Abstract
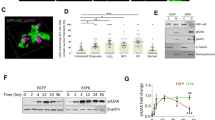
The tumour suppressor protein p53 is localized in the cell nucleus where it serves to initiate cellular responses to a variety of stresses, particularly DNA damage and has the capacity to transactivate stress response genes. An emerging body of evidence indicates that its action is also exerted through direct protein–protein interactions. An approach to understanding p53 function has been to analyse its positioning in relation to nuclear structures and we have shown that p53 can associate with the nuclear matrix. A potential nuclear matrix component for this association is actin. Here we show that p53 interacts with nuclear F-actin and we map the domains involved in this interaction. Using fluorescence resonance energy transfer, we demonstrate that the partition of p53 between F-actin bound and unbound forms is not constant, but is modulated by the presence of DNA damage, which increases binding. Our results indicate that the dynamic interaction of p53 with the nuclear matrix has to be considered for a full understanding of the mechanisms of the p53-mediated cellular response to DNA damage.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Albrechtsen N, Dornreiter I, Grosse F, Kim E, Wiesmuller L, Deppert W . 1999 Oncogene 18: 7706–7717
Andera L, Wasylyk B . 1997 Mol. Med. 3: 852–863
Bakalkin G, Selivanova G, Yakovleva T, Kiseleva E, Kashuba E, Magnusson KP, Szekely L, Klein G, Terenius L, Wiman KG . 1995 Cell 23: 362–369
Bastiaens PI, Majoul IV, Verveer PJ, Söling HD, Jovin TM . 1996 EMBO J. 15: 4246–4253
Cho Y, Gorina S, Jeffrey PD, Pavletich NP . 1994 Science 265: 346–355
Cook PR . 1991 Cell 66: 627–635
Deppert W, Haug M . 1986 Mol. Cell. Biol. 6: 2233–2240
Dutta A, Ruppert JM, Aster JC, Winchester E . 1993 Nature 365: 79–82
Fontoura BM, Sorokina EA, David E, Carroll RB . 1992 Mol. Cell. Biol. 12: 5145–5151
Giaccia AJ, Kastan MB . 1998 Genes Dev. 12: 2973–2983
Haugland RP . 1996 Handbook of fluorescent probes and research chemicals 6th Ed Molecular Probes, Eugene, OR
He D, Nickerson JA, Penman S . 1990 J. Cell Biol. 110: 569–580
Iborra FJ, Pombo A, Jackson DA, Cook PR . 1996 J. Cell Sci. 109: 1427–1436
Jares-Erijman EA, Jovin TM . 1996 J. Mol. Biol. 257: 597–617
Jiang M, Axe T, Holgate R, Rubbi C, Okorokov AL, Mee T, Milner J . 2001 Oncogene 20: 5449–5458
Jimenez GS, Khan SH, Stommel JM, Wahl GM . 1999 Oncogene 18: 7656–7665
Ko LJ, Shieh SY, Chen X, Jayaraman L, Tamai K, Taya Y, Prives C, Pan ZQ . 1997 Mol. Cell. Biol. 17: 7220–7229
Lee S, Elenbaas B, Levine A, Griffith J . 1995 Cell 81: 1013–1020
Léveillard T, Andera L, Bissonette N, Schaeffer L, Bracco L, Egly JM, Wasylyk B . 1996 EMBO J. 15: 1615–1624
Ljungman M . 2000 Neoplasia 2: 208–225
Lu H, Fisher R, Bailey P, Levine AJ . 1997 Mol. Cell. Biol. 17: 5923–5934
Mancini MA, Shan B, Nickerson JA, Penman S, Lee WH . 1994 Proc. Natl. Acad. Sci. USA 91: 418–422
Marechal V, Elenbaas B, Piette J, Nicolas JC, Levine AJ . 1994 Mol. Cell. Biol. 14: 7414–7420
May P, May E . 1999 Oncogene 18: 7621–7636
McCready SJ, Cook PR . 1984 J. Cell Sci. 70: 189–196
Meek DW . 1999 Oncogene 18: 7666–7675
Metcalfe S, Weeds A, Okorokov A, Milner J, Cockman M, Pope B . 1999 Oncogene 18: 2351–2355
Mullenders LH, van Kesteren van Leeuwen AC, van Zeeland AA, Natarajan AT . 1988 Nucleic Acids Res. 16: 10607–10622
Müller M, Strand S, Hug H, Heinemann EM, Walczak H, Hofmann WJ, Stremmel W, Krammer PH, Galle PR . 1997 J. Clin. Invest. 99: 403–413
Nakayasu H, Ueda K . 1985 Experimental Cell Res. 160: 319–330
Nguyen E, Besombes D, Debey P . 1998 Mol. Reprod. Dev. 50: 263–272
Oberosler P, Hloch P, Ramsperger U, Stahl H . 1993 EMBO J. 12: 2389–2396
Okorokov AL, Milner J . 1999 Mol. Cell. Biol. 19: 7501–7510
Pederson T . 1998 J. Molec. Biol. 277: 147–159
Rabinovitz I, Mercurio AM . 1997 J. Cell Biol. 139: 1873–1884
Rubbi CP, Milner J . 2000 Oncogene 19: 85–96
Szak ST, Pietenpol JA . 1999 J. Biol. Chem. 274: 3904–3909
Vogelstein B, Lane D, Levine AJ . 2000 Nature 408: 307–310
Walker KK, Levine AJ . 1996 Proc. Natl. Acad. Sci. USA 93: 15335–15340
Wang XW, Yeh H, Schaeffer L, Roy R, Moncollin V, Egly JM, Wang Z, Friedberg EC, Evans MK, Taffe BG, Bohr VA, Weeda G, Hoeijmakers JHJ, Forrester K, Harris CC . 1995 Nat. Genet. 10: 188–195
Acknowledgements
This work was supported by a Yorkshire Cancer Research program grant to J Milner.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Okorokov, A., Rubbi, C., Metcalfe, S. et al. The interaction of p53 with the nuclear matrix is mediated by F-actin and modulated by DNA damage. Oncogene 21, 356–367 (2002). https://doi.org/10.1038/sj.onc.1205112
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1205112
Keywords
This article is cited by
-
RhoC regulates radioresistance via crosstalk of ROCK2 with the DNA repair machinery in cervical cancer
Journal of Experimental & Clinical Cancer Research (2019)
-
Cofilin-mediated Neuronal Apoptosis via p53 Translocation and PLD1 Regulation
Scientific Reports (2017)
-
RUNX3 is involved in caspase-3-dependent apoptosis induced by a combination of 5-aza-CdR and TSA in leukaemia cell lines
Journal of Cancer Research and Clinical Oncology (2012)
-
The exonuclease ISG20 is directly induced by synthetic dsRNA via NF-κB and IRF1 activation
Oncogene (2004)
-
p53-associated 3′→5′ exonuclease activity in nuclear and cytoplasmic compartments of cells
Oncogene (2003)