. Deregulated proteolysis by the F-box proteins SKP2 and β-TrCP: tipping the scales of cancer. Nature Rev. Cancer 8, 438–449 (2008).
Jia, J. et al. Mechanisms of drug combinations: interaction and network perspectives. Nature Rev. Drug Discov. 8, 111–128 (2009).
Melchor, L. et al. Comprehensive characterization of the DNA amplification at 13q34 in human breast cancer reveals TFDP1 and CUL4A as likely candidate target genes. Breast Cancer Res. 11, R86 (2009).
Nakayama, K. I. & Nakayama, K. Ubiquitin ligases: cell-cycle control and cancer. Nature Rev. Cancer 6, 369–381 (2006).
Chang, F.-M. et al. Inhibition of neddylation represses lipopolysaccharide-induced proinflammatory cytokine production in macrophage cells. J. Biol. Chem. 287, 35756–35767 (2012).
Mathewson, N. et al. Neddylation plays an important role in the regulation of murine and human dendritic cell function. Blood 122, 2062–2073 (2013).
Nawrocki, S. T., Griffin, P., Kelly, K. R. & Carew, J. S. MLN4924: a novel first-in-class inhibitor of NEDD8-activating enzyme for cancer therapy. Expert Opin. Investig. Drugs 21, 1563–1573 (2012).
Brownell, J. E. et al. Substrate-assisted inhibition of ubiquitin-like protein-activating enzymes: the NEDD8 E1 inhibitor MLN4924 forms a NEDD8-AMP mimetic in situ. Mol. Cell 37, 102–111 (2010).
Lin, J. J., Milhollen, M. A., Smith, P. G., Narayanan, U. & Dutta, A. NEDD8-targeting drug MLN4924 elicits DNA rereplication by stabilizing Cdt1 in S phase, triggering checkpoint activation, apoptosis, and senescence in cancer cells. Cancer Res. 70, 10310–10320 (2010).
Milhollen, M. A. et al. Inhibition of NEDD8-activating enzyme induces rereplication and apoptosis in human tumor cells consistent with deregulating CDT1 turnover. Cancer Res. 71, 3042–3051 (2011).
Embade, N. et al. Murine double minute 2 regulates Hu antigen R stability in human liver and colon cancer through NEDDylation. Hepatology 55, 1237–1248 (2012).
McLarnon, A. Cancer: Mdm2-regulated stabilization of HuR by neddylation in HCC and colon cancer—a possible target for therapy. Nature Rev. Gastroenterol. Hepatol. 9, 4 (2012).
Ryu, J.-H. et al. Hypoxia-inducible factor α subunit stabilization by NEDD8 conjugation is reactive oxygen species-dependent. J. Biol. Chem. 286, 6963–6970 (2011).
Artavanis-Tsakonas, K. et al. Characterization and structural studies of the Plasmodium falciparum ubiquitin and Nedd8 hydrolase UCHL3. J. Biol. Chem. 285, 6857–6866 (2010).
Brzovic, P. S., Lissounov, A., Christensen, D. E., Hoyt, D. W. & Klevit, R. E. A. UbcH5/ubiquitin noncovalent complex is required for processive BRCA1-directed ubiquitination. Mol. Cell 21, 873–880 (2006).
Sakata, E. et al. Direct interactions between NEDD8 and ubiquitin E2 conjugating enzymes upregulate cullin-based E3 ligase activity. Nature Struct. Mol. Biol. 14, 167–168 (2007).
Acknowledgements
The authors thank D. Xirodimas, A. Schreiber and A. Smith for critical reading of the manuscript. R.I.E. is supported by a Marie-Curie post-doctoral fellowship, and work in the Peter laboratory is supported by the European Research Council (ERC), the Swiss National Science Foundation (SNF), and the ETH Zürich. B.A.S. acknowledges the Howard Hughes Medical Institute and National Institutes of Health (NIH) R01GM069530, P30CA021765 and ALSAC for support.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information S1 (table)
(PDF 213 kb)
Related links
Glossary
- Cullin–RING ligases
-
A large family of E3 ligases that are nucleated by a cullin scaffold protein and contain a RING domain. Cullin–RING ligases bring together the ubiquitylation substrate (through substrate-specific adaptors) and the E2 enzyme (through the RING domain RBX subunit) to catalyse the ubiquitylation reaction.
- UBA domain
-
A domain that is structurally characterized by a three-helix bundle and that recognizes the Ile44 hydrophobic patch of ubiquitin.
- JAMM motif
-
A metalloprotease His-X-His-X(10)-Asp motif that coordinates a zinc, present in multiple bacterial, archaeal and eukaryotic enzymes, including the deubiquitylating enzymes RPN11, STAMBPL1 and CSN5.
- p97
-
An AAA+ ATPase hexamer involved in eukaryotic signalling and quality control pathways. It delivers ubiquitylated proteins to the 26S proteasome through numerous adaptors.
- UIM
-
A motif that comprises an amphipathic helix with conserved negatively charged residues at its N terminus, a contiguous hydrophobic patch in the middle and a C-terminal Ser residue. The conserved Ala side chain of the UIM inserts in the Ile44 hydrophobic pocket of ubiquitin.
- MIU
-
A motif that is similar to the UIM but in which the helix runs in the opposite direction. MIUs are known to recognize Lys63-linked ubiquitin chains.
- Parkinson's disease
-
A neurodegenerative disease characterized by the loss of dopaminergic neurons of the midbrain and accumulation of insoluble inclusions containing α-synuclein amyloid fibrils.
- Alzheimer's disease
-
A neurodegenerative disease of the cerebral cortex characterized by the accumulation of aggregated amyloid-β.
- Effector caspase
-
An activated caspase that is produced from inactive pro-caspases through cleavage by initiator caspases. Effector caspases subsequently cleave protein substrates to induce the apoptotic process.
- Initiator caspase
-
This type of caspase is typically activated in response to particular stimuli; once activated, they cleave effector pro-caspases to activate them.
Rights and permissions
About this article
Cite this article
Enchev, R., Schulman, B. & Peter, M. Protein neddylation: beyond cullin–RING ligases. Nat Rev Mol Cell Biol 16, 30–44 (2015). https://doi.org/10.1038/nrm3919
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrm3919
This article is cited by
-
Crosstalk between protein post-translational modifications and phase separation
Cell Communication and Signaling (2024)
-
Deciphering the role of neddylation in tumor microenvironment modulation: common outcome of multiple signaling pathways
Biomarker Research (2024)
-
Neddylation is a novel therapeutic target for lupus by regulating double negative T cell homeostasis
Signal Transduction and Targeted Therapy (2024)
-
Targeting NEDD8 suppresses surgical stress-facilitated metastasis of colon cancer via restraining regulatory T cells
Cell Death & Disease (2024)
-
Identification of Potential Neddylation-related Key Genes in Ischemic Stroke based on Machine Learning Methods
Molecular Neurobiology (2024)