Abstract
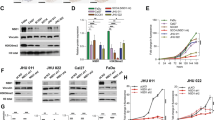
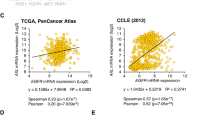
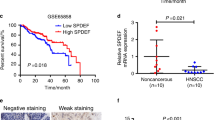
Overexpression of the Yes-associated protein (YAP), and TP53 family members ΔNp63 and p73, have been independently detected in subsets of head and neck squamous cell carcinomas (HNSCCs). YAP may serve as a nuclear cofactor with ΔNp63 and p73, but the functional role of YAP and their potential relationship in HNSCCs are unknown. In this study, we show that in a subset of HNSCC lines and tumors, YAP expression is increased but localized in the cytoplasm in association with increased AKT and YAP phosphorylation, and with decreased expression of ΔNp63 and p73. In another subset, YAP expression is decreased but detectable in the nucleus in association with lower AKT and YAP phosphorylation, and with increased ΔNp63 and p73 expression. Inhibiting AKT decreased serine-127 phosphorylation and enhanced nuclear translocation of YAP. ΔNp63 bound to the YAP promoter and suppressed its expression. Transfection of a YAP-serine-127-alanine phosphoacceptor-site mutant or ΔNp63 knockdown significantly increased nuclear YAP and cell death. Conversely, YAP knockdown enhanced cell proliferation, survival, migration and cisplatin chemoresistance. Thus, YAP function as a tumor suppressor may alternatively be dysregulated by AKT phosphorylation at serine-127 and cytoplasmic sequestration, or by transcriptional repression by ΔNp63, in different subsets of HNSCC. AKT and/or ΔNp63 are potential targets for enhancing YAP-mediated apoptosis and chemosensitivity in HNSCCs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Amornphimoltham P, Leelahavanichkul K, Molinolo A, Patel V, Gutkind JS . (2008). Inhibition of mammalian target of rapamycin by rapamycin causes the regression of carcinogen-induced skin tumor lesions. Clin Cancer Res 14: 8094–8101.
Amornphimoltham P, Sriuranpong V, Patel V, Benavides F, Conti CJ, Sauk J et al. (2004). Persistent activation of the Akt pathway in head and neck squamous cell carcinoma: a potential target for UCN-01. Clin Cancer Res 10: 4029–4037.
Baldwin C, Garnis C, Zhang L, Rosin MP, Lam WL . (2005). Multiple microalterations detected at high frequency in oral cancer. Cancer Res 65: 7561–7567.
Bancroft CC, Chen Z, Yeh J, Sunwoo JB, Yeh NT, Jackson S et al. (2002). Effects of pharmacologic antagonists of epidermal growth factor receptor, PI3K and MEK signal kinases on NF-kappaB and AP-1 activation and IL-8 and VEGF expression in human head and neck squamous cell carcinoma lines. Int J Cancer 99: 538–548.
Barbieri CE, Pietenpol JA . (2006). p63 and epithelial biology. Exp Cell Res 312: 695–706.
Barbieri CE, Tang LJ, Brown KA, Pietenpol JA . (2006). Loss of p63 leads to increased cell migration and up-regulation of genes involved in invasion and metastasis. Cancer Res 66: 7589–7597.
Basu S, Totty NF, Irwin MS, Sudol M, Downward J . (2003). Akt phosphorylates the Yes-associated protein, YAP, to induce interaction with 14-3-3 and attenuation of p73-mediated apoptosis. Mol Cell 11: 11–23.
Bertini E, Oka T, Sudol M, Strano S, Blandino G . (2009). YAP: at the crossroad between transformation and tumor suppression. Cell Cycle 8: 49–57.
Bian Y, Terse A, Du J, Hall B, Molinolo A, Zhang P et al. (2009). Progressive tumor formation in mice with conditional deletion of TGF-beta signaling in head and neck epithelia is associated with activation of the PI3K/Akt pathway. Cancer Res 69: 5918–5926.
Carey TE, Van Dyke DL, Worsham MJ . (1993). Nonrandom chromosome aberrations and clonal populations in head and neck cancer. Anticancer Res 13: 2561–2567.
Chen HI, Sudol M . (1995). The WW domain of Yes-associated protein binds a proline-rich ligand that differs from the consensus established for Src homology 3-binding modules. Proc Natl Acad Sci USA 92: 7819–7823.
Chen Z, Malhotra PS, Thomas GR, Ondrey FG, Duffey DC, Smith CW et al. (1999). Expression of proinflammatory and proangiogenic cytokines in patients with head and neck cancer. Clin Cancer Res 5: 1369–1379.
Danovi SA, Rossi M, Gudmundsdottir K, Yuan M, Melino G, Basu S . (2008). Yes-associated protein (YAP) is a critical mediator of c-Jun-dependent apoptosis. Cell Death Differ 15: 217–219.
Dong G, Chen Z, Li ZY, Yeh NT, Bancroft CC, Van Waes C . (2001a). Hepatocyte growth factor/scatter factor-induced activation of MEK and PI3K signal pathways contributes to expression of proangiogenic cytokines interleukin-8 and vascular endothelial growth factor in head and neck squamous cell carcinoma. Cancer Res 61: 5911–5918.
Dong G, Loukinova E, Chen Z, Gangi L, Chanturita TI, Liu ET et al. (2001b). Molecular profiling of transformed and metastatic murine squamous carcinoma cells by differential display and cDNA microarray reveals altered expression of multiple genes related to growth, apoptosis, angiogenesis, and the NF-kappaB signal pathway. Cancer Res 61: 4797–4808.
Dong G, Loukinova E, Smith CW, Chen Z, Van Waes C . (1997). Genes differentially expressed with malignant transformation and metastatic tumor progression of murine squamous cell carcinoma. J Cell Biochem Suppl 28–29: 90–100.
Downward J, Basu S . (2008). YAP and p73: a complex affair. Mol Cell 32: 749–750.
Forastiere A, Koch W, Trotti A, Sidransky D . (2001). Head and neck cancer. N Engl J Med 345: 1890–1900.
Friedman J, Nottingham L, Duggal P, Pernas FG, Yan B, Yang XP et al. (2007). Deficient TP53 expression, function, and cisplatin sensitivity are restored by quinacrine in head and neck cancer. Clin Cancer Res 13: 6568–6578.
Howell M, Borchers C, Milgram SL . (2004). Heterogeneous nuclear ribonuclear protein U associates with YAP and regulates its co-activation of Bax transcription. J Biol Chem 279: 26300–26306.
Komuro A, Nagai M, Navin NE, Sudol M . (2003). WW domain-containing protein YAP associates with ErbB-4 and acts as a co-transcriptional activator for the carboxyl-terminal fragment of ErbB-4 that translocates to the nucleus. J Biol Chem 278: 33334–33341.
Lee TL, Yang XP, Yan B, Friedman J, Duggal P, Bagain L et al. (2007). A novel nuclear factor-kappaB gene signature is differentially expressed in head and neck squamous cell carcinomas in association with TP53 status. Clin Cancer Res 13: 5680–5691.
Lee TL, Yeh J, Friedman J, Yan B, Yang X, Yeh NT et al. (2008). A signal network involving coactivated NF-kappaB and STAT3 and altered p53 modulates BAX/BCL-XL expression and promotes cell survival of head and neck squamous cell carcinomas. Int J Cancer 122: 1987–1998.
Lee TL, Yeh J, Van Waes C, Chen Z . (2006). Epigenetic modification of SOCS-1 differentially regulates STAT3 activation in response to interleukin-6 receptor and epidermal growth factor receptor signaling through JAK and/or MEK in head and neck squamous cell carcinomas. Mol Cancer Ther 5: 8–19.
Levy D, Adamovich Y, Reuven N, Shaul Y . (2007). The Yes-associated protein 1 stabilizes p73 by preventing Itch-mediated ubiquitination of p73. Cell Death Differ 14: 743–751.
Levy D, Adamovich Y, Reuven N, Shaul Y . (2008). Yap1 phosphorylation by c-Abl is a critical step in selective activation of proapoptotic genes in response to DNA damage. Mol Cell 29: 350–361.
Massarelli E, Liu DD, Lee JJ, El-Naggar AK, Lo Muzio L, Staibano S et al. (2005). Akt activation correlates with adverse outcome in tongue cancer. Cancer 104: 2430–2436.
Matallanas D, Romano D, Yee K, Meissl K, Kucerova L, Piazzolla D et al. (2007). RASSF1A elicits apoptosis through an MST2 pathway directing proapoptotic transcription by the p73 tumor suppressor protein. Mol Cell 27: 962–975.
Molinolo AA, Hewitt SM, Amornphimoltham P, Keelawat S, Rangdaeng S, Meneses Garcia A et al. (2007). Dissecting the Akt/mammalian target of rapamycin signaling network: emerging results from the head and neck cancer tissue array initiative. Clin Cancer Res 13: 4964–4973.
Moral M, Segrelles C, Lara MF, Martinez-Cruz AB, Lorz C, Santos M et al. (2009). Akt activation synergizes with Trp53 loss in oral epithelium to produce a novel mouse model for head and neck squamous cell carcinoma. Cancer Res 69: 1099–1108.
Oda K, Arakawa H, Tanaka T, Matsuda K, Tanikawa C, Mori T et al. (2000). p53AIP1, a potential mediator of p53-dependent apoptosis, and its regulation by ser-46-phosphorylated p53. Cell 102: 849–862.
Oka T, Mazack V, Sudol M . (2008). Mst2 and Lats kinases regulate apoptotic function of YAP. J Biol Chem 283: 27534–27546.
Overholtzer M, Zhang J, Smolen GA, Muir B, Li W, Sgroi DC et al. (2006). Transforming properties of YAP, a candidate oncogene on the chromosome 11q22 amplicon. Proc Natl Acad Sci USA 103: 12405–12410.
Rocco JW, Ellisen LW . (2006). p63 and p73: life and death in squamous cell carcinoma. Cell Cycle 5: 936–940.
Rocco JW, Leong CO, Kuperwasser N, DeYoung MP, Ellisen LW . (2006). p63 mediates survival in squamous cell carcinoma by suppression of p73-dependent apoptosis. Cancer Cell 9: 45–56.
Rosenbluth JM, Pietenpol JA . (2008). The jury is in: p73 is a tumor suppressor after all. Genes Dev 22: 2591–2595.
Snijders AM, Schmidt BL, Fridlyand J, Dekker N, Pinkel D, Jordan RC et al. (2005). Rare amplicons implicate frequent deregulation of cell fate specification pathways in oral squamous cell carcinoma. Oncogene 24: 4232–4242.
Steinhardt AA, Gayyed MF, Klein AP, Dong J, Maitra A, Pan D et al. (2008). Expression of Yes-associated protein in common solid tumors. Hum Pathol 39: 1582–1589.
Strano S, Monti O, Pediconi N, Baccarini A, Fontemaggi G, Lapi E et al. (2005). The transcriptional coactivator Yes-associated protein drives p73 gene-target specificity in response to DNA damage. Mol Cell 18: 447–459.
Strano S, Munarriz E, Rossi M, Castagnoli L, Shaul Y, Sacchi A et al. (2001). Physical interaction with Yes-associated protein enhances p73 transcriptional activity. J Biol Chem 276: 15164–15173.
Sudol M . (1994). Yes-associated protein (YAP65) is a proline-rich phosphoprotein that binds to the SH3 domain of the Yes proto-oncogene product. Oncogene 9: 2145–2152.
Sudol M, Bork P, Einbond A, Kastury K, Druck T, Negrini M et al. (1995). Characterization of the mammalian YAP (Yes-associated protein) gene and its role in defining a novel protein module, the WW domain. J Biol Chem 270: 14733–14741.
Thimmaiah KN, Easton JB, Germain GS, Morton CL, Kamath S, Buolamwini JK et al. (2005). Identification of N10-substituted phenoxazines as potent and specific inhibitors of Akt signaling. J Biol Chem 280: 31924–31935.
Thurfjell N, Coates PJ, Vojtesek B, Benham-Motlagh P, Eisold M, Nylander K . (2005). Endogenous p63 acts as a survival factor for tumour cells of SCCHN origin. Int J Mol Med 16: 1065–1070.
Westfall MD, Mays DJ, Sniezek JC, Pietenpol JA . (2003). The Delta Np63 alpha phosphoprotein binds the p21 and 14-3-3 sigma promoters in vivo and has transcriptional repressor activity that is reduced by Hay-Wells syndrome-derived mutations. Mol Cell Biol 23: 2264–2276.
Worden B, Yang XP, Lee TL, Bagain L, Yeh NT, Cohen JG et al. (2005). Hepatocyte growth factor/scatter factor differentially regulates expression of proangiogenic factors through Egr-1 in head and neck squamous cell carcinoma. Cancer Res 65: 7071–7080.
Yan B, Yang X, Lee TL, Friedman J, Tang J, Van Waes C et al. (2007). Genome-wide identification of novel expression signatures reveal distinct patterns and prevalence of binding motifs for p53, nuclear factor-kappaB and other signal transcription factors in head and neck squamous cell carcinoma. Genome Biol 8: R78.
Yu M, Yeh J, Van Waes C . (2006). Protein kinase casein kinase 2 mediates inhibitor-kappaB kinase and aberrant nuclear factor-kappaB activation by serum factor(s) in head and neck squamous carcinoma cells. Cancer Res 66: 6722–6731.
Yuan M, Tomlinson V, Lara R, Holliday D, Chelala C, Harada T et al. (2008). Yes-associated protein (YAP) functions as a tumor suppressor in breast. Cell Death Differ 15: 1752–1759.
Zender L, Spector MS, Xue W, Flemming P, Cordon-Cardo C, Silke J et al. (2006). Identification and validation of oncogenes in liver cancer using an integrative oncogenomic approach. Cell 125: 1253–1267.
Zhang J, Smolen GA, Haber DA . (2008). Negative regulation of YAP by LATS1 underscores evolutionary conservation of the Drosophila Hippo pathway. Cancer Res 68: 2789–2794.
Zhao B, Wei X, Li W, Udan RS, Yang Q, Kim J et al. (2007). Inactivation of YAP oncoprotein by the Hippo pathway is involved in cell contact inhibition and tissue growth control. Genes Dev 21: 2747–2761.
Acknowledgements
We thank Drs Tsutomu Oka, Maie St John and Ms Cindy Clark for their critical review; Ning Yeh, Dr Liesl Nottingham, Dr Jay Friedman, Dr Yansong Bian and Jonah Cohen for technical assistance; and Dr Paul Albert for assistance with statistical analysis. This study was supported by the NIDCD Intramural project Z1ADC-000073, Z1ADC-000074, Head and Neck Cancer Charity Grant and the Breast Cancer Coalition Grant from the Pennsylvania Department of Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Ehsanian, R., Brown, M., Lu, H. et al. YAP dysregulation by phosphorylation or ΔNp63-mediated gene repression promotes proliferation, survival and migration in head and neck cancer subsets. Oncogene 29, 6160–6171 (2010). https://doi.org/10.1038/onc.2010.339
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2010.339
Keywords
This article is cited by
-
The biology of YAP in programmed cell death
Biomarker Research (2022)
-
YAP1 acts as a negative regulator of pro-tumor TAZ expression in esophageal squamous cell carcinoma
Cellular Oncology (2022)
-
P4HA2-induced prolyl hydroxylation suppresses YAP1-mediated prostate cancer cell migration, invasion, and metastasis
Oncogene (2021)
-
Context-dependent roles of YAP/TAZ in stem cell fates and cancer
Cellular and Molecular Life Sciences (2021)
-
The potential role of YAP in head and neck squamous cell carcinoma
Experimental & Molecular Medicine (2020)