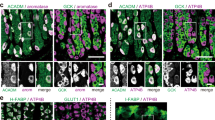
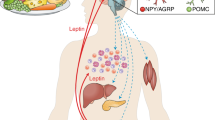
Abstract
The circulating peptide leptin, which is the product of the ob gene1, provides feedback information on the size of fat stores to central Ob receptors2,3 that control food intake and body-weight homeostasis4,5,6. Leptin has so far been reported to be secreted only by adipocytes1 and the placenta7. Here we show that leptin messenger RNA and leptin protein are present in rat gastric epithelium, and that cells in the glands of the gastric fundic mucosa are immunoreactive for leptin. The physiological function of this previously unsuspected source of leptin is unknown. However, both feeding and administration of CCK-8 (the biologically active carboxy-terminal end of cholecystokinin) result in a rapid and large decrease in both leptin cell immunoreactivity and the leptin content of the fundic epithelium, with a concomitant increase in the concentration of leptin in the plasma. These results indicate that gastric leptin may be involved in early CCK-mediated effects activated by food intake, possibly including satiety.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Zhang, Y.et al. Positional cloning of the mouse ob gene and its human homologue. Nature 372, 425–432 (1994).
Tartaglia, L.et al. Identification and expression cloning of a leptin receptor, OB-R. Cell 83, 126–127 (1995).
Lee, G.-H.et al. Abnormal splicing of the leptin receptor in diabetic mice. Nature 379, 632–635 (1996).
Campfield, L. A.et al. Recombinant mouse OB protein: evidence of a peripheral signal linking adiposity and central neural networks. Science 269, 546–549 (1995).
Pelleymounter, M., Cullen, M. & Baker, M. Effects of the obese gene product on body weight regulation in ob/ob mice. Science 269, 540–543 (1995).
Halaas, J. L.et al. Weight-reducing effects of the plasma protein encoded by the obese gene. Science 269, 543–546 (1995).
Masuzaki, H.et al. Non-adipose tissue production of leptin: leptin as a novel placenta-derived hormone in humans. Nature Med. 3, 1029–1033 (1997).
Hainault, I., Guerre-Millo, Guichard, C. & Lavau, M. Differential regulation of adipose tissue glucose transporter in genetic obesity (fatty rat). Selective increase in the adipose cell/muscle glucose transporter (Glu4) expression. J. Clin. Invest. 87, 1127–1131 (1991).
Dugail, I.et al. Adipose-tissue-specific increase in glyceraldehyde-3-phosphate dehydrogenase activity and mRNA amounts in suckling pre-obese Zucker rats. Biochem. J. 254, 483–487 (1988).
Ahim, R. S.et al. Role of leptin in the neuroendocrine response to fasting. Nature 382, 250–252 (1996).
Hardie, L. J., Vernon Rayner, D., Holmes, S. & Trayhum, P. Circulating leptin levels are modulated by fasting, cold exposure and insulin administration in lean but not Zucker (fa/fa) rats as measured by ELISA. Biochem. Biophys. Res. Commun. 223, 660–665 (1996).
Weigle, D. S.et al. Effect of fasting, refeeding, and dietary fat restriction on plasma leptin levels. J. Clin. Endocrinol. Metab. 82, 561–565 (1997).
Saladin, R.et al. Transient increase in obese gene expression after food intake or insulin administration. Nature 377, 527–529 (1995).
Mac Dougald, O. A., Hwang, C. S., Fan, H. & Lane, M. D. Regulated expression of the obese gene product (leptin) in white adipose tissue and 3T3-L1 adipocytes. Proc. Natl Acad. Sci. USA 92, 9034–9037 (1995).
Levy, J. R., leGall-Salmon, E., Santos, M., Pandak, W. M. & Stevens, W. Effect of enteral versus parenteral nutrition on leptin gene expression and release into the circulation. Biochem. Biophys. Res. Commun. 237, 98–102 (1997).
Trayhum, P., Ducan, J. S., Rayner, D. V. & Hardie, L. J. Rapid inhibition of ob gene expression and circulating leptin levels in lean mice by the β3-adrenoceptor agonists BRL35135A and ZD2079. Biochem. Biophys. Res. Commun. 228, 605–610 (1996).
Mantzoros, C. S.et al. Activation of β3-adrenergic receptors suppresses leptin expression and mediates a leptin-independent inhibition of food intake in mice. Diabetes 45, 909–914 (1996).
Barrachina, M. D., Martinez, V., Wang, L., Wei, J. Y. & Taché, Y. . Synergistic interaction between leptin and cholecystokinin to reduce short-term food intake in mice. Proc. Natl Acad. Sci. USA 94, 10455–10460 (1997).
Matson, C. A., Wiater, M. F., Kujiper, J. L. & Weigle, D. S. Synergy between leptin and cholecystokinin (CCK) to control daily caloric intake. Peptides 18, 1275–1278 (1997).
Koulischer, D., Moroder, L. & Deschodt-Lanckman, M. Degradation of cholecystokinin octapeptide, related fragments and analogs by human and rat plasma in vitro. Regul. Pept. 4, 127–139 (1982).
Rose, C.et al. Characterization and inhibition of a cholecystokinin inactivating serine peptidase. Nature 380, 403–409 (1996).
Buffa, R., Solcia, E. & Go, V. L. W. Immunohistochemical identification of the cholecystokinin cell in the intestinal mucosa. Gastroenterology 70, 528–532 (1976).
Liddle, R. A., Goldfine, I. D. & Williams, J. A. Bioassay of plasma cholecystokinin in rats: effects of food, trypsin inhibitor and alcohol. Gastroenterology 87, 542–549 (1984).
Gibbs, J., Young, C. & Smith, G. P. Cholecystokinin elicits satiety in rats with open gastric fistulas. Nature 245, 323–325 (1973).
Smith, GP., Jerome, C., Cushin, B. J., Eterno, T. & Simansky, K. J. Abdominal vagotomy blocks the satiety effect of cholecystokinin in the rat. Science 213, 1036–1037 (1981).
Wang, Y. H.et al. Two types of leptin-responsive gastric afferent terminals: an in vitro single-unit study in rats. Am. J. Physiol. 273, R833–R837 (1997).
Barzilai, N.et al. Leptin selectively decreases visceral adiposity and enhances insulin action. J. Clin. Invest. 100, 3105–3110 (1997).
Tanti, J. F.et al. Overexpression of a constitutively active form of phosphatidylinositol 3-kinase is sufficient to promote Glut4 translocation in adipocytes. J. Biol. Chem. 271, 25227–25232 (1996).
Bado, A.et al. H3-receptor regulation of vascular gastrin and somatostatin releases by the isolated perfused rat stomach. Yale J. Biol. Med. 67, 113–121 (1994).
Acknowledgements
This work was supported by INSERM and by the Ministère de l'Education et de la Recherche Scientifique (A.B.). L.S. was supported by MERST, K.S. by a grant from IRMAD, A.S. by a grant from FRM, and M.N.B. by a grant from Novo-Nordisk.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bado, A., Levasseur, S., Attoub, S. et al. The stomach is a source of leptin. Nature 394, 790–793 (1998). https://doi.org/10.1038/29547
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/29547
This article is cited by
-
Relationship between active Helicobacter pylori infection and risk factors of cardiovascular diseases, a cross-sectional hospital-based study in a Sub-Saharan setting
BMC Infectious Diseases (2022)
-
Reduced gut microbiota diversity in patients with congenital generalized lipodystrophy
Diabetology & Metabolic Syndrome (2022)
-
Leptin as a key regulator of the adipose organ
Reviews in Endocrine and Metabolic Disorders (2022)
-
Metabolic milieu and localization of ovarian leptin and receptor in queens under different reproduction phases
Veterinary Research Communications (2022)
-
Leptin signaling in breast cancer and its crosstalk with peroxisome proliferator-activated receptors α and γ
Clinical and Translational Oncology (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.