Abstract
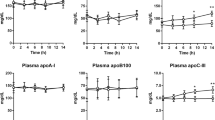
OBJECTIVES: After 10 d of orlistat administration (120 mg three times/day), the primary objective was to determine the drug's effect on postprandial plasma lipoprotein lipase (LPL) and hepatic triglyceride lipase (HTGL) activities on day 10 after an oral fat-load. The secondary objectives were to determine the effects of orlistat on 12 h postprandial measures of: (1) preheparin HTGL and LPL; and (2) serum triglycerides, very-low-density lipoprotein cholesterol, high-density lipoprotein cholesterol, low-density lipoprotein cholesterol and free fatty acids.
METHODS: Twenty-four normal-weight, healthy male volunteers were randomized to either 120 mg orlistat (n=12) or placebo (n=12) three times a day with meals for 10 d. Preheparin LPL and HTGL activities and LPL specific activity were measured in the fasted state on days 1, 5, and 10. On days 5 and 10 the study medication (orlistat or placebo) was taken at the beginning of a fat-rich breakfast and serum lipid and lipoprotein levels monitored for 12 h postprandially. On day 10, 15 min postheparin HTGL activity was measured 8 h after the fat-rich breakfast.
RESULTS: No differences were found between groups in fasting levels of preheparin LPL or HTGL activity or in LPL-specific activity on days 1, 5 and 10. No difference was found between the two treatment groups in postheparin HTGL activity 8 h after the fat-rich breakfast. Also, no differences were found between the two groups in plasma triglycerides or lipoproteins.
CONCLUSION: The results indicate that the oral administration of orlistat (120 mg t.i.d.) does not significantly alter plasma triglycerides or lipoproteins, and that the inhibitory effect of orlistat on lipases is limited to the gastrointestinal tract and is not manifested systemically.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Blum-Kaelin D, Hill H . Effect of tetrahydrolipstatin (RO 180647/002, THL) on rat, dog and human post-heparin plasma lipolytic activity in vitro Hoffman-LaRoche GCR 1990 B-103: 367.
Kamm JJ . Oncogenicity (feeding) study with RO 180647 in the rat Hoffmann-LaRoche GCR 1996 N-138: 885.
Eckel RH, Goldberg IJ, Steiner L, Yost TJ, Paterniti JR Jr . Plasma lipolytic activity. Relationship to postheparin lipolytic activity and evidence for metabolic regulation Diabetes 1988 37: 610–615.
Van Gaal LF, Broom JI, Enzi G, Toplak H . Efficacy and tolerability of orlistat in the treatment of obesity: A 6-month dose-ranging study. Orlistat dose-ranging study group Eur J Clin Pharmacol 1999 54: 125–132.
Glaser DS, Yost TJ, Eckel RH . Preheparin lipoprotein lipolytic activities: Relationship to plasm lipoproteins and postheparin lipolytic activities J Lipid Res 1992 33: 209–214.
Goers JW, Pedersen ME, Kern PA, Ong J, Schotz MC . An enzyme-linked immunoassay for lipoprotein lipase Anal Biochem 1987 166: 27–35.
Van Gent CM, van der Voort HA, de Bruyn AM, Klein F . Cholesterol determinations. A comparative study of methods with special reference to enzymatic procedures Clin Chim Acta 1977 75: 243–251.
Wu LL, Warnick GR, Wu JT, Williams RR, Lalouel J . A rapid micro-scale procedure for determination of the total lipid profile Clin Chem 1989 35: 1486–1491.
Kohlmeier M . Direct enzymic measurement of glycerides in serum and in lipoprotein fractions Clin Chem 1986 32: 63–66.
Demacker PNM, Hijmans AGM, Jansen AP . Enzymatic extraction determinations of free fatty acids in serum compared Clin Chem 1982 28: 1765–1768.
Fernandez M, Borgstrom B . Effects of tetrahydrolipstatin, a lipase inhibitor, on absorption of fat from the intestine of the rat Biochim Biophys Acta 1989 1001: 249–255.
Zhi J, Melia AT, Eggers H, Joly R, Patel IH . Review of limited systemic absorption of orlistat, a lipase inhibitor, in healthy human volunteers J Clin Pharmacol 1995 35: 1103–1108.
Drent ML, Larsson I, William-Olsson T, Quaade F, Czubayko F, von Bergmann K, Strobel W, Sjostrom L, van der Veen EA . Orlistat (RO 19-0647), a lipase inhibitor, in the treatment of human obesity: A multiple dose study Int J Obes 1995 19: 221–226.
Reitsma JB, Castro Cabezas M, de Bruin TW, Erkelens DW . Relationship between improved postprandial lipemia and low-density lipoprotein metabolism during treatment with tetrahydrolipstatin, a pancreatic lipase inhibitor Metabolism 1994 43: 293–298.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shepard, T., Jensen, D., Blotner, S. et al. Orlistat fails to alter postprandial plasma lipid excursions or plasma lipases in normal-weight male volunteers. Int J Obes 24, 187–194 (2000). https://doi.org/10.1038/sj.ijo.0801105
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ijo.0801105