Abstract
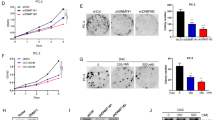
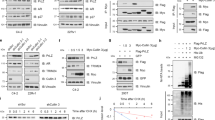
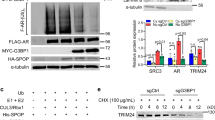
EZH2 is crucial for the progression of prostate cancer (PCa) and castration-resistant prostate cancer (CRPC) through upregulation and activation of progenitor genes, as well as androgen receptor (AR)-target genes. However, the mechanisms by which EZH2 is regulated in PCa and CRPC remain elusive. Here we report that EZH2 is post-transcriptionally regulated by SKP2 in vitro in cultured cells and in vivo in mouse models. We observed aberrant upregulation of Skp2, Ezh2 and histone H3 lysine 27 trimethylation (H3K27me3) in both Pten null mouse embryonic fibroblasts (MEFs) and Pten null mouse prostate tissues. Loss of Skp2 resulted in a striking decrease of Ezh2 levels in Pten/Trp53 double-null MEFs and in prostate tumors of Pten/Trp53 double-null mutant mice. SKP2 knockdown decreased EZH2 levels in human PCa cells through upregulation of TRAF6-mediated and lysine(K) 63-linked ubiquitination of EZH2 for degradation. Ectopic expression of TRAF6 promoted the K63-linked ubiquitination of EZH2 to decrease EZH2 and H3K27me3 levels in PCa cells. In contrast, TRAF6 knockdown resulted in a reduced EZH2 ubiquitination with an increase of EZH2 and H3K27me3 levels in PCa cells. Furthermore, the catalytically dead mutant TRAF6 C70A abolished the TRAF6-mediated polyubiquitination of recombinant human EZH2 in vitro. Most importantly, a concurrent elevation of Skp2 and Ezh2 was found in CRPC tumors of Pten/Trp53 mutant mice, and expression levels of SKP2 and EZH2 were positively correlated in human PCa specimens. Taken together, our findings revealed a novel mechanism on EZH2 ubiquitination and an important signaling network of SKP2-TRAF6-EZH2/H3K27me3, and targeting SKP2-EZH2 pathway may be a promising therapeutic strategy for CRPC treatment.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Chi P, Allis CD, Wang GG . Covalent histone modifications–miswritten, misinterpreted and mis-erased in human cancers. Nat Rev Cancer 2010; 10: 457–469.
Lu W, Liu S, Li B, Xie Y, Adhiambo C, Yang Q et al. SKP2 inactivation suppresses prostate tumorigenesis by mediating JARID1B ubiquitination. Oncotarget 2015; 6: 771–788.
Carrano AC, Eytan E, Hershko A, Pagano M . SKP2 is required for ubiquitin-mediated degradation of the CDK inhibitor p27. Nat Cell Biol 1999; 1: 193–199.
Nakayama KI, Nakayama K . Ubiquitin ligases: cell-cycle control and cancer. Nat Rev Cancer 2006; 6: 369–381.
Lin HK, Chen Z, Wang G, Nardella C, Lee SW, Chan CH et al. Skp2 targeting suppresses tumorigenesis by Arf-p53-independent cellular senescence. Nature 2010; 464: 374–379.
Lin HK, Wang G, Chen Z, Teruya-Feldstein J, Liu Y, Chan CH et al. Phosphorylation-dependent regulation of cytosolic localization and oncogenic function of Skp2 by Akt/PKB. Nat Cell Biol 2009; 11: 420–432.
Chan CH, Lee SW, Li CF, Wang J, Yang WL, Wu CY et al. Deciphering the transcriptional complex critical for RhoA gene expression and cancer metastasis. Nat Cell Biol 2010; 12: 457–467.
Yang G, Ayala G, De Marzo A, Tian W, Frolov A, Wheeler TM et al. Elevated Skp2 protein expression in human prostate cancer: association with loss of the cyclin-dependent kinase inhibitor p27 and PTEN and with reduced recurrence-free survival. Clin Cancer Res 2002; 8: 3419–3426.
Gao D, Wan L, Inuzuka H, Berg AH, Tseng A, Zhai B et al. Rictor forms a complex with Cullin-1 to promote SGK1 ubiquitination and destruction. Mol Cell 2010; 39: 797–808.
Li B, Lu W, Yang Q, Yu X, Matusik RJ, Chen Z . Skp2 regulates androgen receptor through ubiquitin-mediated degradation independent of Akt/mTOR pathways in prostate cancer. Prostate 2014; 74: 421–432.
Wang Z, Gao D, Fukushima H, Inuzuka H, Liu P, Wan L et al. Skp2: a novel potential therapeutic target for prostate cancer. Biochim Biophys Acta 2012; 1825: 11–17.
Kim SY, Herbst A, Tworkowski KA, Salghetti SE, Tansey WP . Skp2 regulates Myc protein stability and activity. Mol Cell 2003; 11: 1177–1188.
von der Lehr N, Johansson S, Wu S, Bahram F, Castell A, Cetinkaya C et al. The F-box protein Skp2 participates in c-Myc proteosomal degradation and acts as a cofactor for c-Myc-regulated transcription. Mol Cell 2003; 11: 1189–1200.
Ngollo M, Lebert A, Dagdemir A, Judes G, Karsli-Ceppioglu S, Daures M et al. The association between histone 3 lysine 27 trimethylation (H3K27me3) and prostate cancer: relationship with clinicopathological parameters. BMC Cancer 2014; 14: 994.
Cao R, Wang L, Wang H, Xia L, Erdjument-Bromage H, Tempst P et al. Role of histone H3 lysine 27 methylation in Polycomb-group silencing. Science 2002; 298: 1039–1043.
Yu J, Cao Q, Mehra R, Laxman B, Tomlins SA, Creighton CJ et al. Integrative genomics analysis reveals silencing of beta-adrenergic signaling by polycomb in prostate cancer. Cancer Cell 2007; 12: 419–431.
Chen H, Tu SW, Hsieh JT . Down-regulation of human DAB2IP gene expression mediated by polycomb Ezh2 complex and histone deacetylase in prostate cancer. J Biol Chem 2005; 280: 22437–22444.
Min J, Zaslavsky A, Fedele G, McLaughlin SK, Reczek EE, De Raedt T et al. An oncogene-tumor suppressor cascade drives metastatic prostate cancer by coordinately activating Ras and nuclear factor-kappaB. Nat Med 2010; 16: 286–294.
Varambally S, Dhanasekaran SM, Zhou M, Barrette TR, Kumar-Sinha C, Sanda MG et al. The polycomb group protein EZH2 is involved in progression of prostate cancer. Nature 2002; 419: 624–629.
Xu K, Wu ZJ, Groner AC, He HH, Cai C, Lis RT et al. EZH2 oncogenic activity in castration-resistant prostate cancer cells is Polycomb-independent. Science 2012; 338: 1465–1469.
Yamaguchi H, Hung MC . Regulation and Role of EZH2 in Cancer. Cancer Res Treat 2014; 46: 209–222.
Yu YL, Chou RH, Shyu WC, Hsieh SC, Wu CS, Chiang SY et al. Smurf2-mediated degradation of EZH2 enhances neuron differentiation and improves functional recovery after ischaemic stroke. EMBO Mol Med 2013; 5: 531–547.
Sahasrabuddhe AA, Chen X, Chung F, Velusamy T, Lim MS, Elenitoba-Johnson KS . Oncogenic Y641 mutations in EZH2 prevent Jak2/beta-TrCP-mediated degradation. Oncogene 2015; 34: 445–454.
Zoabi M, Sadeh R, de Bie P, Marquez VE, Ciechanover A . PRAJA1 is a ubiquitin ligase for the polycomb repressive complex 2 proteins. Biochem Biophys Res Commun 2011; 408: 393–398.
Shen Z, Chen L, Yang X, Zhao Y, Pier E, Zhang X et al. Downregulation of Ezh2 methyltransferase by FOXP3: new insight of FOXP3 into chromatin remodeling? Biochim Biophys Acta 2013; 1833: 2190–2200.
Mamillapalli R, Gavrilova N, Mihaylova VT, Tsvetkov LM, Wu H, Zhang H et al. PTEN regulates the ubiquitin-dependent degradation of the CDK inhibitor p27(KIP1) through the ubiquitin E3 ligase SCF(SKP2). Curr Biol 2001; 11: 263–267.
Gao D, Inuzuka H, Tseng A, Chin RY, Toker A, Wei W . Phosphorylation by Akt1 promotes cytoplasmic localization of Skp2 and impairs APCCdh1-mediated Skp2 destruction. Nat Cell Biol 2009; 11: 397–408.
Ke XS, Qu Y, Rostad K, Li WC, Lin B, Halvorsen OJ et al. Genome-wide profiling of histone h3 lysine 4 and lysine 27 trimethylation reveals an epigenetic signature in prostate carcinogenesis. PLoS One 2009; 4: e4687.
Chen Z, Trotman LC, Shaffer D, Lin HK, Dotan ZA, Niki M et al. Crucial role of p53-dependent cellular senescence in suppression of Pten-deficient tumorigenesis. Nature 2005; 436: 725–730.
Chan CH, Morrow JK, Li CF, Gao Y, Jin G, Moten A et al. Pharmacological inactivation of Skp2 SCF ubiquitin ligase restricts cancer stem cell traits and cancer progression. Cell 2013; 154: 556–568.
Yang WL, Wang J, Chan CH, Lee SW, Campos AD, Lamothe B et al. The E3 ligase TRAF6 regulates Akt ubiquitination and activation. Science 2009; 325: 1134–1138.
Ellinger J, Kahl P, von der Gathen J, Heukamp LC, Gutgemann I, Walter B et al. Global histone H3K27 methylation levels are different in localized and metastatic prostate cancer. Cancer Invest 2012; 30: 92–97.
Valdes-Mora F, Clark SJ . Prostate cancer epigenetic biomarkers: next-generation technologies. Oncogene 2015; 34: 1609–1618.
Xiang Y, Zhu Z, Han G, Ye X, Xu B, Peng Z et al. JARID1B is a histone H3 lysine 4 demethylase up-regulated in prostate cancer. Proc Natl Acad Sci USA 2007; 104: 19226–19231.
Cha TL, Zhou BP, Xia W, Wu Y, Yang CC, Chen CT et al. Akt-mediated phosphorylation of EZH2 suppresses methylation of lysine 27 in histone H3. Science 2005; 310: 306–310.
Chu CS, Lo PW, Yeh YH, Hsu PH, Peng SH, Teng YC et al. O-GlcNAcylation regulates EZH2 protein stability and function. Proc Natl Acad Sci USA 2014; 111: 1355–1360.
Yang WL, Zhang X, Lin HK . Emerging role of Lys-63 ubiquitination in protein kinase and phosphatase activation and cancer development. Oncogene 2010; 29: 4493–4503.
Huang F, Zeng X, Kim W, Balasubramani M, Fortian A, Gygi SP et al. Lysine 63-linked polyubiquitination is required for EGF receptor degradation. Proc Natl Acad Sci USA 2013; 110: 15722–15727.
Zhang L, Xu M, Scotti E, Chen ZJ, Tontonoz P . Both K63 and K48 ubiquitin linkages signal lysosomal degradation of the LDL receptor. J Lipid Res 2013; 54: 1410–1420.
Duncan LM, Piper S, Dodd RB, Saville MK, Sanderson CM, Luzio JP et al. Lysine-63-linked ubiquitination is required for endolysosomal degradation of class I molecules. EMBO J 2006; 25: 1635–1645.
Sugeno N, Hasegawa T, Tanaka N, Fukuda M, Wakabayashi K, Oshima R et al. Lys-63-linked ubiquitination by E3 ubiquitin ligase Nedd4-1 facilitates endosomal sequestration of internalized alpha-synuclein. J Biol Chem 2014; 289: 18137–18151.
Ferreira JV, Soares AR, Ramalho JS, Pereira P, Girao H . K63 linked ubiquitin chain formation is a signal for HIF1A degradation by chaperone-mediated autophagy. Sci Rep 2015; 5: 10210.
Dice JF . Peptide sequences that target cytosolic proteins for lysosomal proteolysis. Trends Biochem Sci 1990; 15: 305–309.
Lin TY, Cheng YC, Yang HC, Lin WC, Wang CC, Lai PL et al. Loss of the candidate tumor suppressor BTG3 triggers acute cellular senescence via the ERK-JMJD3-p16(INK4a) signaling axis. Oncogene 2012; 31: 3287–3297.
Lu W, Xie Y, Ma Y, Matusik RJ, Chen Z . ARF represses androgen receptor transactivation in prostate cancer. Mol Endocrinol 2013; 27: 635–648.
Sudo T, Utsunomiya T, Mimori K, Nagahara H, Ogawa K, Inoue H et al. Clinicopathological significance of EZH2 mRNA expression in patients with hepatocellular carcinoma. Br J Cancer 2005; 92: 1754–1758.
Bhatt KV, Hu R, Spofford LS, Aplin AE . Mutant B-RAF signaling and cyclin D1 regulate Cks1/S-phase kinase-associated protein 2-mediated degradation of p27Kip1 in human melanoma cells. Oncogene 2007; 26: 1056–1066.
Jung HY, Jun S, Lee M, Kim HC, Wang X, Ji H et al. PAF and EZH2 induce Wnt/beta-catenin signaling hyperactivation. Mol Cell 2013; 52: 193–205.
Shembade N, Ma A, Harhaj EW . Inhibition of NF-kappaB signaling by A20 through disruption of ubiquitin enzyme complexes. Science 2010; 327: 1135–1139.
Cocka LJ, Bates P . Identification of alternatively translated Tetherin isoforms with differing antiviral and signaling activities. PLoS Pathog 2012; 8: e1002931.
Jiao S, Zhang Z, Li C, Huang M, Shi Z, Wang Y et al. The kinase MST4 limits inflammatory responses through direct phosphorylation of the adaptor TRAF6. Nat Immunol 2015; 16: 246–257.
Qi J, Tripathi M, Mishra R, Sahgal N, Fazli L, Ettinger S et al. The E3 ubiquitin ligase Siah2 contributes to castration-resistant prostate cancer by regulation of androgen receptor transcriptional activity. Cancer Cell 2013; 23: 332–346.
Wertz IE, O'Rourke KM, Zhou H, Eby M, Aravind L, Seshagiri S et al. De-ubiquitination and ubiquitin ligase domains of A20 downregulate NF-kappaB signalling. Nature 2004; 430: 694–699.
Bracken AP, Pasini D, Capra M, Prosperini E, Colli E, Helin K . EZH2 is downstream of the pRB-E2F pathway, essential for proliferation and amplified in cancer. EMBO J 2003; 22: 5323–5335.
Acknowledgements
We would like to thank Dr Jae-Il Park for Flag-EZH2 plasmid, Dr Paul Bates and Dr Zhaocai (Joe) Zhou for Myc-TRAF6 and Myc-TRAF6-C70A plasmids, Dr Edward W. Harhaj for ubiquitin and ubiquitin mutant plasmids, and Dr Hui-Kuan Lin for GST, GST-TRAF6 and GST-TRAF6-C70A plasmids. HA-tagged EZH2 is a gift from Kristian Helin (Addgene plasmid # 24230).54 This work was supported in part by NIH grants MD004038, DK055748, CA163069, MD007593 and UL1TR000445-06. Human tissue staining, microscopy experiments and data analysis were performed through the Human Tissue & Pathology Core and Morphology Core of Meharry Medical College, supported in part by NIH grants MD007593, MD007586, DA036420 and S10RR0254970.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Lu, W., Liu, S., Li, B. et al. SKP2 loss destabilizes EZH2 by promoting TRAF6-mediated ubiquitination to suppress prostate cancer. Oncogene 36, 1364–1373 (2017). https://doi.org/10.1038/onc.2016.300
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2016.300
This article is cited by
-
Role of K63-linked ubiquitination in cancer
Cell Death Discovery (2022)
-
TRIM25 regulates oxaliplatin resistance in colorectal cancer by promoting EZH2 stability
Cell Death & Disease (2021)
-
Post-translational modifications of EZH2 in cancer
Cell & Bioscience (2020)
-
EZH2: a novel target for cancer treatment
Journal of Hematology & Oncology (2020)
-
Ubiquitin ligases HUWE1 and NEDD4 cooperatively control signal-dependent PRC2-Ezh1α/β-mediated adaptive stress response pathway in skeletal muscle cells
Epigenetics & Chromatin (2019)