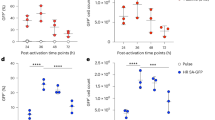
Abstract
We have designed a transgene that encodes a scFv(G250) chimeric receptor, which is specific for carboxyanhydrase IX (G250-ligand, G250L), a molecule overexpressed by renal cell cancer (RCC). Retroviral transduction of this transgene into primary human T lymphocytes confers these cells with specific functional responses towards G250L-positive RCC cells. In preparation of a clinical phase (I/II) study in RCC patients, we set up a protocol for gene transduction and expansion of primary human T cells. For this purpose, we directly compared two packaging cell lines, that is, the GALV-pseudotyped MLV producing cell line PG13, and the MLV-A-producing cell line Phi-NX-Ampho (a.k.a. Phoenix-A). We generated and characterized stable scFv(G250)-positive clones of both PG13 and Phoenix cells and optimized the retrovirus production conditions. Transductions of primary human T cells yielded 30–60% scFv(G250)+ T cells using PG13-derived retrovirus versus up to 90% scFv(G250)+ T cells using Phoenix-derived retrovirus. The median number of transgene integrations per scFv(G250)+ T cell differed only 1.5-fold as determined by real-time PCR (mean number of integrations per T cell 2.6 and 3.7 for PG13 and Phoenix-based transductions, respectively). In addition, T cells transduced with Phoenix-derived retrovirus showed, on a per cell basis, 10–30% higher levels of scFv(G250)-mediated TNFα production and cytolysis of G250L+ RCC cells than T cells transduced with PG13-derived retrovirus. The improved functional transduction efficiency together with a limited increase in the number of integrations per recipient cell, made us select Phoenix clone 58 for our clinical immunogene therapy study.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Lamers CH, Willemsen RA, Luider BA, Debets R, Bolhuis RL . Protocol for gene transduction and expansion of human T lymphocytes for clinical immunogene therapy of cancer. Cancer Gene Ther 2002; 9: 613–623.
Weijtens ME, Willemsen RA, Hart EH, Bolhuis RL . A retroviral vector system ‘STITCH’ in combination with an optimized single chain antibody chimeric receptor gene structure allows efficient gene transduction and expression in human T lymphocytes. Gene Therapy 1998; 5: 1195–1203.
Weijtens ME, Hart EH, Bolhuis RL . Functional balance between T cell chimeric receptor density and tumor associated antigen density: CTL mediated cytolysis and lymphokine production. Gene Therapy 2000; 7: 35–42.
Le Doux JM, Morgan JR, Yarmush ML . Differential inhibition of retrovirus transduction by proteoglycans and free glycosaminoglycans. Biotechnol Prog 1999; 15: 397–406.
Swift S, Lorens J, Achacoso P, Nolan GP . Radid production of retroviruses for efficient gene delivery to mammalian cells using 293T cell-basd systems. Current Protocols in Immunology 1999; 10.17: 14–29.
Kustikova OS, Wahlers A, Kuhlcke K, Stahle B, Zander AR, Baum C et al. Dose finding with retroviral vectors: correlation of retroviral vector copy numbers in single cells with gene transfer efficiency in a cell population. Blood 2003; 102: 3934–3937.
Schaft N, Willemsen RA, de Vries J, Lankiewicz B, Essers BW, Gratama JW et al. Peptide fine specificity of anti-glycoprotein 100 CTL is preserved following transfer of engineered TCR alpha beta genes into primary human T lymphocytes. J Immunol 2003; 170: 2186–2194.
Baum C, Von Kalle C, Staal FJ, Li Z, Fehse B, Schmidt M et al. Chance or necessity? Insertional mutagenesis in gene therapy and its consequences. Mol Ther 2004; 9: 5–13.
Bonini C, Grez M, Traversari C, Ciceri F, Marktel S, Ferrari G et al. Safety of retroviral gene marking with a truncated NGF receptor. Nat Med 2003; 9: 367–369.
Yee C, Savage PA, Lee PP, Davis MM, Greenberg PD . Isolation of high avidity melanoma-reactive CTL from heterogeneous populations using peptide-MHC tetramers. J Immunol 1999; 162: 2227–2234.
Lamers CHJ, Sleijfer S, Willemsen RA, Debets R, Kruit WHJ, Gratama JW et al. Adoptive immuno-gene therapy of cancer with single chain antibody [scFv(Ig)] gene modified T lymphocytes. J Biol Reg Homeost Agents 2004; 18: 134–140.
Acknowledgements
The authors thank Corrien Groot-v. Ruyven, Sabine Langeveld and Barbara Luider-Vrieling for their technical assistance. This work has been supported by the Dutch Cancer Foundation (DDHK99-1865), the EU Grant QLK3-1999-01-262, and the Cancer Research Institute, New York (Clinical Investigation Grant ‘Immunogenetherapy of metastatic renal cell cancer patients’).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lamers, C., Willemsen, R., van Elzakker, P. et al. Phoenix-ampho outperforms PG13 as retroviral packaging cells to transduce human T cells with tumor-specific receptors: implications for clinical immunogene therapy of cancer. Cancer Gene Ther 13, 503–509 (2006). https://doi.org/10.1038/sj.cgt.7700916
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.cgt.7700916
Keywords
This article is cited by
-
T lymphocytes facilitate brain metastasis of breast cancer by inducing Guanylate-Binding Protein 1 expression
Acta Neuropathologica (2018)
-
Enhanced recruitment of genetically modified CX3CR1-positive human T cells into Fractalkine/CX3CL1 expressing tumors: importance of the chemokine gradient
Journal for ImmunoTherapy of Cancer (2016)
-
Treatment of Metastatic Renal Cell Carcinoma With CAIX CAR-engineered T cells: Clinical Evaluation and Management of On-target Toxicity
Molecular Therapy (2013)
-
Retroviral vectors for clinical immunogene therapy are stable for up to 9 years
Cancer Gene Therapy (2008)
-
Gene-modified T cells for adoptive immunotherapy of renal cell cancer maintain transgene-specific immune functions in vivo
Cancer Immunology, Immunotherapy (2007)