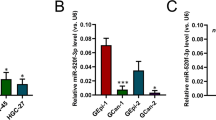
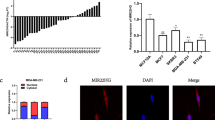
Abstract
Dysregulated microRNAs (miRNAs) have an important role in many malignant tumors. However, elucidating the roles of miRNAs in cancer biology, especially in epithelial cancers, remains an ongoing process. In this study, we show that both miR-143 and miR-145, which belong to the same miRNA cluster, can negatively modulate expression of their target gene, MDM2. The miR-143 and miR-145 is posttranscriptionally activated by upregulated p53, thereby generating a short miRNAs-MDM2-p53 feedback loop. Re-expression of these miRNAs suppresses cellular growth and triggers the apoptosis of epithelial cancer, in vitro and in vivo, by enhancing p53 activity via MDM2 turnover. Moreover, the miRNA-dependent MDM2 turnover contributes to the equilibrium of repeated p53 pulses in response to DNA damage stress. These findings suggest that MDM2 dysregulation caused by downregulation of miR-143 and miR-145 contributes to epithelial cancer development and has a key role in regulating cellular proliferation and apoptosis. Re-expression of miR-143 and miR-145 may be a reasonable strategy for treatment of epithelial cancers.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Fan CY . Epigenetic alterations in head and neck cancer: prevalence, clinical significance, and implications. Curr Oncol Rep 2004; 6: 152–161.
Hanahan D, Weinberg RA . Hallmarks of cancer: the next generation. Cell 2011; 144: 646–674.
Leemans CR, Braakhuis BJ, Brakenhoff RH . The molecular biology of head and neck cancer. Nat Rev Cancer 2011; 11: 9–22.
Vassilev LT . Small-molecule antagonists of p53-MDM2 binding: research tools and potential therapeutics. Cell Cycle 2004; 3: 419–421.
Itahana K, Mao H, Jin A, Itahana Y, Clegg HV, Lindstrom MS et al. Targeted inactivation of Mdm2 RING finger E3 ubiquitin ligase activity in the mouse reveals mechanistic insights into p53 regulation. Cancer Cell 2007; 12: 355–366.
Macias E, Jin A, Deisenroth C, Bhat K, Mao H, Lindstrom MS et al. An ARF-independent c-MYC-activated tumor suppression pathway mediated by ribosomal protein-Mdm2 Interaction. Cancer Cell 2010; 18: 231–243.
Saha MN, Jiang H, Jayakar J, Reece D, Branch DR, Chang H . MDM2 antagonist nutlin plus proteasome inhibitor velcade combination displays a synergistic anti-myeloma activity. Cancer Biol Ther 2010; 9: 936–944.
Stommel JM, Wahl GM . Accelerated MDM2 auto-degradation induced by DNA-damage kinases is required for p53 activation. EMBO J 2004; 23: 1547–1556.
Inuzuka H, Tseng A, Gao D, Zhai B, Zhang Q, Shaik S et al. Phosphorylation by casein kinase I promotes the turnover of the Mdm2 oncoprotein via the SCF(beta-TRCP) ubiquitin ligase. Cancer Cell 2010; 18: 147–159.
Pichiorri F, Suh SS, Rocci A, De Luca L, Taccioli C, Santhanam R et al. Downregulation of p53-inducible microRNAs 192, 194, and 215 impairs the p53/MDM2 autoregulatory loop in multiple myeloma development. Cancer Cell 2010; 18: 367–381.
Brown BD, Naldini L . Exploiting and antagonizing microRNA regulation for therapeutic and experimental applications. Nat Rev Genet 2009; 10: 578–585.
Calin GA, Sevignani C, Dumitru CD, Hyslop T, Noch E, Yendamuri S et al. Human microRNA genes are frequently located at fragile sites and genomic regions involved in cancers. Proc Natl Acad Sci USA 2004; 101: 2999–3004.
Arva NC, Talbott KE, Okoro DR, Brekman A, Qiu WG, Bargonetti J . Disruption of the p53-Mdm2 complex by Nutlin-3 reveals different cancer cell phenotypes. Ethn Dis 2008; 18 (2 Suppl 2): S2-1–8.
Suzuki HI, Yamagata K, Sugimoto K, Iwamoto T, Kato S, Miyazono K . Modulation of microRNA processing by p53. Nature 2009; 460: 529–533.
Liu G, Lozano G . p21 stability: linking chaperones to a cell cycle checkpoint. Cancer Cell 2005; 7: 113–114.
Soriano ME, Scorrano L . Traveling Bax and forth from mitochondria to control apoptosis. Cell 2011; 145: 15–17.
Bouska A, Lushnikova T, Plaza S, Eischen CM . Mdm2 promotes genetic instability and transformation independent of p53. Mol Cell Biol 2008; 28: 4862–4874.
Manfredi JJ . The Mdm2-p53 relationship evolves: Mdm2 swings both ways as an oncogene and a tumor suppressor. Genes Dev 2010; 24: 1580–1589.
Ishimura A, Terashima M, Kimura H, Akagi K, Suzuki Y, Sugano S et al. Jmjd2c histone demethylase enhances the expression of Mdm2 oncogene. Biochem Biophys Res Commun 2009; 389: 366–371.
Ries S, Biederer C, Woods D, Shifman O, Shirasawa S, Sasazuki T et al. Opposing effects of Ras on p53: transcriptional activation of mdm2 and induction of p19ARF. Cell 2000; 103: 321–330.
Chen L, Marechal V, Moreau J, Levine AJ, Chen J . Proteolytic cleavage of the mdm2 oncoprotein during apoptosis. J Biol Chem 1997; 272: 22966–22973.
Asslaber D, Pinon JD, Seyfried I, Desch P, Stocher M, Tinhofer I et al. microRNA-34a expression correlates with MDM2 SNP309 polymorphism and treatment-free survival in chronic lymphocytic leukemia. Blood 2010; 115: 4191–4197.
Dai Y, Xie CH, Neis JP, Fan CY, Vural E, Spring PM . MicroRNA expression profiles of head and neck squamous cell carcinoma with docetaxel-induced multidrug resistance. Head Neck 2011; 33: 786–791.
Langevin SM, Stone RA, Bunker CH, Lyons-Weiler MA, Laframboise WA, Kelly L et al. MicroRNA-137 promoter methylation is associated with poorer overall survival in patients with squamous cell carcinoma of the head and neck. Cancer 2011; 117: 1454–1462.
Boucher JM, Peterson SM, Urs S, Zhang C, Liaw L . The miR-143/145 cluster is a novel transcriptional target of jagged-1/notch signaling in vascular smooth muscle cells. J Biol Chem 2011; 286: 28312–28321.
Kent OA, Chivukula RR, Mullendore M, Wentzel EA, Feldmann G, Lee KH et al. Repression of the miR-143/145 cluster by oncogenic Ras initiates a tumor-promoting feed-forward pathway. Genes Dev 2010; 24: 2754–2759.
Kitade Y, Akao Y . MicroRNAs and their therapeutic potential for human diseases: microRNAs, miR-143 and -145, function as anti-oncomirs and the application of chemically modified miR-143 as an anti-cancer drug. J Pharmacol Sci 2010; 114: 276–280.
Akao Y, Nakagawa Y, Hirata I, Iio A, Itoh T, Kojima K et al. Role of anti-oncomirs miR-143 and -145 in human colorectal tumors. Cancer Gene Ther 2010; 17: 398–408.
Chiyomaru T, Enokida H, Tatarano S, Kawahara K, Uchida Y, Nishiyama K et al. miR-145 and miR-133a function as tumour suppressors and directly regulate FSCN1 expression in bladder cancer. Br J Cancer 2010; 102: 883–891.
Sachdeva M, Mo YY . MicroRNA-145 suppresses cell invasion and metastasis by directly targeting mucin 1. Cancer Res 2010; 70: 378–387.
Chen LH, Tsai KL, Chen YW, Yu CC, Chang KW, Chiou SH et al. MicroRNA as a novel modulator in head and neck squamous carcinoma. J Oncol 2010; 2010: 135632.
Chen X, Guo X, Zhang H, Xiang Y, Chen J, Yin Y et al. Role of miR-143 targeting KRAS in colorectal tumorigenesis. Oncogene 2009; 28: 1385–1392.
Zhang J, Guo H, Zhang H, Wang H, Qian G, Fan X et al. Putative tumor suppressor miR-145 inhibits colon cancer cell growth by targeting oncogene Friend leukemia virus integration 1 gene. Cancer 2011; 117: 86–95.
Sachdeva M, Zhu S, Wu F, Wu H, Walia V, Kumar S et al. p53 represses c-Myc through induction of the tumor suppressor miR-145. Proc Natl Acad Sci USA 2009; 106: 3207–3212.
Zhang J, Guo H, Qian G, Ge S, Ji H, Hu X et al. MiR-145, a new regulator of the DNA fragmentation factor-45 (DFF45)-mediated apoptotic network. Mol Cancer 2010; 9: 211.
Phelps M, Darley M, Primrose JN, Blaydes JP . p53-independent activation of the hdm2-P2 promoter through multiple transcription factor response elements results in elevated hdm2 expression in estrogen receptor alpha-positive breast cancer cells. Cancer Res 2003; 63: 2616–2623.
Araki S, Eitel JA, Batuello CN, Bijangi-Vishehsaraei K, Xie XJ, Danielpour D et al. TGF-beta1-induced expression of human Mdm2 correlates with late-stage metastatic breast cancer. J Clin Invest 2010; 120: 290–302.
Acknowledgements
This study was supported by the National Natural Science Foundation of China (Grant No. 30973343 and 81101515), Projects of the Shanghai Science and Technology Committee (Grant No. 08JC1414400, 11DZ2291800, 10DZ1951300 and 10XD1402500), the Shanghai Leading Academic Discipline Project (S30206), the Program of Shanghai Municipal Education Commission (12YZ052 and shjdy027) and National Basic Research Program of China (2010CB529204).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Zhang, J., Sun, Q., Zhang, Z. et al. Loss of microRNA-143/145 disturbs cellular growth and apoptosis of human epithelial cancers by impairing the MDM2-p53 feedback loop. Oncogene 32, 61–69 (2013). https://doi.org/10.1038/onc.2012.28
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2012.28
Keywords
This article is cited by
-
Expression Profile of Circulating Exosomal microRNAs in Leukoplakia, Oral Submucous Fibrosis, and Combined Lesions of Leukoplakia and Oral Submucous Fibrosis
Head and Neck Pathology (2024)
-
Profiling miRNAs in tear extracellular vesicles: a pilot study with implications for diagnosis of ocular diseases
Japanese Journal of Ophthalmology (2024)
-
Signaling pathways in cancer metabolism: mechanisms and therapeutic targets
Signal Transduction and Targeted Therapy (2023)
-
MicroRNA-143 Sensitizes Cervical Cancer Cells to Cisplatin: a Promising Anticancer Combination Therapy
Reproductive Sciences (2021)
-
microRNAs as therapeutic targets in intestinal diseases
ExRNA (2019)