Abstract
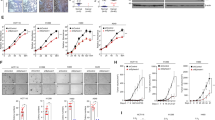
Mitogen-activated protein kinase (MAPK) and AKT pathways are frequently co-activated in melanoma through overexpression of receptor tyrosine kinases, mutations in their signaling surrogates, such as RAS and BRAF, or loss of negative regulators such as PTEN. As RAS can be a positive upstream regulator of PI3-K, it has been proposed that the loss of PTEN and the activation of RAS are redundant events in melanoma pathogenesis. Here, in genetically engineered mouse models of cutaneous melanomas, we sought to better understand the genetic interactions between HRAS activation and PTEN inactivation in melanoma genesis and progression in vivo. We showed that HRAS activation cooperates with Pten+/− and Ink4a/Arf−/− to increase melanoma penetrance and promote metastasis. Correspondingly, gain- and loss-of-function studies established that Pten loss increases invasion and migration of melanoma cells and non-transformed melanocytes, and such biological activity correlates with a shift to phosphorylation of AKT2 isoform and E-cadherin down-regulation. Thus, Pten inactivation can drive the genesis and promote the metastatic progression of RAS activated Ink4a/Arf deficient melanomas.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Ackermann J, Frutschi M, Kaloulis K, McKee T, Trumpp A, Beermann F . (2005). Metastasizing melanoma formation caused by expression of activated N-RasQ61K on an INK4a-deficient background. Cancer Res 65: 4005–4011.
Arboleda MJ, Lyons JF, Kabbinavar FF, Bray MR, Snow BE, Ayala R et al. (2003). Overexpression of AKT2/protein kinase Bbeta leads to up-regulation of beta1 integrins, increased invasion, and metastasis of human breast and ovarian cancer cells. Cancer Res 63: 196–206.
Bardeesy N, Bastian BC, Hezel A, Pinkel D, DePinho RA, Chin L . (2001). Dual inactivation of RB and p53 pathways in RAS-induced melanomas. Mol Cell Biol 21: 2144–2153.
Bardeesy N, Kim M, Xu J, Kim RS, Shen Q, Bosenberg MW et al. (2005). Role of epidermal growth factor receptor signaling in RAS-driven melanoma. Mol Cell Biol 25: 4176–4188.
Birck A, Ahrenkiel V, Zeuthen J, Hou-Jensen K, Guldberg P . (2000). Mutation and allelic loss of the PTEN/MMAC1 gene in primary and metastatic melanoma biopsies. J Invest Dermatol 114: 277–280.
Blanco-Aparicio C, Renner O, Leal JF, Carnero A . (2007). PTEN, more than the AKT pathway. Carcinogenesis 28: 1379–1386.
Brummelkamp TR, Bernards R, Agami R . (2002). A system for stable expression of short interfering RNAs in mammalian cells. Science 296: 550–553.
Chin L, Garraway LA, Fisher DE . (2006). Malignant melanoma: genetics and therapeutics in the genomic era. Genes Dev 20: 2149–2182.
Chin L, Pomerantz J, Polsky D, Jacobson M, Cohen C, Cordon-Cardo C et al. (1997). Cooperative effects of INK4a and ras in melanoma susceptibility in vivo. Genes Dev 11: 2822–2834.
Chin L, Tam A, Pomerantz J, Wong M, Holash J, Bardeesy N et al. (1999). Essential role for oncogenic Ras in tumour maintenance. Nature 400: 468–472.
Chudnovsky Y, Adams AE, Robbins PB, Lin Q, Khavari PA . (2005). Use of human tissue to assess the oncogenic activity of melanoma-associated mutations. Nat Genet 37: 745–749.
Cully M, You H, Levine AJ, Mak TW . (2006). Beyond PTEN mutations: the PI3K pathway as an integrator of multiple inputs during tumorigenesis. Nat Rev Cancer 6: 184–192.
Dankort D, Curley DP, Cartlidge RA, Nelson B, Karnezis AN, Damsky Jr WE et al. (2009). Braf(V600E) cooperates with Pten loss to induce metastatic melanoma. Nat Genet 41: 544–552.
Davies H, Bignell GR, Cox C, Stephens P, Edkins S, Clegg S et al. (2002). Mutations of the BRAF gene in human cancer. Nature 417: 949–954.
Davies MA, Stemke-Hale K, Tellez C, Calderone TL, Deng W, Prieto VG et al. (2008). A novel AKT3 mutation in melanoma tumours and cell lines. Br J Cancer 99: 1265–1268.
Dhawan P, Singh AB, Ellis DL, Richmond A . (2002). Constitutive activation of Akt/protein kinase B in melanoma leads to up-regulation of nuclear factor-kappaB and tumor progression. Cancer Res 62: 7335–7342.
Dhomen N, Reis-Filho JS, da Rocha Dias S, Hayward R, Savage K, Delmas V et al. (2009). Oncogenic Braf induces melanocyte senescence and melanoma in mice. Cancer Cell 15: 294–303.
Di Cristofano A, Pesce B, Cordon-Cardo C, Pandolfi PP . (1998). Pten is essential for embryonic development and tumour suppression. Nat Genet 19: 348–355.
Dinulescu DM, Ince TA, Quade BJ, Shafer SA, Crowley D, Jacks T . (2005). Role of K-ras and Pten in the development of mouse models of endometriosis and endometrioid ovarian cancer. Nat Med 11: 63–70.
Forbes SA, Bhamra G, Bamford S, Dawson E, Kok C, Clements J et al. (2008). The Catalogue of Somatic Mutations in Cancer (COSMIC). Curr Protoc Hum Genet 57: 10.11.1–10.11.26.
Goel VK, Ibrahim N, Jiang G, Singhal M, Fee S, Flotte T et al. (2009). Melanocytic nevus-like hyperplasia and melanoma in transgenic BRAFV600E mice. Oncogene 28: 2289–2298.
Gonzalez E, McGraw TE . (2009). The Akt kinases: isoform specificity in metabolism and cancer. Cell Cycle 8: 2502–2508.
Guldberg P, thor Straten P, Birck A, Ahrenkiel V, Kirkin AF, Zeuthen J . (1997). Disruption of the MMAC1/PTEN gene by deletion or mutation is a frequent event in malignant melanoma. Cancer Res 57: 3660–3663.
Gupta S, Ramjaun AR, Haiko P, Wang Y, Warne PH, Nicke B et al. (2007). Binding of ras to phosphoinositide 3-kinase p110alpha is required for ras-driven tumorigenesis in mice. Cell 129: 957–968.
Ikeda T, Yoshinaga K, Suzuki A, Sakurada A, Ohmori H, Horii A . (2000). Anticorresponding mutations of the KRAS and PTEN genes in human endometrial cancer. Oncol Rep 7: 567–570.
Iliopoulos D, Polytarchou C, Hatziapostolou M, Kottakis F, Maroulakou IG, Struhl K et al. (2009). MicroRNAs differentially regulated by Akt isoforms control EMT and stem cell renewal in cancer cells. Sci Signal 2: ra62.
Jafari M, Papp T, Kirchner S, Diener U, Henschler D, Burg G et al. (1995). Analysis of ras mutations in human melanocytic lesions: activation of the ras gene seems to be associated with the nodular type of human malignant melanoma. J Cancer Res Clin Oncol 121: 23–30.
Kim M, Gans JD, Nogueira C, Wang A, Paik JH, Feng B et al. (2006). Comparative oncogenomics identifies NEDD9 as a melanoma metastasis gene. Cell 125: 1269–1281.
Kotelevets L, van Hengel J, Bruyneel E, Mareel M, van Roy F, Chastre E . (2005). Implication of the MAGI-1b/PTEN signalosome in stabilization of adherens junctions and suppression of invasiveness. FASEB J 19: 115–117.
Lahtz C, Stranzenbach R, Fiedler E, Helmbold P, Dammann RH . (2010). Methylation of PTEN as a prognostic factor in malignant melanoma of the skin. J Invest Dermatol 130: 620–622.
Li J, Yen C, Liaw D, Podsypanina K, Bose S, Wang SI et al. (1997). PTEN, a putative protein tyrosine phosphatase gene mutated in human brain, breast, and prostate cancer. Science 275: 1943–1947.
Lin WM, Baker AC, Beroukhim R, Winckler W, Feng W, Marmion JM et al. (2008). Modeling genomic diversity and tumor dependency in malignant melanoma. Cancer Res 68: 664–673.
Liu H, Radisky DC, Nelson CM, Zhang H, Fata JE, Roth RA et al. (2006). Mechanism of Akt1 inhibition of breast cancer cell invasion reveals a protumorigenic role for TSC2. Proc Natl Acad Sci USA 103: 4134–4139.
Ma X, Ziel-van der Made AC, Autar B, van der Korput HA, Vermeij M, van Duijn P et al. (2005). Targeted biallelic inactivation of Pten in the mouse prostate leads to prostate cancer accompanied by increased epithelial cell proliferation but not by reduced apoptosis. Cancer Res 65: 5730–5739.
Majumder PK, Yeh JJ, George DJ, Febbo PG, Kum J, Xue Q et al. (2003). Prostate intraepithelial neoplasia induced by prostate restricted Akt activation: the MPAKT model. Proc Natl Acad Sci USA 100: 7841–7846.
Maroulakou IG, Oemler W, Naber SP, Tsichlis PN . (2007). Akt1 ablation inhibits, whereas Akt2 ablation accelerates, the development of mammary adenocarcinomas in mouse mammary tumor virus (MMTV)-ErbB2/neu and MMTV-polyoma middle T transgenic mice. Cancer Res 67: 167–177.
Mirmohammadsadegh A, Marini A, Nambiar S, Hassan M, Tannapfel A, Ruzicka T et al. (2006). Epigenetic silencing of the PTEN gene in melanoma. Cancer Res 66: 6546–6552.
Podsypanina K, Ellenson LH, Nemes A, Gu J, Tamura M, Yamada KM et al. (1999). Mutation of Pten/Mmac1 in mice causes neoplasia in multiple organ systems. Proc Natl Acad Sci USA 96: 1563–1568.
Pollock PM, Harper UL, Hansen KS, Yudt LM, Stark M, Robbins CM et al. (2003). High frequency of BRAF mutations in nevi. Nat Genet 33: 19–20.
Salmena L, Carracedo A, Pandolfi PP . (2008). Tenets of PTEN tumor suppression. Cell 133: 403–414.
Serrano M, Lee H, Chin L, Cordon-Cardo C, Beach D, DePinho RA . (1996). Role of the INK4a locus in tumor suppression and cell mortality. Cell 85: 27–37.
Sharpless NE, Kannan K, Xu J, Bosenberg MW, Chin L . (2003). Both products of the mouse Ink4a/Arf locus suppress melanoma formation in vivo. Oncogene 22: 5055–5059.
Stahl JM, Cheung M, Sharma A, Trivedi NR, Shanmugam S, Robertson GP . (2003). Loss of PTEN promotes tumor development in malignant melanoma. Cancer Res 63: 2881–2890.
Stahl JM, Sharma A, Cheung M, Zimmerman M, Cheng JQ, Bosenberg MW et al. (2004). Deregulated Akt3 activity promotes development of malignant melanoma. Cancer Res 64: 7002–7010.
Stambolic V, Tsao MS, Macpherson D, Suzuki A, Chapman WB, Mak TW . (2000). High incidence of breast and endometrial neoplasia resembling human Cowden syndrome in pten+/− mice. Cancer Res 60: 3605–3611.
Steck PA, Pershouse MA, Jasser SA, Yung WK, Lin H, Ligon AH et al. (1997). Identification of a candidate tumour suppressor gene, MMAC1, at chromosome 10q23.3 that is mutated in multiple advanced cancers. Nat Genet 15: 356–362.
Suzuki A, de la Pompa JL, Stambolic V, Elia AJ, Sasaki T, del Barco Barrantes I et al. (1998). High cancer susceptibility and embryonic lethality associated with mutation of the PTEN tumor suppressor gene in mice. Curr Biol 8: 1169–1178.
Sviderskaya EV, Bennett DC, Ho L, Bailin T, Lee ST, Spritz RA . (1997). Complementation of hypopigmentation in p-mutant (pink-eyed dilution) mouse melanocytes by normal human P cDNA, and defective complementation by OCA2 mutant sequences. J Invest Dermatol 108: 30–34.
Tamura M, Gu J, Danen EH, Takino T, Miyamoto S, Yamada KM . (1999). PTEN interactions with focal adhesion kinase and suppression of the extracellular matrix-dependent phosphatidylinositol 3-kinase/Akt cell survival pathway. J Biol Chem 274: 20693–20703.
Trotman LC, Niki M, Dotan ZA, Koutcher JA, Di Cristofano A, Xiao A et al. (2003). Pten dose dictates cancer progression in the prostate. PLoS Biol 1: E59.
Trotman LC, Wang X, Alimonti A, Chen Z, Teruya-Feldstein J, Yang H et al. (2007). Ubiquitination regulates PTEN nuclear import and tumor suppression. Cell 128: 141–156.
Tsao H, Goel V, Wu H, Yang G, Haluska FG . (2004). Genetic interaction between NRAS and BRAF mutations and PTEN/MMAC1 inactivation in melanoma. J Invest Dermatol 122: 337–341.
Tsao H, Zhang X, Benoit E, Haluska FG . (1998). Identification of PTEN/MMAC1 alterations in uncultured melanomas and melanoma cell lines. Oncogene 16: 3397–3402.
Tsao H, Zhang X, Fowlkes K, Haluska FG . (2000). Relative reciprocity of NRAS and PTEN/MMAC1 alterations in cutaneous melanoma cell lines. Cancer Res 60: 1800–1804.
Vogelmann R, Nguyen-Tat MD, Giehl K, Adler G, Wedlich D, Menke A . (2005). TGFbeta-induced downregulation of E-cadherin-based cell-cell adhesion depends on PI3-kinase and PTEN. J Cell Sci 118: 4901–4912.
Wang S, Gao J, Lei Q, Rozengurt N, Pritchard C, Jiao J et al. (2003). Prostate-specific deletion of the murine Pten tumor suppressor gene leads to metastatic prostate cancer. Cancer Cell 4: 209–221.
You MJ, Castrillon DH, Bastian BC, O'Hagan RC, Bosenberg MW, Parsons R et al. (2002). Genetic analysis of Pten and Ink4a/Arf interactions in the suppression of tumorigenesis in mice. Proc Natl Acad Sci USA 99: 1455–1460.
Yuan ZQ, Feldman RI, Sussman GE, Coppola D, Nicosia SV, Cheng JQ . (2003). AKT2 inhibition of cisplatin-induced JNK/p38 and Bax activation by phosphorylation of ASK1: implication of AKT2 in chemoresistance. J Biol Chem 278: 23432–23440.
Acknowledgements
C Nogueira was supported by a fellowship from FCT (PRAXIS/BD/21794/99). M Kim was supported by the Claudia Adams Barr Program, the Dermatology Foundation, and Melanoma Research Foundation. JH Dannenberg was supported by Damon-Runyon Cancer Research Foundation and the Dutch Cancer Society. This work was supported by grants from the NIH (UO1 CA84313; RO1 CA93947) to L Chin and from the Bankhead Coley Pilot Research Award and American Cancer Society Institutional Research Grant (#93-032-13) to M Kim. We thank Dr Jin Q Cheng (Moffitt Cancer Center) for helpful discussion and adenoviruses for PTEN and DN-AKT2 expression, and Dr Ronald A DePinho for critical reading of this paper.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Nogueira, C., Kim, KH., Sung, H. et al. Cooperative interactions of PTEN deficiency and RAS activation in melanoma metastasis. Oncogene 29, 6222–6232 (2010). https://doi.org/10.1038/onc.2010.349
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2010.349
Keywords
This article is cited by
-
Combination of chemotherapy with BRAF inhibitors results in effective eradication of malignant melanoma by preventing ATM-dependent DNA repair
Oncogene (2021)
-
WNT/β-catenin signaling regulates mitochondrial activity to alter the oncogenic potential of melanoma in a PTEN-dependent manner
Oncogene (2017)
-
Directed migration of cancer cells guided by the graded texture of the underlying matrix
Nature Materials (2016)
-
Activating PIK3CA mutations coexist with BRAF or NRAS mutations in a limited fraction of melanomas
Journal of Translational Medicine (2015)
-
RUNX2 and the PI3K/AKT axis reciprocal activation as a driving force for tumor progression
Molecular Cancer (2015)