Abstract
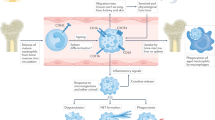
Systemic lupus erythematosus (SLE) is an autoimmune disease of unclear etiology that affects mostly women of childbearing age. Profound abnormalities in both innate and adaptive immunity triggered by genetic and environmental factors are well documented to play an important part in the pathogenesis of SLE. Nonetheless, the role of neutrophils—the most abundant immune cell type—in the pathology of this disease has been unclear. Over the past decade, compelling evidence has emerged that implicates neutrophils in the initiation and perpetuation of SLE and also in the resultant organ damage frequently observed in patients with this disease. SLE-derived low-density granulocytes (LDGs) induce vascular damage and synthesize increased amounts of type I interferons and, as such, could play a prominent part in the pathogenesis of SLE. Furthermore, increased cell death and enhanced extracellular trap formation observed in SLE-derived neutrophils might have key roles in the induction of autoimmunity and the development of organ damage in patients with SLE. Together, these events could have significant deleterious effects and promote aberrant immune responses in this disease. This Review highlights the role of neutrophils in the pathogenesis of SLE, with a particular focus on the putative deleterious effects of LDGs and neutrophil extracellular trap formation.
Key Points
-
Patients with systemic lupus erythematosus (SLE) display marked abnormalities in neutrophil phenotype and function, and enhanced neutrophil death through apoptosis and 'NETosis'
-
A distinct subset of proinflammatory low-density granulocytes isolated from patients with SLE induces vascular damage, displays enhanced bactericidal gene signatures and synthesizes increased amounts of type I IFNs
-
Enhanced NETosis observed in SLE-derived neutrophils might have key roles in the induction of autoimmunity and the development of organ damage in SLE
-
Neutrophil dysfunction and increased NETosis might contribute to SLE pathology and disease manifestations, such as vascular complications, lupus nephritis and cutaneous lupus erythematosus
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Crispin, J. C., Kyttaris, V. C., Terhorst, C. & Tsokos, G. C. T cells as therapeutic targets in SLE. Nat. Rev. Rheumatol. 6, 317–325 (2010).
Dorner, T., Jacobi, A. M., Lee, J. & Lipsky, P. E. Abnormalities of B cell subsets in patients with systemic lupus erythematosus. J. Immunol. Methods 363, 187–197 (2011).
Denny, M. F. et al. Accelerated macrophage apoptosis induces autoantibody formation and organ damage in systemic lupus erythematosus. J. Immunol. 176, 2095–2104 (2006).
Denny, M. F. et al. Interferon-α promotes abnormal vasculogenesis in lupus: a potential pathway for premature atherosclerosis. Blood 110, 2907–2915 (2007).
Denny, M. F. et al. A distinct subset of proinflammatory neutrophils isolated from patients with systemic lupus erythematosus induces vascular damage and synthesizes type I IFNs. J. Immunol. 184, 3284–3297 (2010).
Bennett, L. et al. Interferon and granulopoiesis signatures in systemic lupus erythematosus blood. J. Exp. Med. 197, 711–723 (2003).
Blanco, P., Palucka, A. K., Gill, M., Pascual, V. & Banchereau, J. Induction of dendritic cell differentiation by IFN-α in systemic lupus erythematosus. Science 294, 1540–1543 (2001).
Ding, D., Mehta, H., McCune, W. J. & Kaplan, M. J. Aberrant phenotype and function of myeloid dendritic cells in systemic lupus erythematosus. J. Immunol. 177, 5878–5889 (2006).
Banchereau, J. & Pascual, V. Type I interferon in systemic lupus erythematosus and other autoimmune diseases. Immunity 25, 383–392 (2006).
Kaplan, M. J. Apoptosis in systemic lupus erythematosus. Clin. Immunol. 112, 210–218 (2004).
Hua, J., Kirou, K., Lee, C. & Crow, M. K. Functional assay of type I interferon in systemic lupus erythematosus plasma and association with anti-RNA binding protein autoantibodies. Arthritis Rheum. 54, 1906–1916 (2006).
Kariuki, S. et al. Cutting edge: autoimmune disease risk variant of STAT4 confers increased sensitivity to IFN-α in lupus patients in vivo. J. Immunol. 182, 34–38 (2009).
Lee, P. Y. et al. A novel type I IFN-producing cell subset in murine lupus. J. Immunol. 180, 5101–5108 (2008).
Kaplan, M. J. & Salmon, J. E. How does interferon-α insult the vasculature? Let me count the ways. Arthritis Rheum. 63, 334–336 (2011).
Holman, H. R. The L. E. cell phenomenon. Annu. Rev. Med. 11, 231–242 (1960).
Henson, P. M. Pathologic mechanisms in neutrophil-mediated injury. Am. J. Pathol. 68, 593–612 (1972).
Kobayashi, S. D. & DeLeo, F. R. Role of neutrophils in innate immunity: a systems biology-level approach. Wiley Interdiscip. Rev. Syst. Biol. Med. 1, 309–333 (2009).
Brinkmann, V. et al. Neutrophil extracellular traps kill bacteria. Science 303, 1532–1535 (2004).
Faurschou, M. & Borregaard, N. Neutrophil granules and secretory vesicles in inflammation. Microbes Infect. 5, 1317–1327 (2003).
Lande, R. et al. Plasmacytoid dendritic cells sense self-DNA coupled with antimicrobial peptide. Nature 449, 564–569 (2007).
Murphy, H. S., Bakopoulos, N., Dame, M. K., Varani, J. & Ward, P. A. Heterogeneity of vascular endothelial cells: differences in susceptibility to neutrophil-mediated injury. Microvasc. Res. 56, 203–211 (1998).
Marzocchi-Machado, C. M. et al. Fcgamma and complement receptors: expression, role and co-operation in mediating the oxidative burst and degranulation of neutrophils of Brazilian systemic lupus erythematosus patients. Lupus 11, 240–248 (2002).
Shirafuji, N. et al. Granulocyte colony-stimulating factor stimulates human mature neutrophilic granulocytes to produce interferon-α. Blood 75, 17–19 (1990).
Tamassia, N. et al. Activation of an immunoregulatory and antiviral gene expression program in poly(I:C)-transfected human neutrophils. J. Immunol. 181, 6563–6573 (2008).
Abramson, S. B., Given, W. P., Edelson, H. S. & Weissmann, G. Neutrophil aggregation induced by sera from patients with active systemic lupus erythematosus. Arthritis Rheum. 26, 630–636 (1983).
Brandt, L. & Hedberg, H. Impaired phagocytosis by peripheral blood granulocytes in systemic lupus erythematosus. Scand. J. Haematol. 6, 348–353 (1969).
Courtney, P. A. et al. Increased apoptotic peripheral blood neutrophils in systemic lupus erythematosus: relations with disease activity, antibodies to double stranded DNA, and neutropenia. Ann. Rheum. Dis. 58, 309–314 (1999).
Hsieh, S. C. et al. Abnormal in vitro CXCR2 modulation and defective cationic ion transporter expression on polymorphonuclear neutrophils responsible for hyporesponsiveness to IL-8 stimulation in patients with active systemic lupus erythematosus. Rheumatology (Oxford) 47, 150–157 (2008).
Wu, C. H., Hsieh, S. C., Li, K. J., Lu, M. C. & Yu, C. L. Premature telomere shortening in polymorphonuclear neutrophils from patients with systemic lupus erythematosus is related to the lupus disease activity. Lupus 16, 265–272 (2007).
Molad, Y., Buyon, J., Anderson, D. C., Abramson, S. B. & Cronstein, B. N. Intravascular neutrophil activation in systemic lupus erythematosus (SLE): dissociation between increased expression of CD11b/CD18 and diminished expression of L-selectin on neutrophils from patients with active SLE. Clin. Immunol. Immunopathol. 71, 281–286 (1994).
Arvieux, J., Jacob, M. C., Roussel, B., Bensa, J. C. & Colomb, M. G. Neutrophil activation by anti-β2 glycoprotein I monoclonal antibodies via Fcγ receptor II. J. Leukoc. Biol. 57, 387–394 (1995).
Ronnefarth, V. M. et al. TLR2/TLR4-independent neutrophil activation and recruitment upon endocytosis of nucleosomes reveals a new pathway of innate immunity in systemic lupus erythematosus. J. Immunol. 177, 7740–7749 (2006).
Sthoeger, Z. M., Bezalel, S., Chapnik, N., Asher, I. & Froy, O. High α-defensin levels in patients with systemic lupus erythematosus. Immunology 127, 116–122 (2009).
Vordenbaumen, S. et al. Elevated levels of human β-defensin 2 and human neutrophil peptides in systemic lupus erythematosus. Lupus 19, 1648–1653, (2010).
Ma, C. Y. et al. Elevated plasma level of HMGB1 is associated with disease activity and combined alterations with IFN-α and TNF-α in systemic lupus erythematosus. Rheumatol. Int. http://dx.doi.org/10.1007/s00296-010-1636-6.
Bakkaloglu, A. et al. Antineutrophil cytoplasmic antibodies in childhood systemic lupus erythematosus. Clin. Rheumatol. 17, 265–267 (1998).
Ren, Y. et al. Increased apoptotic neutrophils and macrophages and impaired macrophage phagocytic clearance of apoptotic neutrophils in systemic lupus erythematosus. Arthritis Rheum. 48, 2888–2897 (2003).
Donnelly, S. et al. Impaired recognition of apoptotic neutrophils by the C1q/calreticulin and CD91 pathway in systemic lupus erythematosus. Arthritis Rheum. 54, 1543–1556 (2006).
Budman, D. R. & Steinberg, A. D. Hematologic aspects of systemic lupus erythematosus. Current concepts. Ann. Intern. Med. 86, 220–229 (1977).
Kramers, C. et al. Anti-nucleosome antibodies complexed to nucleosomal antigens show anti-DNA reactivity and bind to rat glomerular basement membrane in vivo. J. Clin. Invest. 94, 568–577 (1994).
Arenas, M., Abad, A., Valverde, V., Ferriz, P. & Pascual, R. Selective inhibition of granulopoiesis with severe neutropenia in systemic lupus erythematosus. Arthritis Rheum. 35, 979–980 (1992).
Cairns, A. P., Crockard, A. D., McConnell, J. R., Courtney, P. A. & Bell, A. L. Reduced expression of CD44 on monocytes and neutrophils in systemic lupus erythematosus: relations with apoptotic neutrophils and disease activity. Ann. Rheum. Dis. 60, 950–955 (2001).
Hsieh, S. C. et al. Anti-SSB/La is one of the antineutrophil autoantibodies responsible for neutropenia and functional impairment of polymorphonuclear neutrophils in patients with systemic lupus erythematosus. Clin. Exp. Immunol. 131, 506–516, (2003).
Nassberger, L., Sjoholm, A. G., Jonsson, H., Sturfelt, G. & Akesson, A. Autoantibodies against neutrophil cytoplasm components in systemic lupus erythematosus and in hydralazine-induced lupus. Clin. Exp. Immunol. 81, 380–383 (1990).
Galeazzi, M. et al. Anti-neutrophil cytoplasmic antibodies in 566 European patients with systemic lupus erythematosus: prevalence, clinical associations and correlation with other autoantibodies. European Concerted Action on the Immunogenetics of SLE. Clin. Exp. Rheumatol. 16, 541–546 (1998).
Kurien, B. T., Newland, J., Paczkowski, C., Moore, K. L. & Scofield, R. H. Association of neutropenia in systemic lupus erythematosus (SLE) with anti-Ro and binding of an immunologically cross-reactive neutrophil membrane antigen. Clin. Exp. Immunol. 120, 209–217 (2000).
Hartman, K. R. et al. Antibodies to myeloid precursor cells in autoimmune neutropenia. Blood 84, 625–631 (1994).
Liu, H. et al. Suppression of haematopoiesis by IgG autoantibodies from patients with systemic lupus erythematosus (SLE). Clin. Exp. Immunol. 100, 480–485 (1995).
Hacbarth, E. & Kajdacsy-Balla, A. Low density neutrophils in patients with systemic lupus erythematosus, rheumatoid arthritis, and acute rheumatic fever. Arthritis Rheum. 29, 1334–1342 (1986).
Villanueva, E. et al. Netting neutrophils induce endothelial damage, infiltrate tissues, and expose immunostimulatory molecules in systemic lupus erythematosus. J. Immunol. 187, 538–552 (2011).
Nakou, M. et al. Gene expression in systemic lupus erythematosus: bone marrow analysis differentiates active from inactive disease and reveals apoptosis and granulopoiesis signatures. Arthritis Rheum. 58, 3541–3549 (2008).
Lin, A. M. et al. Mast cells and neutrophils release IL-17 through extracellular trap formation in psoriasis. J. Immunol. 187, 490–500 (2011).
Mohan, C., Adams, S., Stanik, V. & Datta, S. K. Nucleosome: a major immunogen for pathogenic autoantibody-inducing T cells of lupus. J. Exp. Med. 177, 1367–1381 (1993).
Bruns, A., Blass, S., Hausdorf, G., Burmester, G. R. & Hiepe, F. Nucleosomes are major T and B cell autoantigens in systemic lupus erythematosus. Arthritis Rheum. 43, 2307–2315 (2000).
Amoura, Z. et al. Nucleosome-restricted antibodies are detected before anti-dsDNA and/or antihistone antibodies in serum of MRL-Mp lpr/lpr and +/+ mice, and are present in kidney eluates of lupus mice with proteinuria. Arthritis Rheum. 37, 1684–1688 (1994).
Licht, R., van Bruggen, M. C., Oppers-Walgreen, B., Rijke, T. P. & Berden, J. H. Plasma levels of nucleosomes and nucleosome-autoantibody complexes in murine lupus: effects of disease progression and lipopolyssacharide administration. Arthritis Rheum. 44, 1320–1330 (2001).
McHugh, N. J. Systemic lupus erythematosus and dysregulated apoptosis—what is the evidence? Rheumatology (Oxford) 41, 242–245 (2002).
Fuchs, T. A. et al. Novel cell death program leads to neutrophil extracellular traps. J. Cell Biol. 176, 231–241 (2007).
Papayannopoulos, V., Metzler, K. D., Hakkim, A. & Zychlinsky, A. Neutrophil elastase and myeloperoxidase regulate the formation of neutrophil extracellular traps. J. Cell Biol. 191, 677–691 (2010).
Remijsen, Q. et al. Neutrophil extracellular trap cell death requires both autophagy and superoxide generation. Cell Res. 21, 290–304 (2011).
Buchanan, J. T. et al. DNase expression allows the pathogen group A Streptococcus to escape killing in neutrophil extracellular traps. Curr. Biol. 16, 396–400 (2006).
Neeli, I., Khan, S. N. & Radic, M. Histone deimination as a response to inflammatory stimuli in neutrophils. J. Immunol. 180, 1895–1902 (2008).
Kessenbrock, K. et al. Netting neutrophils in autoimmune small-vessel vasculitis. Nat. Med. 15, 623–625 (2009).
Fuchs, T. A. et al. Extracellular DNA traps promote thrombosis. Proc. Natl Acad. Sci. USA 107, 15880–15885 (2010).
Gupta, A. K. et al. Activated endothelial cells induce neutrophil extracellular traps and are susceptible to NETosis-mediated cell death. FEBS Lett. 584, 3193–3197 (2010).
Hakkim, A. et al. Impairment of neutrophil extracellular trap degradation is associated with lupus nephritis. Proc. Natl Acad. Sci. USA 107, 9813–9818 (2010).
Garcia-Romo, G. S. et al. Netting neutrophils are major inducers of type I IFN production in pediatric systemic lupus erythematosus. Sci. Transl. Med. 3, 73ra20 (2011).
Lande, R. et al. Neutrophils activate plasmacytoid dendritic cells by releasing self-DNA-peptide complexes in systemic lupus erythematosus. Sci. Transl. Med. 3, 73ra19 (2011).
Guiducci, C. et al. Autoimmune skin inflammation is dependent on plasmacytoid dendritic cell activation by nucleic acids via TLR7 and TLR9. J. Exp. Med. 207, 2931–2942 (2010).
Ward, M. M. Premature morbidity from cardiovascular and cerebrovascular diseases in women with systemic lupus erythematosus. Arthritis Rheum. 42, 338–346 (1999).
Rajagopalan, S. et al. Endothelial cell apoptosis in systemic lupus erythematosus: a common pathway for abnormal vascular function and thrombosis propensity. Blood 103, 3677–3683 (2004).
Lee, P. et al. Type I interferon as a novel risk factor for endothelial progenitor cell depletion and endothelial dysfunction in systemic lupus erythematosus. Arthritis Rheum. 56, 3759–3769 (2007).
Cochrane, C. G., Unanue, E. R. & Dixon, F. J. A role of polymorphonuclear leukocytes and complement in nephrotoxic nephritis. J. Exp. Med. 122, 99–116 (1965).
Hotta, O. et al. Role of neutrophil elastase in the development of renal necrotizing vasculitis. Clin. Nephrol. 45, 211–216 (1996).
Camussi, G. et al. The polymorphonuclear neutrophil (PMN) immunohistological technique: detection of immune complexes bound to the PMN membrane in acute poststreptococcal and lupus nephritis. Clin. Nephrol. 14, 280–287 (1980).
Johnson, R. J. et al. The human neutrophil serine proteinases, elastase and cathepsin G, can mediate glomerular injury in vivo. J. Exp. Med. 168, 1169–1174 (1988).
Scapini, P. et al. Proinflammatory mediators elicit secretion of the intracellular B-lymphocyte stimulator pool (BLyS) that is stored in activated neutrophils: implications for inflammatory diseases. Blood 105, 830–837 (2005).
Hinze, C. H. et al. Neutrophil gelatinase-associated lipocalin is a predictor of the course of global and renal childhood-onset systemic lupus erythematosus disease activity. Arthritis Rheum. 60, 2772–2781 (2009).
Bouali, H. et al. Association of the G-463A myeloperoxidase gene polymorphism with renal disease in African Americans with systemic lupus erythematosus. J. Rheumatol. 34, 2028–2034 (2007).
Rovin, B. H., Lu, L. & Zhang, X. A novel interleukin-8 polymorphism is associated with severe systemic lupus erythematosus nephritis. Kidney Int. 62, 261–265 (2002).
Obermoser, G., Sontheimer, R. D. & Zelger, B. Overview of common, rare and atypical manifestations of cutaneous lupus erythematosus and histopathological correlates. Lupus 19, 1050–1070 (2010).
Kieffer, C., Cribier, B. & Lipsker, D. Neutrophilic urticarial dermatosis: a variant of neutrophilic urticaria strongly associated with systemic disease. Report of 9 new cases and review of the literature. Medicine (Baltimore) 88, 23–31 (2009).
Gulati, A. et al. Palisaded neutrophilic granulomatous dermatitis associated with systemic lupus erythematosus presenting with the burning rope sign. J. Am. Acad. Dermatol. 61, 711–714 (2009).
Misago, N., Inoue, H., Inoue, T., Nagasawa, K. & Narisawa, Y. Palisaded neutrophilic granulomatous dermatitis in systemic lupus erythematosus with a butterfly rash-like lesion. Eur. J. Dermatol. 20, 128–129 (2010).
Hospach, T., von den Driesch, P. & Dannecker, G. E. Acute febrile neutrophilic dermatosis (Sweet's syndrome) in childhood and adolescence: two new patients and review of the literature on associated diseases. Eur. J. Pediatr. 168, 1–9 (2009).
Lipsker, D. & Saurat, J. H. Neutrophilic cutaneous lupus erythematosus. At the edge between innate and acquired immunity? Dermatology 216, 283–286 (2008).
Yang, J. et al. TH17 and natural TREG cell population dynamics in systemic lupus erythematosus. Arthritis Rheum. 60, 1472–1483 (2009).
Acknowledgements
The writing of this manuscript was supported by the NIH through Public Health Service Grant HL-088419.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The author declares no competing financial interests.
Rights and permissions
About this article
Cite this article
Kaplan, M. Neutrophils in the pathogenesis and manifestations of SLE. Nat Rev Rheumatol 7, 691–699 (2011). https://doi.org/10.1038/nrrheum.2011.132
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2011.132
This article is cited by
-
Proteomic analyses reveal cystatin c is a promising biomarker for evaluation of systemic lupus erythematosus
Clinical Proteomics (2023)
-
Oxidized galectin-1 in SLE fails to bind the inhibitory receptor VSTM1 and increases reactive oxygen species levels in neutrophils
Cellular & Molecular Immunology (2023)
-
The ratio of neutrophil to lymphocyte as a potential marker of clinicopathological activity for lupus nephritis
International Urology and Nephrology (2023)
-
The potential roles of type I interferon activated neutrophils and neutrophil extracellular traps (NETs) in the pathogenesis of primary Sjögren’s syndrome
Arthritis Research & Therapy (2022)
-
Identification of biomarkers related to neutrophils and two molecular subtypes of systemic lupus erythematosus
BMC Medical Genomics (2022)