Abstract
Tissue macrophages (Mϕ) are essential effector cells in rheumatoid arthritis (RA), contributing to autoimmune tissue inflammation through diverse effector functions. Their arthritogenic potential depends on their proficiency to survive in the glucose-depleted environment of the inflamed joint. Here, we identify a mechanism that links metabolic adaptation to nutrient stress with the efficacy of tissue Mϕ to activate adaptive immunity by presenting antigen to tissue-invading T cells. Specifically, Mϕ populating the rheumatoid joint produce and respond to the small cytokine CCL18, which protects against cell death induced by glucose withdrawal. Mechanistically, CCL18 induces the transcription factor RFX5 that selectively upregulates glutamate dehydrogenase 1 (GLUD1), thus enabling glutamate utilization to support energy production. In parallel, RFX5 enhances surface expression of HLA-DR molecules, promoting Mϕ-dependent expansion of antigen-specific T cells. These data place CCL18 at the top of a RFX5–GLUD1 survival pathway and couple adaptability to nutrient conditions in the tissue environment to antigen-presenting function in autoimmune tissue inflammation.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
All data that support the findings of this study are available from the corresponding author upon reasonable request. Source data are provided with this paper.
References
Weyand, C. M. & Goronzy, J. J. The immunology of rheumatoid arthritis. Nat. Immunol. 22, 10–18 (2021).
Dinarello, C. A., Simon, A. & van der Meer, J. W. Treating inflammation by blocking interleukin-1 in a broad spectrum of diseases. Nat. Rev. Drug Discov. 11, 633–652 (2012).
Sanin, D. E. et al. A common framework of monocyte-derived macrophage activation. Sci. Immunol. 7, eabl7482 (2022).
Barrat, F. J., Crow, M. K. & Ivashkiv, L. B. Interferon target-gene expression and epigenomic signatures in health and disease. Nat. Immunol. 20, 1574–1583 (2019).
Chakarov, S. et al. Two distinct interstitial macrophage populations coexist across tissues in specific subtissular niches. Science 363, eaau0964 (2019).
Molawi, K. & Sieweke, M. H. Monocytes compensate Kupffer cell loss during bacterial infection. Immunity 42, 10–12 (2015).
Udalova, I. A., Mantovani, A. & Feldmann, M. Macrophage heterogeneity in the context of rheumatoid arthritis. Nat. Rev. Rheumatol. 12, 472–485 (2016).
Smiljanovic, B. et al. Monocyte alterations in rheumatoid arthritis are dominated by preterm release from bone marrow and prominent triggering in the joint. Ann. Rheum. Dis. 77, 300–308 (2018).
Zhang, F. et al. Defining inflammatory cell states in rheumatoid arthritis joint synovial tissues by integrating single-cell transcriptomics and mass cytometry. Nat. Immunol. 20, 928–942 (2019).
Culemann, S. et al. Locally renewing resident synovial macrophages provide a protective barrier for the joint. Nature 572, 670–675 (2019).
Kuo, D. et al. HBEGF+ macrophages in rheumatoid arthritis induce fibroblast invasiveness. Sci. Transl. Med. 11, eaau8587 (2019).
Jager, E. et al. Calcium-sensing receptor-mediated NLRP3 inflammasome response to calciprotein particles drives inflammation in rheumatoid arthritis. Nat. Commun. 11, 4243 (2020).
Boutet, M. A. et al. Novel insights into macrophage diversity in rheumatoid arthritis synovium. Autoimmun. Rev. 20, 102758 (2021).
Dance, A. Core concept: cells nibble one another via the under-appreciated process of trogocytosis. Proc. Natl Acad. Sci. USA 116, 17608–17610 (2019).
Li, G. et al. T cell antigen discovery via trogocytosis. Nat. Methods 16, 183–190 (2019).
Orange, D. E. et al. Identification of three rheumatoid arthritis disease subtypes by machine learning integration of synovial histologic features and RNA sequencing data. Arthritis Rheumatol. 70, 690–701 (2018).
Wu, B. et al. Mitochondrial aspartate regulates TNF biogenesis and autoimmune tissue inflammation. Nat. Immunol. 22, 1551–1562 (2021).
Wu, X. et al. Single-cell sequencing of immune cells from anticitrullinated peptide antibody positive and negative rheumatoid arthritis. Nat. Commun. 12, 4977 (2021).
He, T. et al. Bidirectional membrane molecule transfer between dendritic and T cells. Biochem. Biophys. Res. Commun. 359, 202–208 (2007).
Shen, Y. et al. Metabolic control of the scaffold protein TKS5 in tissue-invasive, proinflammatory T cells. Nat. Immunol. 18, 1025–1034 (2017).
Zhang, S., Carriere, J., Lin, X., Xie, N. & Feng, P. Interplay between cellular metabolism and cytokine responses during viral infection. Viruses 10, 521 (2018).
Shi, J., Fan, J., Su, Q. & Yang, Z. Cytokines and abnormal glucose and lipid metabolism. Front Endocrinol. 10, 703 (2019).
Woetzel, D. et al. Identification of rheumatoid arthritis and osteoarthritis patients by transcriptome-based rule set generation. Arthritis Res. Ther. 16, R84 (2014).
Schraufstatter, I. U., Zhao, M., Khaldoyanidi, S. K. & Discipio, R. G. The chemokine CCL18 causes maturation of cultured monocytes to macrophages in the M2 spectrum. Immunology 135, 287–298 (2012).
van Lieshout, A. W. et al. Novel insights in the regulation of CCL18 secretion by monocytes and dendritic cells via cytokines, toll-like receptors and rheumatoid synovial fluid. BMC Immunol. 7, 23 (2006).
Shultz, L. D., Brehm, M. A., Garcia-Martinez, J. V. & Greiner, D. L. Humanized mice for immune system investigation: progress, promise and challenges. Nat. Rev. Immunol. 12, 786–798 (2012).
Walsh, N. C. et al. Humanized mouse models of clinical disease. Annu Rev. Pathol. 12, 187–215 (2017).
Chang, N. H., Inman, R. D., Dick, J. E. & Wither, J. E. Bone marrow-derived human hematopoietic stem cells engraft NOD/SCID mice and traffic appropriately to an inflammatory stimulus in the joint. J. Rheumatol. 37, 496–502 (2010).
Jin, L. et al. Glutamate dehydrogenase 1 signals through antioxidant glutathione peroxidase 1 to regulate redox homeostasis and tumor growth. Cancer Cell 27, 257–270 (2015).
Landt, S. G. et al. ChIP–seq guidelines and practices of the ENCODE and modENCODE consortia. Genome Res. 22, 1813–1831 (2012).
Novakovic, B. et al. β-glucan reverses the epigenetic state of LPS-induced immunological tolerance. Cell 167, 1354–1368(2016).
Wong, D. et al. Genomic mapping of the MHC transactivator CIITA using an integrated ChIP–seq and genetical genomics approach. Genome Biol. 15, 494 (2014).
Couture, A. et al. HLA-class II artificial antigen presenting cells in CD4+ T cell-based immunotherapy. Front. Immunol. 10, 1081 (2019).
Davis, M. M., Altman, J. D. & Newell, E. W. Interrogating the repertoire: broadening the scope of peptide–MHC multimer analysis. Nat. Rev. Immunol. 11, 551–558 (2011).
Huang, J. et al. Detection, phenotyping, and quantification of antigen-specific T cells using a peptide–MHC dodecamer. Proc. Natl Acad. Sci. USA 113, E1890–E1897 (2016).
Morou, A. et al. Altered differentiation is central to HIV-specific CD4+ T cell dysfunction in progressive disease. Nat. Immunol. 20, 1059–1070 (2019).
Gautier, E. L. & Yvan-Charvet, L. Understanding macrophage diversity at the ontogenic and transcriptomic levels. Immunol. Rev. 262, 85–95 (2014).
Shirai, T. et al. The glycolytic enzyme PKM2 bridges metabolic and inflammatory dysfunction in coronary artery disease. J. Exp. Med. 213, 337–354 (2016).
Zeisbrich, M. et al. Hypermetabolic macrophages in rheumatoid arthritis and coronary artery disease due to glycogen synthase kinase 3b inactivation. Ann. Rheum. Dis. 77, 1053–1062 (2018).
Gautier, E. L. et al. Gene-expression profiles and transcriptional regulatory pathways that underlie the identity and diversity of mouse tissue macrophages. Nat. Immunol. 13, 1118–1128 (2012).
Weyand, C. M. & Goronzy, J. J. Pathogenesis of rheumatoid arthritis. Med. Clin. North Am. 81, 29–55 (1997).
Bettadapur, A., Miller, H. W. & Ralston, K. S. Biting off what can be chewed: trogocytosis in health, infection, and disease. Infect. Immun. 88, e00930-19 (2020).
Dopfer, E. P., Minguet, S. & Schamel, W. W. A new vampire saga: the molecular mechanism of T cell trogocytosis. Immunity 35, 151–153 (2011).
Snyder, J. P. & Amiel, E. Regulation of dendritic cell immune function and metabolism by cellular nutrient sensor mammalian target of rapamycin (mTOR). Front. Immunol. 9, 3145 (2018).
Roche, P. A. & Furuta, K. The ins and outs of MHC class II-mediated antigen processing and presentation. Nat. Rev. Immunol. 15, 203–216 (2015).
Greten, F. R. & Grivennikov, S. I. Inflammation and cancer: triggers, mechanisms, and consequences. Immunity 51, 27–41 (2019).
Riera-Domingo, C. et al. Immunity, hypoxia, and metabolism—the menage a trois of cancer: implications for immunotherapy. Physiol. Rev. 100, 1–102 (2020).
DeBerardinis, R. J. et al. Beyond aerobic glycolysis: transformed cells can engage in glutamine metabolism that exceeds the requirement for protein and nucleotide synthesis. Proc. Natl Acad. Sci. USA 104, 19345–19350 (2007).
Garcia-Canaveras, J. C., Chen, L. & Rabinowitz, J. D. The tumor metabolic microenvironment: lessons from lactate. Cancer Res. 79, 3155–3162 (2019).
Mergenthaler, P., Lindauer, U., Dienel, G. A. & Meisel, A. Sugar for the brain: the role of glucose in physiological and pathological brain function. Trends Neurosci. 36, 587–597 (2013).
Ferretti, A. C., Larocca, M. C. & Favre, C. Nutritional stress in eukaryotic cells: oxidative species and regulation of survival in time of scarceness. Mol. Genet. Metab. 105, 186–192 (2012).
Jha, A. K. et al. Network integration of parallel metabolic and transcriptional data reveals metabolic modules that regulate macrophage polarization. Immunity 42, 419–430 (2015).
Shang, M. et al. Macrophage-derived glutamine boosts satellite cells and muscle regeneration. Nature 587, 626–631 (2020).
Meiser, J. et al. Pro-inflammatory macrophages sustain pyruvate oxidation through pyruvate dehydrogenase for the synthesis of itaconate and to enable cytokine expression. J. Biol. Chem. 291, 3932–3946 (2016).
Tannahill, G. M. et al. Succinate is an inflammatory signal that induces IL-1beta through HIF-1α. Nature 496, 238–242 (2013).
Mills, E. L. et al. Itaconate is an anti-inflammatory metabolite that activates Nrf2 via alkylation of KEAP1. Nature 556, 113–117 (2018).
Mills, E. L. et al. Succinate dehydrogenase supports metabolic repurposing of mitochondria to drive inflammatory macrophages. Cell 167, 457–470 e413 (2016).
O’Neill, L. A. J. & Artyomov, M. N. Itaconate: the poster child of metabolic reprogramming in macrophage function. Nat. Rev. Immunol. 19, 273–281 (2019).
Wang, F. et al. Glycolytic stimulation is not a requirement for M2 macrophage differentiation. Cell Metab. 28, 463–475(2018).
Watanabe, R. et al. Pyruvate controls the checkpoint inhibitor PD-L1 and suppresses T cell immunity. J. Clin. Invest. 127, 2725–2738 (2017).
Huang, Y. et al. Microglia/macrophage-derived human CCL18 promotes glioma progression via CCR8–ACP5 axis analyzed in humanized slice model. Cell Rep. 39, 110670 (2022).
Kraaijeveld, A. O. et al. CC chemokine ligand-5 (CCL5/RANTES) and CC chemokine ligand-18 (CCL18/PARC) are specific markers of refractory unstable angina pectoris and are transiently raised during severe ischemic symptoms. Circulation 116, 1931–1941 (2007).
Chen, P. et al. Identification and validation of four hub genes involved in the plaque deterioration of atherosclerosis. Aging 11, 6469–6489 (2019).
Wu, B. et al. Succinyl-CoA ligase deficiency in pro-inflammatory and tissue-invasive T cells. Cell Metab. 32, 967–980(2020).
Li, Y. et al. The DNA repair nuclease MRE11A functions as a mitochondrial protector and prevents T cell pyroptosis and tissue inflammation. Cell Metab. 30, 477–492(2019).
Pucino, V. et al. Lactate buildup at the site of chronic inflammation promotes disease by inducing CD4+ T. Cell Metab. Rewiring. Cell Metab. 30, 1055–1074(2019).
Jin, K. et al. NOTCH-induced rerouting of endosomal trafficking disables regulatory T cells in vasculitis. J. Clin. Invest. 131, e136042 (2021).
Acknowledgements
This work was supported by the National Institutes of Health (R01AR042527, R01AI108906, R01HL142068, and P01HL129941 to CMW and R01AI108891, R01AG045779, U19AI057266, R01AI129191 to J.J.G.) and by the Encrantz Family Discovery Fund.
Author information
Authors and Affiliations
Contributions
Conceptualization, C.M.W., J.J.G.; Formal Analysis, Z.H., T.V.Z.; Investigation, Z.H., T.V.Z., T.H., S.O., K.J., I.N.G., B.W., G.J.B.; Clinical Samples, M.P.A., J.W.B.; Writing (original), C.M.W., Z.H., T.V.Z., I.N.G.; Supervision, C.M.W., J.J.G., G.J.B.; Funding Acquisition, C.M.W., J.J.G.
Corresponding author
Ethics declarations
Competing interests
The authors have no competing interests.
Peer review
Peer review information
Nature Metabolism thanks Anne Davidson, Ping-Chih Ho and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Editor recognition statement Primary handling editors: Alfredo Gimenez-Cassina and George Caputa, in collaboration with the Nature Metabolism team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
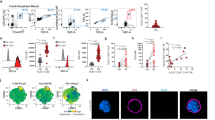
Extended Data Fig. 1 Characterization of synovial Mϕ.
a,b. Reanalysis of the original data from Nat Commun. 2021 Aug 17;12(1):4977 “Single-cell sequencing of immune cells from anticitrullinated peptide antibody positive and negative rheumatoid arthritis” by Wu et al. a, tSNE visualization of pooled scRNA-seq data from 71,073 CD45 + cells isolated the synovial tissue of rheumatoid arthritis patients (n = 20). We identified 21 clusters, including T cells (4 clusters), NK cells (1 cluster), B cells (3 clusters), fibroblasts (2 clusters), pericyte (1 cluster), endothelial cells (1 cluster), mast cells (1 cluster), dendritic cells (2 clusters), and macrophages (6 clusters). b, Gene transcript expression related to antigen presentation in different cell clusters. Data are shown as violin blots, and the 6 macrophage subsets are marked by a red frame.
Extended Data Fig. 2 HLA-DR and trogocytosis of synovial Mϕ.
a, HLA-DR protein expression was measured by flow cytometry in CD206 + CD163 + cells and CD206-CD163low cells isolated from low- and high-grade synovitis (n = 5). b, Identification of trogocytic Mϕ in the synovial tissue. Single cell suspensions were prepared from synovial tissues collected from RA patients. Multiparametric flow cytometry identified CD68 + Mϕ, which had integrated T cell-derived membrane fragments. Captured membrane fragments stained positive with two independent anti-CD3 antibodies (UCHT clone (clone 2) and SK7 clone (clone 1)). CD68 + CD3/UCHTI + CD3/SK7 + cells were considered trogocytic macrophages. Gating strategy and a representative dot plot overlaying CD68 + cells from a tissue with low-grade and high-grade synovitis.
Extended Data Fig. 3 Dynamics of Mϕ ingress into human synovium engrafted into NSG mice.
a–c, Human synovial tissues were implanted into NSG mice subcutaneously. Mϕ were induced from monocytes with M-CSF. After 7 days, CFSE-labelled Mϕ were mixed with autologous PBMC (1:10) and adoptively transferred into the mice. Synovial tissue grafts were harvested and digested on days 3, 5, 7 and 14 and tissue-residing macrophages were analyzed in the single cell suspensions by flow cytometry. a, Gating strategy for CFSE-labeled CD68 + cells in the tissue. b,c, Representative dot blots and frequencies of CFSE + CD68 + Mϕ were measured in the peripheral blood (n = 4), the spleen (n = 4) and the synovial tissue (n = 8) on days 3, 5, 7 and 14 after adoptive transfer. Data presented as violin blots.
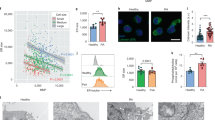
Extended Data Fig. 4 Glutamate and a-KG sustain survival and mitochondrial respiration in monocyte-derived Mϕ.
a,b, Oxygen consumption rates (OCR) of monocyte-derived Mϕ generated from RA patients and healthy controls were compared by Seahorse analysis (n = 4). Basal OCR, ATP-coupled OCR and maximum respiration were measured by seahorse experiment on day 3. c,d, Mϕ from healthy controls and RA patients were kept in glucose and glutamine-free medium. Parallel cultures were supplemented with increasing doses of glutamine (0–2 mM) or glutamate (0–1 mM). Cell survival was quantified by LDH release assays on day 7 (n = 4). e,f, RA patient derived Mϕ were cultured under glucose depleted conditions and supplemented with the amino acids glutamine or glutamate or α-KG (0.5 mM). e, Metabolic activity of these Mϕ was determined by Seahorse analysis (n = 6). (f) ATP-coupled OCR was measured by seahorse experiment on day 3 (n = 4). Data are mean ± SEM. b–d, Two-tailed unpaired t test. f, One-way ANOVA with post hoc Tukey’s multiple comparisons test.
Extended Data Fig. 5 Expression of CCL18 in synovial cell populations and ex vivo induction of CCL18 transcripts.
a, Tissue samples from low-grade and high-grade RA synovitis were digested and single cell suspensions were treated with ionomycin/PMA and LPS. After 6 hours, CCL18 protein was measured via flow cytometry. Histograms comparing the mean fluorescence intensity (MFI) for CCL18 in fibroblasts, T cells and B cells. FMO shown in light grey. b, Monocyte-derived macrophages were induced ex vivo with M-CSF over 6 days and then stimulated with different stimuli as indicated. M0 cells remained unstimulated. CCL18 transcripts were quantified by RT-PCR. Data are mean ± SEM from 4 experiments.
Extended Data Fig. 6 CCL18 and the CCL18 target GLUD1 regulate survival of tissue-infiltrating Mϕ.
CFSE-labeled Mϕ were adoptively transferred into NSG mice that had been engrafted with human synovium. Synovial grafts were harvested after 7 days and tissue-infiltrating Mϕ were analyzed by flow cytometry. a, Chimeric mice were treated with rhCCL18 (50 μg/mouse, n = 9) or vehicle control (n = 10) for 7 days. Grafts were explanted and percentages of CFSE + Mϕ within the CD3neg cell population were measured, Data are presented as violin plots. b, Before the adoptive transfer, Mϕ were transfected with a control (n = 10), GLUD1-expressing vector (n = 9). Synovial grafts were harvested and processed for flow cytometric analysis. Percentages of CFSE + Mϕ within the CD3neg cell population were measured. Data are presented as violin plots. c, Chimeras were reconstituted with CFSE-labeled Mϕ originating from RA patients and treated with the GLUD1 inhibitor R162 (10 mg/kg, n = 12) or vehicle control (n = 12). Frequencies of CFSE + Mϕ within the CD3neg cell population were measured. Data are presented as violin plots. d–f, Proliferation indices of Mϕ isolated from the explanted human synovial tissue were determined based on CSFE dilution. Data are in box and whisker plots.
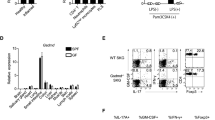
Extended Data Fig. 7 CCL18-induced upregulation of transcription factors.
MDM from healthy individuals and RA patients were induced with M-CSF over 6 days and differentiated with LPS/IFN-γ. Color scales are presented by fold change. a, Gene expression profiling for 20 transcription factors in Mϕ treated with rhCCL18 (50 ng/mL, n = 5) or vehicle (n = 5) for 48 h. Transcripts were quantified by RT-PCR. b, Gene expression of 20 transcription factors in Mϕ generated from controls and RA patients. mRNA expression was measured by RT-PCR.
Extended Data Fig. 8 Knock down efficiency for CCL18, GLUD1 and RFX5.
MDM were generated from RA patients. a,b, CCL18 was knocked down by transfecting the Mϕ with CCL18 siRNA. CCL18 protein and transcripts were quantified by FACS (a, n = 6) and qPCR (b, n = 5). c,d, To knock down GLUD1, Mϕ were transfected with GLUD1 siRNA. GLUD1 expression was quantified by immunoblot (c, n = 3) and qPCR (d, n = 4). e,f, RFX5 was knocked down by transfecting Mϕ with RFX5 siRNA. RFX5 protein and transcripts were quantified by immunoblotting (e, n = 3) and qPCR (f, n = 4). Data are mean ± SEM from 3–5 experiments in each group. Two-tailed paired t test.
Extended Data Fig. 9 Gating strategy to detect antigen-reactive CD4+ T cells.
PBMC were stimulated with Candida albicans antigen (0.05 Units/mL) for 6 days. Antigen-reactive T cells were detected by flow cytometry. a–c, Cells of interest were identified by forward versus side scatter (FSC vs SSC) based on size and granularity (a). Alive cells were identified as 7-AADnegative (b). CD4 + T cells were identified as CD3 + CD4 + cells (c). d, Antigen-reactive CD3 + CD4 + T cells were identified as CD69 + CD40L +. Frequencies in FMO, in non-stimulated cells and in antigen-stimulated cells. e, T cells were primed with antigen as in a–d and restimulated with Mϕ that were loaded with vehicle or antigen. CD3 + CD4 + CD69 + CD40L + T cells were measured after 6 hours.
Supplementary information
Supplementary Tables
Supplementary Tables 1–7
Source data
Source Data Fig. 1
Statistical Source Data
Source Data Fig. 2
Statistical Source Data
Source Data Fig. 3
Statistical Source Data
Source Data Fig. 4
Statistical Source Data
Source Data Fig. 5
Unprocessed western blots for Fig. 5d and 5e
Source Data Fig. 5
Statistical Source Data
Source Data Fig. 6
Unprocessed western blots for Fig. 6a and 6b
Source Data Fig. 6
Statistical Source Data
Source Data Fig. 7
Statistical Source Data
Source Data Extended Data Fig. 3
Statistical Source Data
Source Data Extended Data Fig. 4
Statistical Source Data
Source Data Extended Data Fig. 5
Statistical Source Data
Source Data Extended Data Fig. 6
Statistical Source Data
Source Data Extended Data Fig. 7
Statistical Source Data
Source Data Extended Data Fig. 8
Statistical Source Data
Source Data Extended Data Fig. 8
Unprocessed western blots for Extended data Fig. 8c and 8e
Rights and permissions
About this article
Cite this article
Hu, Z., Zhao, T.V., Huang, T. et al. The transcription factor RFX5 coordinates antigen-presenting function and resistance to nutrient stress in synovial macrophages. Nat Metab 4, 759–774 (2022). https://doi.org/10.1038/s42255-022-00585-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s42255-022-00585-x
This article is cited by
-
Mitochondrial and metabolic dysfunction of peripheral immune cells in multiple sclerosis
Journal of Neuroinflammation (2024)
-
Investigation of the stent induced deformation on hemodynamic of internal carotid aneurysms by computational fluid dynamics
Scientific Reports (2023)
-
Computational study of blood flow inside MCA aneurysm with/without endovascular coiling
Scientific Reports (2023)
-
Single-cell multi-omics analysis of human testicular germ cell tumor reveals its molecular features and microenvironment
Nature Communications (2023)
-
Reduction of rupture risk in ICA aneurysms by endovascular techniques of coiling and stent: numerical study
Scientific Reports (2023)