Abstract
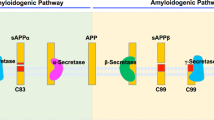
Signalling through the receptor protein Notch, which is involved in crucial cell-fate decisions during development, requires ligand-induced cleavage of Notch. This cleavage occurs within the predicted transmembrane domain, releasing the Notch intracellular domain (NICD), and is reminiscent of γ-secretase-mediated cleavage of β-amyloid precursor protein (APP), a critical event in the pathogenesis of Alzheimer's disease. A deficiency in presenilin-1 (PS1) inhibits processing of APP by γ-secretase in mammalian cells, and genetic interactions between Notch and PS1 homologues in Caenorhabditis elegans indicate that the presenilins may modulate the Notch signalling pathway1,2,3,4. Here we report that, in mammalian cells, PS1 deficiency also reduces the proteolytic release of NICD from a truncated Notch construct, thus identifying the specific biochemical step of the Notch signalling pathway that is affected by PS1. Moreover, several γ-secretase inhibitors block this same step in Notch processing, indicating that related protease activities are responsible for cleavage within the predicted transmembrane domains of Notch and APP. Thus the targeting of γ-secretase for the treatment of Alzheimer's disease may risk toxicity caused by reduced Notch signalling.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Levitan, D. & Greenwald, I. Facilitation of lin-12-mediated signalling by sel-12, a Caenorhabditis elegans S182 Alzheimer's disease gene. Nature 377, 351–354 (1995).
Wong, P. C. et al. Presenilin 1 is required for Notch1 and DII1 expression in the paraxial mesoderm. Nature 387, 288–292 (1997).
Shen, J. et al. Skeletal and CNS defects in presenilin-1-deficient mice. Cell 89, 629–639 (1997).
De Strooper, B. et al. Deficiency of presenilin-1 inhibits the normal cleavage of amyloid precursor protein. Nature 391, 387–390 (1998).
Logeat, F. et al. The Notch1 receptor is cleaved constitutively by a furin-like convertase. Proc. Natl Acad. Sci. USA 95, 8108–8112 (1998).
Blaumueller, C. M., Qi, H., Zagouras, P. & Artavanis-Tsakonas, S. Intracellular cleavage of Notch leads to a heterdimeric receptor on the plasma membrane. Cell 90, 281–291 (1997).
Schroeter, E. H., Kisslinger, J. A. & Kopan, R. Notch-1 signalling requires ligand-induced proteolytic release of intracellular domain. Nature 393, 382–386 (1998).
Struhl, G. & Adachi, A. Nuclear access and action of notch in vivo. Cell 93, 649–660 (1998).
Lecourtois, M. & Schweisguth, F. Indirect evidence for Delta-dependent intracellular processing of notch in Drosophila embryos. Curr. Biol. 8, 771–774 (1998).
Lendon, C., Ashall, F. & Goate, A. Exploring the etiology of Alzheimer's disease using molecular genetics. J. Am. Med. Assoc. 277, 825–831 (1997).
Levitan, D. & Greenwald, I. Effects of SEL-12 presenilin on LIN-12 localization and function in Caenorhabditis elegans. Development 125, 3599–3606 (1998).
De Strooper, B. et al. Production of intracellular amyloid-containing fragments in hippocampal neurons expressing human amyloid precursor protein and protection against amyloidogenesis by subtle amino acid substitutions in the rodent sequence. EMBO J. 14, 4932–4938 (1995).
Kopan, R., Schroeter, E. H., Weintraub, H. & Nye, J. S. Signal transduction by activated mNotch: importance of proteolytic processing and its regulation by the extracellular domain. Proc. Natl Acad. Sci. USA 93, 1683–1688 (1996).
Tischer, E. & Cordell, B. Beta-amyloid precursor protein. Location of transmembrane domain and specificity of gamma-secretase cleavage. J. Biol. Chem. 271, 21914–21919 (1996).
Brown, M. S. & Goldstein, J. L. The SREBP pathway: regulation of cholesterol metabolism by proteolysis of a membrane-bound transcription factor. Cell 89, 331–340 (1997).
Ross, S. L. et al. Amyloid precursor protein processing in sterol regulatory element-binding protein site 2 protease-deficient Chinese hamster ovary cells. J. Biol. Chem. 273, 15309–15312 (1998).
Wolfe, M. S. et al. Asubstrate-based difluoro ketone selectively inhibits Alzheimer's gamma-secretase activity. J. Med. Chem. 41, 6–9 (1998).
Higaki, J., Quon, D., Zhong, Z. & Cordell, B. Inhibition of beta-amyloid formation identifies proteolytic precursors and subcellular site of catabolism. Neutron 14, 651–659 (1995).
Citron, M. et al. Evidence that the 42- and 40-amino acid forms of amyloid beta protein are generated from the beta-amyloid precursor protein by different protease activities. Proc. Natl Acad. Sci. USA 93, 13170–13175 (1996).
Klafki, H., Abramowski, D., Swoboda, R., Paganetti, P. A. & Staufenbiel, M. The carboxyl termini of beta-amyloid peptides 1–40 and 1–42 are generated by distinct gamma-secretase activities. J. Biol. Chem. 271, 28655–28659 (1996).
Yamazaki, T., Haass, C., Saido, T. C., Omura, S. & Ihara, Y. Specific increase in amyloid beta-protein 42 secretion ratio by calpain inhibition. Biochemistry 36, 8377–8383 (1997).
Wolfe, M. S. et al. Peptidomimetic probes and molecular modelling suggest Alzheimer's γ-secretase is an intra-membrane cleaving aspartyl protease. Biochemistry (in the press).
Ray, W. J. et al. Evidence for a physical interaction between presenilin and Notch. Proc. Natl Acad. Sci. USA 96, 3263–3268 ((1999).
Weidermann, A. et al. Formation of stable complexes between two Alzheimer's disease gene products: presenilin-2 and beta-amyloid precursor protein. Nature Med. 3, 328–332 (1997).
Xia, W., Zhang, J., Perez, R., Koo, E. H. & Selkoe, D. J. Interaction between amyloid precursor protein and presenilins in mammalian cells: implications for the pathogenesis of Alzheimer disease. Proc. Natl Acad. Sci. USA 94, 8208–8213 (1997).
Saftig, P. & de Strooper, B. Downregulation of PS1 expression in neurons decreases beta-amyloid production: a biochemical link between the two major familial Alzheimer's disease genes. Mol. Psychiat. 3, 287–289 (1998).
Rawson, R. B. et al. Complementation cloning of S2P, a gene encoding a putative metalloprotease required for intramembrane cleavage of SREBPs. Mol. Cell 1, 47–57 (1997).
Varnum-Finney, B. et al. The Notch ligand, Jagged-1, influences the development of primitive hematopoietic precursor cells. Blood 91, 4084–4091 (1998).
Robey, E. & Fowlkes, B. J. The αβ versus αδ T-cell lineage choice. Curr. Opin. Immunol. 10, 181–187 (1998).
Chan, Y. M. & Jan, Y. N. Roles for proteolysis and trafficking in notch maturation and signal transduction. Cell 94, 423–426 (1998).
Acknowledgements
This work was supported by the FWO-Vlaanderen, the Human Frontier of Science Program, the Flemish Institute for Biotechnology (VIB) and the K. U. Leuven. B.D.S., W.A., P.C. and K.C. are researchers of the FWO. R.K., M.S.W. and A.G. are supported by the NIH; W.J.R. is supported by a fellowship from the KECK foundation; J.S.M. is supported in part by the Markey Special Emphasis Pathway in Human Pathobiology; P.S. is supported by the Deutsche Forschungsgemeinschaft. We thank A. Roebroek and J. Gordon for helpful comments.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
De Strooper, B., Annaert, W., Cupers, P. et al. A presenilin-1-dependent γ-secretase-like protease mediates release of Notch intracellular domain. Nature 398, 518–522 (1999). https://doi.org/10.1038/19083
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/19083
This article is cited by
-
Non-canonical non-genomic morphogen signaling in anucleate platelets: a critical determinant of prothrombotic function in circulation
Cell Communication and Signaling (2024)
-
The advent of Alzheimer treatments will change the trajectory of human aging
Nature Aging (2024)
-
TM2D3, a mammalian homologue of Drosophila neurogenic gene product Almondex, regulates surface presentation of Notch receptors
Scientific Reports (2023)
-
Amyloid β-based therapy for Alzheimer’s disease: challenges, successes and future
Signal Transduction and Targeted Therapy (2023)
-
Molecular sensing of mechano- and ligand-dependent adhesion GPCR dissociation
Nature (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.