Abstract
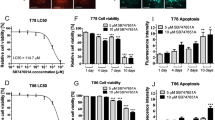
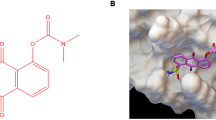
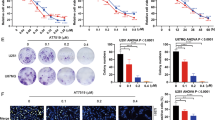
Signal transducer and activator of transcription-3 (STAT3) is constitutively activated in a variety of cancer types, including malignant gliomas. STAT3 is activated by phosphorylation of a tyrosine residue, after which it dimerizes and translocates into the nucleus. There it regulates the expression of several genes responsible for proliferation and survival at the transcriptional level. A selective inhibitor of STAT3 phosphorylation, AG490, has been shown to inhibit growth and induce apoptosis in some cancer cell types. However, although AG490 routinely shows in vitro anticancer activity, it has not consistently demonstrated an in vivo anticancer effect in animal models. Here, we have tested WP1066, a novel inhibitor structurally related to AG490 but significantly more potent and active, against human malignant glioma U87-MG and U373-MG cells in vitro and in vivo. IC50 values for WP1066 were 5.6 μ M in U87-MG cells and 3.7 μ M in U373-MG cells, which represents 18-fold and eightfold increases in potency, respectively, over that of AG490. WP1066 activated Bax, suppressed the expression of c-myc, Bcl-XL and Mcl-1, and induced apoptosis. Systemic intraperitoneal administration of WP1066 in mice significantly (P<0.001) inhibited the growth of subcutaneous malignant glioma xenografts during the 30-day follow-up period. Immunohistochemical analysis of the excised tumors revealed that phosphorylated STAT3 levels in the WP1066 treatment group remained inhibited at 3 weeks after the final WP1066 injection, whereas tumors from the control group expressed high levels of phosphorylated STAT3. We conclude that WP1066 holds promise as a therapeutic agent against malignant gliomas.
This is a preview of subscription content, access via your institution
Access options





Similar content being viewed by others
References
Bhattacharya S, Ray RM, Johnson LR . (2005). STAT3-mediated transcription of Bcl-2, Mcl-1 and c-IAP2 prevents apoptosis in polyamine-depleted cells. Biochem J 392: 335–344.
Bogler O, Weller M . (2002). Apoptosis in gliomas, and its role in their current and future treatment. Front Biosci 7: e339–e353.
Bowman T, Garcia R, Turkson J, Jove R . (2000). STATs in oncogenesis. Oncogene 19: 2474–2488.
Bromberg J . (2002). Stat proteins and oncogenesis. J Clin Invest 109: 1139–1142.
Bromberg JF . (2001). Activation of STAT proteins and growth control. Bioessays 23: 161–169.
Bromberg JF, Wrzeszczynska MH, Devgan G, Zhao Y, Pestell RG, Albanese C et al. (1999). Stat3 as an oncogene. Cell 98: 295–303.
Darnell Jr JE . (1997). STATs and gene regulation. Science 277: 1630–1635.
Epling-Burnette PK, Liu JH, Catlett-Falcone R, Turkson J, Oshiro M, Kothapalli R et al. (2001). Inhibition of STAT3 signaling leads to apoptosis of leukemic large granular lymphocytes and decreased Mcl-1 expression. J Clin Invest 107: 351–362.
Fesik SW . (2005). Promoting apoptosis as a strategy for cancer drug discovery. Nat Rev Cancer 5: 876–885.
Ihle JN . (2001). The Stat family in cytokine signaling. Curr Opin Cell Biol 13: 211–217.
Ito H, Kanzawa T, Miyoshi T, Hirohata S, Kyo S, Iwamaru A et al. (2005). Therapeutic efficacy of PUMA for malignant glioma cells regardless of p53 status. Hum Gene Ther 16: 685–698.
Kanzawa T, Iwado E, Aoki H, Iwamaru A, Hollingsworth EF, Sawaya R et al. (2006). Ionizing radiation induces apoptosis and inhibits neuronal differentiation in rat neural stem cells via the c-Jun NH(2)-terminal kinase (JNK) pathway. Oncogene 25: 3638–3648.
Kanzawa T, Zhang L, Xiao L, Germano IM, Kondo Y, Kondo S . (2005). Arsenic trioxide induces autophagic cell death in malignant glioma cells by upregulation of mitochondrial cell death protein BNIP3. Oncogene 24: 980–991.
Kapoor GS, O'Rourke DM . (2003). Mitogenic signaling cascades in glial tumors. Neurosurgery 52: 1425–1434.
Kiuchi N, Nakajima K, Ichiba M, Fukada T, Narimatsu M, Mizuno K et al. (1999). STAT3 is required for the gp130-mediated full activation of the c-myc gene. J Exp Med 189: 63–73.
Kleihues P, Louis DN, Scheithauer BW, Rorke LB, Reifenberger G, Burger PC et al. (2002). The WHO classification of tumors of the nervous system. J Neuropathol Exp Neurol 61: 215–225; discussion 226–219.
Kondo Y, Hollingsworth EF, Kondo S . (2004). Molecular targeting for malignant gliomas (Review). Int J Oncol 24: 1101–1109.
Konnikova L, Simeone MC, Kruger MM, Kotecki M, Cochran BH . (2005). Signal transducer and activator of transcription 3 (STAT3) regulates human telomerase reverse transcriptase (hTERT) expression in human cancer and primary cells. Cancer Res 65: 6516–6520.
Levy DE, Darnell Jr JE . (2002). Stats: transcriptional control and biological impact. Nat Rev Mol Cell Biol 3: 651–662.
Lichtor T, Libermann TA . (1994). Coexpression of interleukin-1 beta and interleukin-6 in human brain tumors. Neurosurgery 34: 669–672.
Meydan N, Grunberger T, Dadi H, Shahar M, Arpaia E, Lapidot Z et al. (1996). Inhibition of acute lymphoblastic leukaemia by a Jak-2 inhibitor. Nature 379: 645–648.
Minn AJ, Gupta GP, Siegel PM, Bos PD, Shu W, Giri DD et al. (2005). Genes that mediate breast cancer metastasis to lung. Nature 436: 518–524.
Niu G, Wright KL, Huang M, Song L, Haura E, Turkson J et al. (2002). Constitutive Stat3 activity up-regulates VEGF expression and tumor angiogenesis. Oncogene 21: 2000–2008.
Ohgaki H, Dessen P, Jourde B, Horstmann S, Nishikawa T, Di Patre PL et al. (2004). Genetic pathways to glioblastoma: a population-based study. Cancer Res 64: 6892–6899.
Rahaman SO, Harbor PC, Chernova O, Barnett GH, Vogelbaum MA, Haque SJ . (2002). Inhibition of constitutively active Stat3 suppresses proliferation and induces apoptosis in glioblastoma multiforme cells. Oncogene 21: 8404–8413.
Reardon DA, Rich JN, Friedman HS, Bigner DD . (2006). Recent advances in the treatment of malignant astrocytoma. J Clin Oncol 24: 1253–1265.
Riedl SJ, Shi Y . (2004). Molecular mechanisms of caspase regulation during apoptosis. Nat Rev Mol Cell Biol 5: 897–907.
Schaefer LK, Ren Z, Fuller GN, Schaefer TS . (2002). Constitutive activation of Stat3alpha in brain tumors: localization to tumor endothelial cells and activation by the endothelial tyrosine kinase receptor (VEGFR-2). Oncogene 21: 2058–2065.
Song H, Wang R, Wang S, Lin J . (2005). A low-molecular-weight compound discovered through virtual database screening inhibits Stat3 function in breast cancer cells. Proc Natl Acad Sci USA 102: 4700–4705.
Sonoda Y, Ozawa T, Hirose Y, Aldape KD, McMahon M, Berger MS et al. (2001). Formation of intracranial tumors by genetically modified human astrocytes defines four pathways critical in the development of human anaplastic astrocytoma. Cancer Res 61: 4956–4960.
Sun J, Blaskovich MA, Jove R, Livingston SK, Coppola D, Sebti SM . (2005). Cucurbitacin Q: a selective STAT3 activation inhibitor with potent antitumor activity. Oncogene 24: 3236–3245.
Turkson J, Zhang S, Palmer J, Kay H, Stanko J, Mora LB et al. (2004). Inhibition of constitutive signal transducer and activator of transcription 3 activation by novel platinum complexes with potent antitumor activity. Mol Cancer Ther 3: 1533–1542.
Van Meir E, Sawamura Y, Diserens AC, Hamou MF, de Tribolet N . (1990). Human glioblastoma cells release interleukin 6 in vivo and in vitro. Cancer Res 50: 6683–6688.
Wang P, Valentijn AJ, Gilmore AP, Streuli CH . (2003). Early events in the anoikis program occur in the absence of caspase activation. J Biol Chem 278: 19917–19925.
Weissenberger J, Loeffler S, Kappeler A, Kopf M, Lukes A, Afanasieva TA et al. (2004). IL-6 is required for glioma development in a mouse model. Oncogene 23: 3308–3316.
Yau CY, Wheeler JJ, Sutton KL, Hedley DW . (2005). Inhibition of integrin-linked kinase by a selective small molecule inhibitor, QLT0254, inhibits the PI3K/PKB/mTOR, Stat3, and FKHR pathways and tumor growth, and enhances gemcitabine-induced apoptosis in human orthotopic primary pancreatic cancer xenografts. Cancer Res 65: 1497–1504.
Acknowledgements
We thank Ms E Faith Hollingsworth for her technical help and Ms Kathryn Carnes for editing. This work was supported by a generous donation to Y Kondo and S Kondo from the Anthony D Bullock III Foundation and by Cancer Center Support Grant CA16672 to MD Anderson Cancer Center from the National Cancer Institute.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Iwamaru, A., Szymanski, S., Iwado, E. et al. A novel inhibitor of the STAT3 pathway induces apoptosis in malignant glioma cells both in vitro and in vivo. Oncogene 26, 2435–2444 (2007). https://doi.org/10.1038/sj.onc.1210031
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210031
Keywords
This article is cited by
-
Elevated FAM134B expression induces radiation-sensitive in hepatocellular carcinoma
BMC Cancer (2023)
-
Deciphering STAT3 signaling potential in hepatocellular carcinoma: tumorigenesis, treatment resistance, and pharmacological significance
Cellular & Molecular Biology Letters (2023)
-
Molecular characterization and function of JAK/STAT pathway in IPEC-J2 cells during Clostridium perfringens beta2 toxin stimulation
Veterinary Research Communications (2023)
-
p52 signaling promotes cellular senescence
Cell & Bioscience (2022)
-
EGFR signaling promotes nuclear translocation of plasma membrane protein TSPAN8 to enhance tumor progression via STAT3-mediated transcription
Cell Research (2022)