Abstract
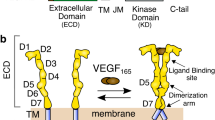
Receptor tyrosine kinases are activated upon ligand-induced dimerization. Here we show that the monomeric extracellular domain of vascular endothelial growth factor (VEGF) receptor-2 (VEGFR-2) has a flexible structure. Binding of VEGF to membrane-distal immunoglobulin-like domains causes receptor dimerization and promotes further interaction between receptor monomers through the membrane-proximal immunoglobulin-like domain 7. By this mechanism, ligand-induced dimerization of VEGFR-2 can be communicated across the membrane, activating the intracellular tyrosine kinase domains.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Hubbard, S.R. Nat. Rev. Mol. Cell Biol. 5, 464–471 (2004).
Schlessinger, J. Science 300, 750–752 (2003).
Olsson, A.K., Dimberg, A., Kreuger, J. & Claesson-Welsh, L. Nat. Rev. Mol. Cell Biol. 7, 359–371 (2006).
Christinger, H.W., Fuh, G., de Vos, A.M. & Wiesmann, C. J. Biol. Chem. 279, 10382–10388 (2003).
Wiesmann, C. et al. Cell 91, 695–704 (1997).
Muller, Y.A. et al. Proc. Natl. Acad. Sci. USA 94, 7192–7197 (1997).
Pieren, M. et al. J. Biol. Chem. 281, 19578–19587 (2006).
Shinkai, A. et al. J. Biol. Chem. 273, 31283–31288 (1998).
Fuh, G., Li, B., Crowley, C., Cunningham, B. & Wells, J.A. J. Biol. Chem. 273, 11197–11204 (1998).
Skiniotis, G., Boulanger, M.J., Garcia, K.C. & Walz, T. Nat. Struct. Mol. Biol. 12, 545–551 (2005).
Blechman, J.M. et al. Cell 80, 103–113 (1995).
Shulman, T., Sauer, F.G., Jackman, R.M., Chang, C.N. & Landolfi, N.F. J. Biol. Chem. 272, 17400–17404 (1997).
Omura, T., Heldin, C.H. & Ostman, A. J. Biol. Chem. 272, 12676–12682 (1997).
Barleon, B. et al. J. Biol. Chem. 272, 10382–10388 (1997).
Tao, Q., Backer, M.V., Backer, J.M. & Terman, B.I. J. Biol. Chem. 276, 21916–21923 (2001).
Acknowledgements
This work was supported by Swiss National Science Foundation grant 3100A0-100204 (to K.B.-H.) and US National Institutes of Health grant GM62580 (to T.W.). G.S. is a Damon Runyon Fellow, supported by the Damon Runyon Cancer Research Foundation (DRG no. 1824-04). The molecular EM facility at Harvard Medical School was established by a generous donation from the Giovanni Armenise Harvard Center for Structural Biology. We thank F. Winkler and A. Prota for critical reading of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Schematic representation of VEGFR-2 ECD constructs (PDF 14 kb)
Supplementary Fig. 2
Biochemical analysis of VEGFR-2 ECDs and VEGFR-2 ECD?ligand (PDF 57 kb)
Supplementary Fig. 3
Raw image of monomeric VEGFR-2 ECD in negative stain (PDF 5251 kb)
Supplementary Fig. 4
Raw image of predimerized VEGFR-2 ECD GCN4 in negative stain (PDF 4473 kb)
Supplementary Fig. 5
Raw image of negatively stained predimerized VEGFR-2 ECD GCN4 in complex with VEGF-A (PDF 4189 kb)
Supplementary Fig. 6
Raw image of negatively stained monomeric VEGFR-2 ECD in complex with VEGF-A (PDF 948 kb)
Supplementary Fig. 7
Class averages of predimerized VEGFR-2 ECD GCN4 in complex with VEGF-A (PDF 623 kb)
Supplementary Fig. 8
Class averages of monomeric VEGFR-2 ECD in complex with VEGF-A (PDF 4657 kb)
Supplementary Fig. 9
Raw image of negatively stained monomeric VEGFR-2 ECD in which immunoglobulin-like domain 7 was substituted by immunoglobulin-like domain 6 of VEGFR-1, in complex with VEGF-A (PDF 4437 kb)
Supplementary Fig. 10
Class averages of monomeric VEGFR-2 ECD in which immunoglobulin-like domain 7 was substituted by immunoglobulin-like domain 6 of VEGFR-1, in complex with VEGF-A (PDF 3462 kb)
Supplementary Table 1
Complex formation between VEGFR-2 and VEGF ligands (PDF 11 kb)
Rights and permissions
About this article
Cite this article
Ruch, C., Skiniotis, G., Steinmetz, M. et al. Structure of a VEGF–VEGF receptor complex determined by electron microscopy. Nat Struct Mol Biol 14, 249–250 (2007). https://doi.org/10.1038/nsmb1202
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb1202
This article is cited by
-
NRP1 interacts with endoglin and VEGFR2 to modulate VEGF signaling and endothelial cell sprouting
Communications Biology (2024)
-
Molecular basis of VEGFR1 autoinhibition at the plasma membrane
Nature Communications (2024)
-
Biomechanical stimulation promotes blood vessel growth despite VEGFR-2 inhibition
BMC Biology (2023)
-
Biology and therapeutic targeting of vascular endothelial growth factor A
Nature Reviews Molecular Cell Biology (2023)
-
Biochemical and Conformational Characterization of Recombinant VEGFR2 Domain 7
Molecular Biotechnology (2019)