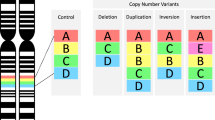
Key Points
-
Children with 22q11.2 deletion syndrome (22q11.2DS) show substantial impairments in several areas of attention. Spatial aspects of attention are likely to contribute to numerical impairments, and executive attention or cognitive control impairments may have a role in later psychosis.
-
Most but not all children with 22q11.2DS develop borderline to low average intellectual functioning abilities, with strengths tending to be in verbal domains and weaknesses in non-verbal domains.
-
Neural abnormalities seem to occur more in the midline of the brain and in subcortical as well as cortical regions.
-
Up to one-third of all individuals carrying the 22q11.2 deletion develop schizophrenia or schizoaffective disorder as defined strictly in the Diagnostic and Statistical Manual of Mental Disorders, although the risk factors that are predictive of the development of psychosis remain unclear and continue to be the focus of active investigation. 22q11.2 deletions account for up to 1–2% of schizophrenia cases and represent the only confirmed recurrent structural mutation responsible for introducing sporadic cases of schizophrenia to the population.
-
Work on genetically engineered animal models of 22q11.2DS has uncovered behavioural and cognitive alterations, as well as a number of affected neural processes (such as compromised dendritogenesis, synaptogenesis, neurogenesis and long-range connectivity) and molecular pathways (including abnormal brain microRNA biogenesis, palmitoylation of proteins and dopaminergic activity) that all have important roles in the observed cognitive and behavioural dysfunction.
-
Human genetic and animal model studies suggest that the combined effect of the imbalance of several genes in the 22q11.2 deletion determines the overall phenotype.
Abstract
Recent studies are beginning to paint a clear and consistent picture of the impairments in psychological and cognitive competencies that are associated with microdeletions in chromosome 22q11.2. These studies have highlighted a strong link between this genetic lesion and schizophrenia. Parallel studies in humans and animal models are starting to uncover the complex genetic and neural substrates altered by the microdeletion. In addition to offering a deeper understanding of the effects of this genetic lesion, these findings may guide analysis of other copy-number variants associated with cognitive dysfunction and psychiatric disorders.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Shprintzen, R. J. et al. A new syndrome involving cleft palate, cardiac anomalies, typical facies, and learning disabilities: velo-cardio-facial syndrome. Cleft Palate J. 15, 56–62 (1978).
DiGeorge, A. A new concept of the cellular basis of immunity. Disabil. Rehabil. 67, 907–908 (1965).
Robin, N. H. & Shprintzen, R. J. Defining the clinical spectrum of deletion. 22q11.2. J. Pediatr. 147, 90–96 (2005).
Kobrynski, L. J. & Sullivan, K. E. Velocardiofacial syndrome, DiGeorge syndrome: the chromosome 22q11.2 deletion syndromes. Lancet 370, 1443–1452 (2007).
Botto, L. D. et al. A population-based study of the 22q11.2 deletion: phenotype, incidence, and contribution to major birth defects in the population. Pediatrics 112, 101–107 (2003).
Urban, A. E. et al. High-resolution mapping of DNA copy alterations in human chromosome 22 using high-density tiling oligonucleotide arrays. Proc. Natl Acad. Sci. USA 103, 4534–4539 (2006).
Edelmann, L., Pandita, R. K. & Morrow, B. E. Low-copy repeats mediate the common 3-Mb deletion in patients with velo-cardio-facial syndrome. Am. J. Hum. Genet. 64, 1076–1086 (1999).
Shaikh, T. H. et al. Chromosome 22-specific low copy repeats and the 22q11.2 deletion syndrome: genomic organization and deletion endpoint analysis. Hum. Mol. Genet. 9, 489–501 (2000).
Maynard, T. M. et al. A comprehensive analysis of 22q11 gene expression in the developing and adult brain. Proc. Natl Acad. Sci. USA 24, 14433–14438 (2003).
Ryan, A. K. et al. Spectrum of clinical features associated with interstitial chromosome 22q11 deletions: a European collaborative study. J. Med. Genet. 34, 798–804 (1997).
Carlson, C. et al. Molecular definition of 22q11 deletions in 151 velo-cardio-facial syndrome patients. Am. J. Hum. Genet. 61, 620–629 (1997).
Sullivan, K. E. The clinical, immunological, and molecular spectrum of chromosome 22q11.2 deletion syndrome and DiGeorge syndrome. Curr. Opin. Allergy Clin. Immunol. 4, 505–512 (2004).
Cook, E. H. Jr & Scherer, S. W. Copy-number variations associated with neuropsychiatric conditions. Nature 455, 919–923 (2008).
De Smedt, B. et al. Mathematical disabilities in children with velo-cardio-facial syndrome. Neuropsychologia 45, 885–895 (2007).
Campbell, L. & Swillen, A. in Velo-Cardio-Facial Syndrome: A Model for Understanding Microdeletion Disorders (eds Murphy, K. C. & Scambler, P. J.) 147–164 (Cambridge Univ. Press, 2005).
Wang, P. P. et al. Research on behavioral phenotypes: velocardiofacial syndrome (deletion 22q11.2). Dev. Med. Child Neurol. 42, 422–427 (2000).
Moss, E. M. et al. Psychoeducational profile of the 22q11.2 microdeletion: a complex pattern. J. Pediatrics 134, 193–198 (1999).
Gerdes, M. et al. Cognitive and behavior profile of preschool children with chromosome 22q11.2 deletion. Am. J. Med. Genet. 85, 127–133 (1999).
Scherer, N. J., D'Antonio, L. L. & Kalbfleisch, J. H. Early speech and language development in children with velocardiofacial syndrome. Am. J. Med. Genet. 88, 714–723 (1999).
Solot, C. B. et al. Communication disorders in the 22Q11.2 microdeletion syndrome. J. Commun. Disord. 33, 187–203 (2000).
Solot, C. B. et al. Communication issues in 22q11.2 deletion syndrome: children at risk. Genet. Med. 3, 67–71 (2001).
Swillen, A. et al. Neuropsychological, learning and psychosocial profile of primary school aged children with the velo-cardio-facial syndrome (22q11 deletion): evidence for a nonverbal learning disability? Child Neuropsychol. 5, 230–241 (1999).
Woodin, M. et al. Neuropsychological profile of children and adolescents with the 22q11.2 microdeletion. Genet. Med. 3, 34–39 (2001). An early comprehensive review of the neuropsychological profile of school-aged children with 22q11.2DS.
Bearden, C. E. et al. The neurocognitive phenotype of the 22q11.2 deletion syndrome: selective deficit in visual-spatial memory. J. Clin. Exper. Neuropsychol. 23, 447–464 (2001).
Lajiness-O'Neill, R. R. et al. Memory and learning in children with 22q11.2 deletion syndrome: evidence for ventral and dorsal stream disruption? Neuropsychol. Dev. Cogn. C Child Neuropsychol. 11, 55–71 (2005).
Sobin, C. et al. Neuropsychological characteristics of children with the 22q11 deletion syndrome: a descriptive analysis. Neuropsychol. Dev. Cogn. C Child Neuropsychol. 11, 39–53 (2005).
Sobin, C. et al. Networks of attention in children with the 22q11 deletion syndrome. Dev. Neuropsychol. 26, 611–626 (2004).
Simon, T. J. et al. Overlapping numerical cognition impairments in children with chromosome 22q11.2 deletion or Turner syndromes. Neuropsychologia 46, 82–94 (2008).
Cavanagh, P. in Cognitive Neuroscience of Attention (ed. Posner, M. I.) 13–28 (Guilford Press, New York, 2004).
Fan, J., McCandliss, B. D., Sommer, T., Raz, A. & Posner, M. I. Testing the efficiency and independence of attentional networks. J. Cogn. Neurosci. 14, 340–347 (2002).
Posner, M. I. & Petersen, S. E. The attention system of human brain. Ann. Rev. Neurosci. 13, 25–42 (1990).
Simon, T. J. et al. Volumetric, connective, and morphologic changes in the brains of children with chromosome 22q11.2 deletion syndrome: an integrative study. Neuroimage 25, 169–180 (2005).
Simon, T. J. et al. A multiple levels analysis of cognitive dysfunction and psychopathology associated with chromosome 22q11.2 deletion syndrome in children. Dev. Psychopathol. 17, 753–784 (2005). An integrative review of evidence from cognitive experimental studies, standardized measures, neuroimaging and genetics concerning children with 22q11.2DS.
Bish, J. P. et al. Domain specific attentional impairments in children with chromosome 22q11.2 deletion syndrome. Brain Cogn. 64, 265–273 (2007).
Corbetta, M. & Shulman, G. L. Control of goal-directed and stimulus-driven attention in the brain. Nature Rev. Neurosci. 3, 201–215 (2002).
Simon, T. J. et al. Atypical cortical connectivity and visuospatial cognitive impairments are related in children with chromosome 22q11.2 deletion syndrome. Behav. Brain Funct. 4, 25 (2008).
Piazza, M., Giacomini, E., Le Bihan, D. & Dehaene, S. Single-trial classification of parallel pre-attentive and serial attentive processes using functional magnetic resonance imaging. Proc. Biol. Sci. 270, 1237–1245 (2003).
Sathian, K. et al. Neural evidence linking visual object enumeration and attention. J. Cogn. Neurosci. 11, 36–51 (1999).
Simon, T. J. & Vaishnavi, S. Subitizing and counting depend on different attentional mechanisms: evidence from visual enumeration in afterimages. Percept. Psychophys. 58, 915–926 (1996).
Debbané, M., Glaser, B., Gex-Fabry, M. & Eliez, S. Temporal perception in velo-cardio-facial syndrome. Neuropsychologia 43, 1754–1762 (2005).
Ansari, D., Lyons, I. M., van Eimeren, L. & Xu, F. Linking visual attention and number processing in the brain: the role of the temporo-parietal junction in small and large symbolic and nonsymbolic number comparison. J. Cogn. Neurosci. 19, 1845–1853 (2007).
Molko, N. et al. Functional and structural alterations of the intraparietal sulcus in a developmental dyscalculia of genetic origin. Neuron 40, 847–858 (2003).
Shuman, M. & Kanwisher, N. Numerical magnitude in the human parietal lobe; tests of representational generality and domain specificity. Neuron 44, 557–569 (2004).
Zorzi, M., Priftis, K., Meneghello, F., Marenzi, R. & Umiltà, C. The spatial representation of numerical and non-numerical sequences: evidence from neglect. Neuropsychologia 44, 1061–1067 (2006).
Bish, J. P. et al. Maladaptive conflict monitoring as evidence for executive dysfunction in children with chromosome 22q11.2 deletion syndrome. Dev. Sci. 8, 36–43 (2005).
Gratton, G., Coles, M. G. & Donchin, E. Optimizing the use of information: strategic control of activation of responses. J. Exp. Psychol. 121, 480–506 (1992).
Takarae, Y., Schmidt, L., Tassone, F. & Simon, T. J. Catechol-O-methyltransferase polymorphism modulates cognitive control in children with chromosome 22q11.2 deletion syndrome. Cogn. Affect. Behav. Neurosci. 9, 83–90 (2009).
Sobin, C., Kiley-Brabeck, K. & Karayiorgou, M. Lower prepulse inhibition in children with the 22q11 deletion syndrome. Am. J. Psychiatry 162, 1090–1099 (2005).
Baker, K. et al. COMT Val108/158Met modifies mismatch negativity and cognitive function in 22q11 deletion syndrome. Biol. Psychiatry 58, 23–31 (2005).
Cheour, M. et al. The first neurophysiological evidence for cognitive brain dysfunctions in children with CATCH. Neuroreport 8, 1785–1787 (1997).
Kates, W. et al. The neural correlates of non-spatial working memory in velocardiofacial syndrome (22q11.2 deletion syndrome). Neuropsychologia 45, 2863–2873 (2007).
van Amelsvoort, T. et al. Cognitive deficits associated with schizophrenia in velo-cardio-facial syndrome. Schizophr. Res. 70, 223–232 (2004).
Karayiorgou, M. et al. Schizophrenia susceptibility associated with interstitial deletions of chromosome 22q11. Proc. Natl Acad. Sci. USA 92, 7612–7616 (1995). A seminal paper that provided the first evidence to support the importance of CNVs in schizophrenia vulnerability.
Xu, B. et al. Strong association of de novo copy number mutations with sporadic schizophrenia. Nature Genet. 40, 880–885 (2008). A systematic study that confirms the extensive contribution of de novo CNVs, such as the 22q11.2 microdeletions, in schizophrenia vulnerability.
Arnold, P. D. et al. Velo-cardio-facial syndrome: implications of microdeletion 22q11 for schizophrenia and mood disorders. Am. J. Med. Genet. 105, 354–362 (2001).
Feinstein, C. et al. Psychiatric disorders and behavioral problems in children with velocardiofacial syndrome: usefulness as phenotypic indicators of schizophrenia risk. Biol. Psychiatry 51, 312–318 (2002).
Antshel, K. M. et al. ADHD, major depressive disorder, and simple phobias are prevalent psychiatric conditions in youth with velocardiofacial syndrome. J. Am. Acad. Child Adolesc. Psychiatry 45, 596–603 (2006).
Antshel, K. M. et al. Autistic spectrum disorders in velo-cardiofacial syndrome (22q11.2 deletion). J. Autism Dev. Disord. 37, 1776–1786 (2007).
Vorstman, J. A. et al. The 22q11.2 deletion in children: high rate of autistic disorders and early onset of psychotic symptoms. J. Am. Acad. Child Adolesc. Psychiatry 45, 1104–1113 (2006).
Flint, J. & Yule W. Behavioural Phenotypes, Child and Adolescent Psychiatry 3rd edn (eds Rutter, M., Taylor, E. & Hersov, L.) 666–687 (Blackwell Scientific, Oxford, 1994).
Flint, J. Behavioral phenotypes: conceptual and methodological issues. Am. J. Med. Genet. 81, 235–240 (1998).
Ogilvie, C. M., Moore, J., Daker, M., Palferman, S. & Docherty, Z. Chromosome 22q11 deletions are not found in autistic patients identified using strict diagnostic criteria. IMGSAC. International Molecular Genetics Study of Autism Consortium. Am. J. Med. Genet. 96, 15–17 (2000).
Elia, J. et al. Rare structural variants found in attention-deficit hyperactivity disorder are preferentially associated with neurodevelopmental genes. Mol. Psychiatry 23 Jun 2009 (doi: 10.1038/mp.2009.57).
Glessner, J. T. et al. Autism genome-wide copy number variation reveals ubiquitin and neuronal genes. Nature 459, 569–573 (2009).
Eliez, S. Autism in children with 22Q11.2 deletion syndrome. J. Am. Acad. Child Adolesc. Psychiatry 46, 433–434 (2007).
Pulver, A. E. et al. Psychotic illness in patients diagnosed with velo-cardio-facial syndrome and their relatives. J. Nerv. Ment. Dis. 182, 476–478 (1994).
Murphy, K. C., Jones, L. A. & Owen, M. J. High rates of schizophrenia in adults with velocardio-facial syndrome. Arch. Gen. Psychiatry 56, 940–945 (1999).
Gothelf, D. et al. Risk factors for the emergence of psychotic disorders in adolescents with 22q11.2 deletion syndrome. Am. J. Psychiatry 164, 663–669 (2007).
Green, T. et al. Psychiatric disorders and intellectual functioning throughout development in velocardiofacial (22q11.2 deletion) syndrome. J. Am. Acad. Child Adolesc. Psychiatry 48, 1060–1068 (2009).
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders 4th edn (American Psychiatric Publishing, Washington, DC, 1994).
International Schizophrenia Consortium. Rare chromosomal deletions and duplications increase risk of schizophrenia. Nature 455, 237–241 (2008). A comprehensive large-scale study that confirmed the role of 22q11.2 microdeletions in schizophrenia and identified additional candidate pathogenic CNVs.
Stefansson, H. et al. Large recurrent microdeletions associated with schizophrenia. Nature 455, 232–236 (2008).
Bassett, A. S. et al. The schizophrenia phenotype in 22q11 deletion syndrome. Am. J. Psychiatry 160, 1580–1586 (2003).
Bassett, A. S. et al. 22q11 deletion syndrome in adults with schizophrenia. Am. J. Med. Genet. 81, 328–337 (1998).
Johnson, M. H., Halit, H., Grice, S. J. & Karmiloff-Smith, A. Neuroimaging of typical and atypical development: a perspective from multiple levels of analysis. Dev. Psychopathol. 14, 521–536 (2002). An important review of special interpretive assumptions as they relate to neuroimaging studies of typically and, more importantly, atypically developing children.
Campbell, L. E. et al. Brain and behaviour in children with 22q11.2 deletion syndrome: a volumetric and voxel-based morphometry MRI study. Brain 129, 1218–1228 (2006).
Eliez, S. et al. Children and adolescents with velocardiofacial syndrome: a volumetric study. Am. J. Psychiatry 3, 409–415 (2000).
Kates, W. R. et al. Regional cortical white matter reductions in velocardiofacial syndrome: a volumetric MRI analysis. Biol. Psychiatry 49, 677–684 (2001).
Kates, W. R. et al. Frontal and caudate alterations in velocardiofacial syndrome (deletion at chromosome 22q11.2). J. Child Neurol. 5, 337–342 (2004).
Simon, T. J. et al. Visuospatial and numerical cognitive deficits in children with chromosome 22q11.2 deletion syndrome. Cortex 2, 145–155 (2005).
Tan, G. et al. Meta-analysis of magnetic resonance imaging studies in chromosome 22q11.2 deletion syndrome (velocardiofacial syndrome). Schizophr. Res. 115, 173–181 (2009). An extensive review of structural neuroimaging findings in 22q11.2DS.
Bearden, C. et al. Mapping cortical thickness in children with 22q11.2 deletions. Cereb. Cortex 8, 1889–1898 (2006).
Bearden, C. E. et al. Alterations in midline cortical thickness and gyrification patterns mapped in children with 22q11.2 deletions. Cereb. Cortex 19, 115–126 (2009).
Schaer, M. et al. Abnormal patterns of cortical gyrification in velo-cardio-facial syndrome (deletion 22q11.2): an MRI study. Psychiatry Res. 146, 1–11 (2006).
Schaer, M. et al. Congenital heart disease affects local gyrification in 22q11.2 deletion syndrome. Dev. Med. Child Neurol. 51, 746–753 (2009).
Van Essen, D. C. et al. Symmetry of cortical folding abnormalities in Williams syndrome revealed by surface-based analyses. J. Neurosci. 26, 5470–5483 (2006).
Barnea-Goraly, N. et al. Investigation of white matter structure in velocardiofacial syndrome: a diffusion tensor imaging study. Am. J. Psychiatry 160, 1863–1869 (2003).
Machado, A. M. et al. Corpus callosum morphology and ventricular size in chromosome 22q11.2 deletion syndrome. Brain Res. 1131, 197–210 (2007).
Barnea-Goraly, N. et al. Arithmetic ability and parietal alterations: a diffusion tensor imaging study in velocardiofacial syndrome. Brain Res. Cogn. Brain Res. 25, 735–740 (2005).
Eliez, S. et al. Functional brain imaging study of mathematical reasoning abilities in velocardiofacial syndrome (del22q11.2). Genet. Med. 3, 49–55 (2001).
Gothelf, D. et al. Abnormal cortical activation during response inhibition in 22q11.2 deletion syndrome. Hum. Brain Mapp. 28, 533–542 (2007).
van Amelsvoort, T. et al. Brain anatomy in adults with velocardiofacial syndrome with and without schizophrenia: preliminary results of a structural magnetic resonance imaging study. Arch. Gen. Psychiatry 61, 1085–1096 (2004).
Chow, E. W. et al. Structural brain abnormalities in patients with schizophrenia and 22q11 deletion syndrome. Biol. Psychiatry 51, 208–215 (2002).
Chow, E. W., Watson, M., Young, D. A. & Bassett, A. S. Neurocognitive profile in 22q11 deletion syndrome and schizophrenia. Schizophr. Res. 87, 270–278 (2006).
Gothelf, D. et al. COMT genotype predicts longitudinal cognitive decline and psychosis in 22q11.2 deletion syndrome. Nature Neurosci. 8, 1500–1502 (2005).
Raux, G. et al. Involvement of hyperprolinemia in cognitive and psychiatric features of the 22q11 deletion syndrome. Hum. Mol. Genet. 16, 83–91 (2007).
Vorstman, J. A. et al. Proline affects brain function in 22q11DS children with the low activity COMT158 allele. Neuropsychopharmacology 34, 739–746 (2009).
Taddei, I. et al. Genetic factors are major determinants of phenotypic variability in a mouse model of the DiGeorge/del22q11 syndromes. Proc. Natl Acad. Sci. USA 98, 11428–11431 (2001).
Karayiorgou, M. & Gogos, J. A. The molecular genetics of the 22q11-associated schizophrenia. Brain Res. Mol. Brain Res. 132, 95–104 (2004).
Arinami, T. Analyses of the associations between the genes of 22q11 deletion syndrome and schizophrenia. J. Hum. Genet. 51, 1037–1045 (2006).
Mukai, J. et al. Evidence that the gene encoding ZDHHC8 contributes to the risk of schizophrenia. Nature Genet. 36, 725–731 (2004).
Liu, H. et al. Genetic variation in the 22q11 locus and susceptibility to schizophrenia. Proc. Natl Acad. Sci. USA 26, 16859–16864 (2002).
Liu, H. et al. Genetic variation at the 22q11 PRODH2/DGCR6 locus presents an unusual pattern and increases susceptibility to schizophrenia. Proc. Natl Acad. Sci. USA 99, 3717–3722 (2002).
Karayiorgou, M. et al. Genotype determining low catechol-O-methyltransferase activity as a risk factor for obsessive-compulsive disorder. Proc. Natl Acad. Sci. USA 94, 4572–4575 (1997).
Karayiorgou, M. et al. Family-based association studies support a sexually dimorphic effect of COMT and MAOA on genetic susceptibility to obsessive-compulsive disorder. Biol. Psychiatry 45, 1178–1189 (1999).
Pooley, E. C., Fineberg, N. & Harrison, P. J. The met158 allele of catechol-O-methyltransferase (COMT) is associated with obsessive-compulsive disorder in men: case–control study and meta-analysis. Mol. Psychiatry 12, 556–561 (2007).
Williams, N. M. et al. Strong evidence that GNB1L is associated with schizophrenia. Hum. Mol. Genet. 17, 555–566 (2008).
Goldstein, D. B. Common genetic variation and human traits. N. Engl. J. Med. 360, 1696–1698 (2009).
Arguello, P. A. & Gogos, J. A. Modeling madness in mice: one piece at a time. Neuron 52, 179–196 (2006).
Wise, S. P. Forward frontal fields: phylogeny and fundamental function. Trends Neurosci. 31, 599–608 (2008).
Meyer-Lindenberg, A. S. et al. Regionally specific disturbance of dorsolateral prefrontal-hippocampal functional connectivity in schizophrenia. Arch. Gen. Psychiatry 62, 379–386 (2005).
Lawrie, S. M. et al. Reduced frontotemporal functional connectivity in schizophrenia associated with auditory hallucinations. Biol. Psychiatry 51, 1008–1011 (2002).
Ford, J. M., Mathalon, D. H., Whitfield, S., Faustman, W. O. & Roth, W. T. Reduced communication between frontal and temporal lobes during talking in schizophrenia. Biol. Psychiatry 51, 485–492 (2002).
Stark, K. L. et al. Altered brain microRNA biogenesis contributes to phenotypic deficits in a 22q11-deletion mouse model. Nature Genet. 40, 751–760 (2008). This paper provided compelling evidence that the 22q11.2 microdeletion results in abnormal processing of brain miRNAs, which results in altered neuronal connectivity, behaviour and cognition in mice, including deficits in WM.
Ambros, V. The functions of animal microRNAs. Nature 431, 350–355 (2004).
Tomari, Y. & Zamore, P. D. MicroRNA biogenesis: drosha can't cut it without a partner. Curr. Biol. 15, R61–R64 (2005).
Hornstein, E. & Shomron, N. Canalization of development by microRNAs. Nature Genet. 38, S20–S24 (2006).
Mukai, J. et al. Palmitoylation-dependent neurodevelopmental deficits in a mouse model of 22q11 microdeletion. Nature Neurosci. 11, 1302–1310 (2008). This paper provided evidence that the 22q11.2 microdeletion results in impaired development of dendrites, dendritic spines and excitatory synapses, in part due to abnormal palmitoylation of neuronal proteins.
Yuste, R. & Tank, D. W. Dendritic integration in mammalian neurons, a century after Cajal. Neuron 16, 701–716 (1996).
Mainen, Z. F. & Sejnowski, T. J. Influence of dendritic structure on firing pattern in model neocortical neurons. Nature 382, 363–366 (1996).
Meechan, D. W., Tucker, E. S., Maynard, T. M. & LaMantia, A. S. Diminished dosage of 22q11 genes disrupts neurogenesis and cortical development in a mouse model of 22q11 deletion/DiGeorge syndrome. Proc. Natl Acad. Sci. USA 106, 16434–16445 (2009). This paper provided evidence that diminished dosage of 22q11.2 genes subtly compromises neurogenesis and subsequent neuronal differentiation in the cerebral cortex.
Kosik, K. S. The neuronal microRNA system. Nature Rev. Neurosci. 7, 911–920 (2006).
Fiore, R. et al. Mef2-mediated transcription of the miR379–410 cluster regulates activity-dependent dendritogenesis by fine-tuning Pumilio2 protein levels. EMBO J. 28, 697–710 (2009).
Schratt, G. M. et al. A brain-specific microRNA regulates dendritic spine development. Nature 439, 283–289 (2006); erratum in 441, 902 (2006).
Fukata, M., Fukata, Y., Adesnik, H., Nicoll, R. A. & Bredt, D. S. Identification of PSD-95 palmitoylating enzymes. Neuron 44, 987–996 (2004).
Mitchell, D. A., Vasudevan, A., Linder, M. E. & Deschenes, R. J. Protein palmitoylation by a family of DHHC protein S-acyltransferases. J. Lipid Res. 47, 1118–1127 (2006).
Ohno, Y., Kihara, A., Sano, T. & Igarashi, Y. Intracellular localization and tissue-specific distribution of human and yeast DHHC cysteine-rich domain-containing proteins. Biochim. Biophys. Acta 1761, 474–483 (2006).
Maynard, T. M. et al. Mitochondrial localization and function of a subset of 22q11 deletion syndrome candidate genes. Mol. Cell. Neurosci. 39, 439–451 (2008).
El-Husseini, A. E. D. & Bredt, D. S. Protein palmitoylation: a regulator of neuronal development and function. Nature Rev. Neurosci. 3, 791–802 (2002).
Kang, R. et al. Neural palmitoyl-proteomics reveals dynamic synaptic palmitoylation. Nature 456, 904–909 (2008).
Hsu, R. et al. Nogo receptor 1 (RTN4R) as a candidate gene for schizophrenia: analysis using human and mouse genetic approaches. PLoS ONE 2, e1234 (2007).
Tsang, C. W. et al. Superfluous role of mammalian septins 3 and 5 in neuronal development and synaptic transmission. Mol. Cell. Biol. 28, 7012–7029 (2008).
Su, Q., Mochida, S., Tian, J. H., Mehta, R. & Shen, Z. H. SNAP-29: a general SNARE protein that inhibits SNARE disassembly and is implicated in synaptic transmission. Proc. Natl Acad. Sci. USA 98, 14038–14043 (2001).
Park, T. J. & Curran, T. Crk and Crk-like play essential overlapping roles downstream of disabled-1 in the Reelin pathway. J. Neurosci. 28, 13551–13562 (2008).
Sprecher, E. et al. A mutation in SNAP29, coding for a SNARE protein involved in intracellular trafficking, causes a novel neurocutaneous syndrome characterized by cerebral dysgenesis, neuropathy, ichthyosis, and palmoplantar keratoderma. Am. J. Hum. Genet. 77, 242–251 (2005).
Jurata, L. W. et al. Altered expression of hippocampal dentate granule neuron genes in a mouse model of human 22q11 deletion syndrome. Schizophr. Res. 88, 251–259 (2006).
Yavich, L., Forsberg, M. M., Karayiorgou, M., Gogos, J. A. & Männistö, P. T. Site-specific role of catechol-O-methyltransferase in dopamine overflow within prefrontal cortex and dorsal striatum. J. Neurosci. 27, 10196–10209 (2007).
Huotari, M. et al. Brain catecholamine metabolism in catechol-O-methyltransferase (COMT)-deficient mice. Eur. J. Neurosci. 15, 246–256 (2002).
Huotari, M., García-Horsman, J. A., Karayiorgou, M., Gogos, J. A. & Männistö, P. T. D-Amphetamine responses in catechol-O-methyltransferase (COMT) disrupted mice. Psychopharmacology (Berlin) 172, 1–10 (2004).
Hayward, D. C. et al. The sluggish-A gene of Drosophila melanogaster is expressed in the nervous system and encodes proline oxidase, a mitochondrial enzyme involved in glutamate biosynthesis. Proc. Natl Acad. Sci. USA 90, 2979–2983 (1993).
Goodman, B. K., Rutberg, J., Lin, W. W., Pulver, A. E. & Thomas, G. H. Hyperprolinaemia in patients with deletion (22)(q11.2) syndrome. J. Inherit. Metab. Dis. 23, 847–848 (2000).
Jacquet, H. et al. The severe form of type I hyperprolinaemia results from homozygous inactivation of the PRODH gene. J. Med. Genet. 40, e7 (2003).
Jacquet, H. et al. PRODH mutations and hyperprolinemia in a subset of schizophrenic patients. Hum. Mol. Genet. 11, 2243–2249 (2002).
Jacquet, H. et al. Hyperprolinemia is a risk factor for schizoaffective disorder. Mol. Psychiatry 10, 479–485 (2005).
Gogos, J. A. et al. The gene encoding proline dehydrogenase modulates sensorimotor gating in mice. Nature Genet. 21, 434–439 (1999).
Paterlini, M. et al. Transcriptional and behavioral interaction between 22q11.2 orthologs modulates schizophrenia-related phenotypes in mice. Nature Neurosci. 8, 1586–1594 (2005). This paper and reference 96 provided the first demonstration of an epistatic interaction between two candidate schizophrenia risk genes in the 22q11.2 locus and showed the power of iterative human genetic and animal model studies for dissecting the genetic and neural substrates of the 22q11.2DS.
Arguello, P. A. & Gogos, J. A. Cognition in mouse models of schizophrenia susceptibility genes. Schizophr. Bull. 36, 289–300 (2010).
Paylor, R. et al. Mice deleted for the DiGeorge/velocardiofacial syndrome region show abnormal sensorimotor gating and learning and memory impairments. Hum. Mol. Genet. 10, 2645–2650 (2001).
Kimber, W. L. et al. Deletion of 150 kb in the minimal DiGeorge/velocardiofacial syndrome critical region in mouse. Hum. Mol. Genet. 8, 2229–2237 (1999).
Gogos, J. A. et al. Catechol-O-methyltransferase-deficient mice exhibit sexually dimorphic changes in catecholamine levels and behavior. Proc. Natl Acad. Sci. USA 95, 9991–9996 (1998).
Papaleo, F. et al. Genetic dissection of the role of catechol-O-methyltransferase in cognition and stress reactivity in mice. J. Neurosci. 28, 8709–8723 (2008).
Babovic, D. et al. Phenotypic characterization of cognition and social behavior in mice with heterozygous versus homozygous deletion of catechol-O-methyltransferase. Neuroscience 155, 1021–1029 (2008).
Long, J. M. et al. Behavior of mice with mutations in the conserved region deleted in velocardiofacial/DiGeorge syndrome. Neurogenetics 7, 247–257 (2006).
Paylor, R. et al. Tbx1 haploinsufficiency is linked to behavioral disorders in mice and humans: implications for 22q11 deletion syndrome. Proc. Natl Acad. Sci. USA 103, 7729–7734 (2006). This paper described the first traditional deletion-mapping approach to dissect genotype–phenotype relationships in the 22q11.2 locus.
Nitta, T. et al. Size-selective loosening of the blood–brain barrier in claudin-5-deficient mice. J. Cell Biol. 161, 653–660 (2003).
Steriade, M., Gloor, P., Llinas, R. R., Lopes de Silva, F. H. & Mesulam, M. M. Report of IFCN Committee on Basic Mechanisms. Basic mechanisms of cerebral rhythmic activities. Electroencephalogr. Clin. Neurophysiol. 76, 481–508 (1990).
Singer, W. Neuronal synchrony: a versatile code for the definition of relations? Neuron 24, 49–65 (1999).
Jones, M. W. & Wilson, M. A. Theta rhythms coordinate hippocampal–prefrontal interactions in a spatial memory task. PLoS Biol. 3, e402 (2005).
Sigurdsson, T., Stark, K. L., Karayiorgou, M., Gogos, J. A. & Gordon, J. A. Impaired hippocampal–prefrontal synchrony in a genetic mouse model of schizophrenia. Nature 464, 763–767 (2010). Building on the findings of reference 114, this paper provides compelling evidence that impaired long-range connectivity and synchrony of neural activity is one consequence of the 22q11.2 deletion and could be a fundamental component of the pathophysiology underlying schizophrenia.
Bertolino, A. et al. Prefrontal–hippocampal coupling during memory processing is modulated by COMT val158met genotype. Biol. Psychiatry 60, 1250–1258 (2006).
Esslinger, C. et al. Neural mechanisms of a genome-wide supported psychosis variant. Science 324, 605 (2009).
Bodmer, W. & Bonilla, C. Common and rare variants in multifactorial susceptibility to common diseases. Nature Genet. 40, 695–701 (2008).
Van Veen, V. & Carter, C. S. The anterior cingulated as a conflict monitor: fMRI and ERP studies. Physiol. Behav. 77, 477–482 (2002).
Devrim-Uçok, M., Keskin-Ergen, H. Y. & Uçok, A. Mismatch negativity at acute and post-acute phases of first-episode schizophrenia. Eur. Arch. Psychiatry Clin. Neurosci. 258, 179–185 (2008).
Carroll, C. A., Boggs, J., O'Donnell, B. F., Shekhar, A. & Hetrick, W. P. Temporal processing dysfunction in schizophrenia. Brain Cogn. 67, 150–161 (2008).
Elvevåg, B. & Goldberg, T. E. Cognitive impairment in schizophrenia is the core of the disorder. Crit. Rev. Neurobiol. 14, 1–21 (2000).
Debbané, M., Glaser, B., David, M. K., Feinstein, C. & Eliez, S. Psychotic symptoms in children and adolescents with 22q11.2 deletion syndrome: neuropsychological and behavioral implications. Schizophr. Res. 84, 187–193 (2006).
Sugama, S. et al. Morphometry of the head of the caudate nucleus in patients with velocardiofacial syndrome (del 22q11.2). Acta Paediatr. 89, 546–549 (2000).
Eliez, S. et al. Increased basal ganglia volumes in velo-cardio-facial syndrome (deletion 22q11.2). Biol. Psychiatry 52, 68–70 (2002).
Eliez, S. et al. A quantitative MRI study of posterior fossa development in velocardiofacial syndrome. Biol. Psychiatry 49, 540–546 (2001).
Bish, J. P. et al. Specific cerebellar reductions in children with chromosome 22q11.2 deletion syndrome. Neurosci. Lett. 399, 245–248 (2006).
Fanselow, M. S. & Poulos, A. M. The neuroscience of mammalian associative learning. Annu Rev. Psychol. 56, 207–234 (2005).
Aultman, J. M. & Moghaddam, B. Distinct contributions of glutamate and dopamine receptors to temporal aspects of rodent working memory using a clinically relevant task. Psychopharmacology (Berlin) 153, 353–364 (2001).
Lee, I. & Kesner, R. P. Time-dependent relationship between the dorsal hippocampus and the prefrontal cortex in spatial memory. J. Neurosci. 23, 1517–1523 (2003).
Goldberg, M. C., Maurer, D. & Lewis, T. L. Developmental changes in attention: the effects of endogenous cueing and of distractors. Dev. Sci. 4, 209–219 (2001).
Posner, M. I. et al. Effects of parietal injury on covert orienting of attention. J. Neurosci. 4, 1863–1874 (1984).
Rueda, M. R. et al. Development of attentional networks in childhood. Neuropsychologia 42, 1029–1040 (2004).
Egly, R., Driver, J. & Rafal, R. D. Shifting visual attention between objects and locations: evidence from normal and parietal lesion subjects. J. Exp. Psychol. Gen. 123, 161–177 (1994).
Klein, R. M. Inhibition of return. Trends Cogn. Sci. 4, 138–147 (2000).
MacPherson, A. C., Klein, R. M. & Moore, C. Inhibition of return in children and adolescents. J. Exp. Child Psychol. 85, 337–351 (2003).
Chi, M. T. & Klahr, D. Span and rate of apprehension in children and adults. J. Exp. Child Psychol. 19, 434–439 (1975).
Trick, L. M. & Pylyshyn, Z. W. Why are small and large numbers enumerated differently? A limited-capacity preattentive stage in vision. Psychol. Rev. 101, 80–102 (1994).
Ansari, D. & Karmiloff-Smith, A. Atypical trajectories of number development: a neuroconstructivist perspective. Trends Cogn. Sci. 6, 511–516 (2002).
Dehaene, S. & Cohen, L. Towards an anatomical and functional model of number processing. Math. Cogn. 1, 83–120 (1995).
Ornitz, E. M., Guthrie, D., Kaplan, A. R., Lane, S. J. & Norman, R. J. Maturation of startle modulation. Psychophysiology 23, 624–634 (1986).
Ahmmed, A. U., Clarke, E. M. & Adams, C. Mismatch negativity and frequency representational width in children with specific language impairment. Dev. Med. Child Neurol. 50, 938–944 (2008).
Näätänen, R. Mismatch negativity (MMN): perspectives for application. Int. J. Psychophysiol. 37, 3–10 (2000).
Casey, B. J. et al. Activation of prefrontal cortex in children during a nonspatial working memory task with functional MRI. Neuroimage 2, 221–229 (1995).
Durston, S., Thomas, K. M., Worden, M. S., Yang, Y. & Casey, B. J. The effect of preceding context on inhibition: an event-related fMRI study. Neuroimage 16, 449–453 (2002).
Schulz, K. P. et al. Response inhibition in adolescents diagnosed with attention deficit hyperactivity disorder during childhood: an event-related FMRI study. Am. J. Psychiatry 161, 1650–1657 (2004).
Acknowledgements
The authors wish to acknowledge grant support for their work from the US National Institutes of Health (grant R01MH67068 to M.K. and J.A.G. and grant R01HD42974 to T.J.S.). J.A.G. also gratefully acknowledges support from the Simons Foundation and M.K. from the McKnight and March of Dimes Foundations. The authors wish to thank A. Arguello for help with creating figures 2,3 and 4, B. Xu for help with Supplementary information S1 (table) and Supplementary information S3 (figure) and S. Srivastava for help with box 3 and figure 1. The authors also thank A. Arguello, L. Drew, B. Xu and other members of their laboratories for comments and critical feedback.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information S1 (table)
RefSeq genes located in the most common 3-Mb 22q11.2 microdeletiona (PDF 86 kb)
Supplementary information S2 (table)
DTI results from children with 22q11.2DS (PDF 84 kb)
Supplementary information S3 (figure)
miRNA dysregulation and altered gene expression in 22q11.2 animal models. (PDF 397 kb)
Related links
Glossary
- Microdeletion
-
A submicroscopic loss of a segment of DNA of varying size, typically several kilobases long.
- Breakpoint
-
A specific site of chromosomal breakage associated with a chromosomal abnormality.
- Full scale IQ
-
A standardized composite measure of global intellectual functioning generated from scores in specific domains, such as verbal, perceptual, memory and speeded functions. Typically the median age-adjusted score is 100 ± 15 points.
- Attention
-
A cognitive process that is mainly thought to be involved in selectively processing or focusing on one aspect of the environment at the expense of others. Several types of attention are thought to exist, including focused, sustained and divided attention, each of which seems to depend on different cognitive, neural and neurotransmitter systems.
- Executive function
-
Also referred to as 'cognitive control', this is a broad category of cognitive functions that are generally associated in typical humans with the prefrontal cortex. Executive functions are thought to modulate or control the use of other cognitive resources and include planning, problem solving, error monitoring, decision making and the use of working memory.
- Prepulse inhibition
-
A reduction in the magnitude of the startle reflex that occurs when an organism is presented with a non-startling stimulus (a prepulse) before being presented with the startling stimulus. Deficits in prepulse inhibition have been observed in patients with schizophrenia as well as in patients with other psychiatric and neurological disorders.
- Mismatch negativity
-
A component of the electro-encephalographic (EEG) brain response that is typically generated 150–250 ms after an unusual stimulus is detected in a sequence of similar stimuli.
- Diffusion tensor imaging
-
An MRI imaging technique that takes advantage of the restricted diffusion of water through myelinated nerve fibres in the brain to map the anatomical connectivity among brain areas.
- Genetic modifiers
-
Genetic variation in (cis) or outside (trans) a gene or genetic locus that alters the phenotypic expression of the gene.
- Oligogenic
-
A phenotypic trait produced by two or more (but only a few) genes working together.
- Next-generation sequencing
-
High-throughput parallel sequencing of several megabases of DNA.
- Haploinsufficiency
-
The situation in which one copy of a gene is incapable of providing sufficient protein production to ensure normal function.
- Endophenotype
-
A state-independent biomarker or cognitive marker of an illness (present whether or not the illness is active) that is heritable and present in unaffected relatives of subjects that have the illness.
Rights and permissions
About this article
Cite this article
Karayiorgou, M., Simon, T. & Gogos, J. 22q11.2 microdeletions: linking DNA structural variation to brain dysfunction and schizophrenia. Nat Rev Neurosci 11, 402–416 (2010). https://doi.org/10.1038/nrn2841
Issue Date:
DOI: https://doi.org/10.1038/nrn2841
This article is cited by
-
Atypical cortical networks in children at high-genetic risk of psychiatric and neurodevelopmental disorders
Neuropsychopharmacology (2024)
-
Event-related potential (ERP) markers of 22q11.2 deletion syndrome and associated psychosis
Journal of Neurodevelopmental Disorders (2023)
-
DiGeorge syndrome critical region gene 2 (DGCR2), a schizophrenia risk gene, regulates dendritic spine development through cell adhesion
Cell & Bioscience (2023)
-
Altered corollary discharge signaling in the auditory cortex of a mouse model of schizophrenia predisposition
Nature Communications (2023)
-
Structural Connectivity and Emotion Recognition Impairment in Children and Adolescents with Chromosome 22q11.2 Deletion Syndrome
Journal of Autism and Developmental Disorders (2023)