Abstract
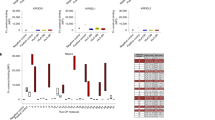
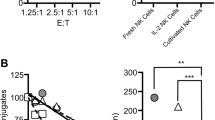
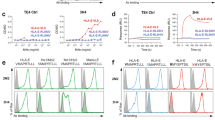
Natural killer (NK) cells destroy virus-infected and tumour cells, apparently without the need for previous antigen stimulation1. In part, target cells are recognized by their diminished expression of major histocompatibility complex (MHC) class I molecules, which normally interact with inhibitory receptors on the NK cell surface2,3,4,5,6,7,8. NK cells also express triggering receptors that are specific for non-MHC ligands; but the nature of the ligands recognized on target cells is undefined9,10,11,12,13,14. NKp46 is thought to be the main activating receptor for human NK cells9,15. Here we show that a soluble NKp46–immunoglobulin fusion protein binds to both the haemagglutinin of influenza virus and the haemagglutinin–neuraminidase of parainfluenza virus. In a substantial subset of NK cells, recognition by NKp46 is required to lyse cells expressing the corresponding viral glycoproteins. The binding requires the sialylation of NKp46 oligosaccharides, which is consistent with the known sialic binding capacity of the viral glycoproteins. These findings indicate how NKp46-expressing NK cells may recognize target cells infected by influenza or parainfluenza without the decreased expression of target-cell MHC class I protein.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Trinchieri, G. Biology of natural killer cells. Adv. Immunol. 47, 187–376 (1989).
Lanier, L. L. NK cell receptors. Annu. Rev. Immunol. 16, 359–393 (1998).
Ljunggren, H. G. & Karre, K. In search of the ‘missing self’: MHC molecules and NK recognition. Immunol. Today 11, 7–10 (1990).
Davis, D. M. et al. The human natural killer cell immune synapse. Proc. Natl Acad. Sci. USA 96, 15062–15067 (1999).
Braud, V. M. et al. HLA-E binds to natural killer cell receptors CD94/NKG2A, B and C. Nature 391, 795–799 (1998).
Colonna, M. & Samaridis, J. Cloning of immunoglobulin-superfamily members associated with HLA-C and HLA-B recognition by human natural killer cells. Science 268, 405–408 (1995).
Wagtmann, N. et al. Molecular clones of the p58 NK cell receptor reveal immunoglobulin-related molecules with diversity in both the extra- and intracellular domains. Immunity 2, 439–449 (1995).
D'Andrea, A. et al. Molecular cloning of NKB1. A natural killer cell receptor for HLA-B allotypes. J. Immunol. 155, 2306–2310 (1995).
Bottino, C., Biassoni, R., Millo R., Moretta, L. & Moretta, A. The human natural cytotoxicity receptors (NCR) that induce HLA class I independent NK cell triggering. Hum. Immunol. 61, 1–6 (2000).
Mandelboim, O. et al. Human CD16 as a lysis receptor mediating direct natural killer (NK) cell cytotoxicity. Proc. Natl Acad. Sci. USA 96, 5640–5644 (1999).
Pende, D. et al. Identification and molecular characterization of NKp30, a novel triggering receptor involved in natural cytotoxicity mediated by human natural killer cells. J. Exp. Med. 190, 1505–1516 (1999).
Cantoni, C. et al. NKp44, A triggering receptor involved in tumor cell lysis by activated human natural killer cells, is a novel member of the immunoglobulin superfamily. J. Exp. Med. 189, 787–796 (1999).
Biassoni, R. et al. The murine homologue of the human NKp46, a triggering receptor involved in the induction of natural cytotoxicity. Eur. J. Immunol. 29, 1014–1020 (1999).
Pessino, A. et al. Molecular cloning of NKp46: novel member of the immunoglobulin superfamily involved in triggering of natural cytotocxicity. J. Exp. Med. 188, 953–960 (1998).
Sivori, S. et al. NKp46 is the major triggering receptor involved in the natural cytotoxicity of fresh or cultured human NK cells. Correlation between surface density of NKp46 and natural cytotoxicity against autologous, allogeneic or xenogeneic target cells. Eur. J. Immunol. 29, 1656–1666 (1999).
Alsheikhly, A. et al. Sendai-virus-induced cell-mediated cytotoxicity in vitro. The role of viral glycoproteins in cell-mediated cytotoxicity. Scand. J. Immunol. 17, 129–138 (1983).
Alsheikhly, A. R., Orvell, C., Andersson, T. & Perlmann, P. The role of serologically defined epitopes on mumps virus HN–glycoprotein in the induction of virus-dependent cell-mediated cytotoxicity. Analysis with monoclonal antibodies. Scand. J. Immunol. 22, 529–538 (1985).
Maisner, A. & Herrler, G. Membrane cofactor protein with different types of N-glycans can serve as measles virus receptor. Virology 210, 479–481 (1995).
Varki, A. Sialic acids as ligands in recognition phenomena. FASEB J. 11, 248–255 (1997).
Weis, W. et al. Structure of the influenza virus hemagglutinin complexed with its receptor, sialic acid. Nature 333, 426–431 (1988).
De Lau, W. B., Kuipers, J., Voshol, H., Clevers, H. & Bast, B. J. HB4 antibody recognizes a carbohydrate structure on lymphocyte surface proteins related to HB6, CDw75, and CD76 antigens. J. Immunol. 150, 4911–4919 (1993).
Pinola, M., Renkonen, R., Majuri, M. L., Tiisala, S. & Saksela, E. Characterization of the E-selectin ligand on NK cells. J. Immunol. 152, 3586–3594 (1994).
Mandelboim, O. et al. Protection from lysis by natural killer cells of group 1 and 2 specificity is mediated by residue 80 in human histocompatibility leukocyte antigen C alleles and also occurs with empty major histocompatibility complex molecules. J. Exp. Med. 184, 913–922 (1996).
Peterhans, E., Bachi, T. & Yewdell, J. W. Evidence for different receptor sites in mouse spleen cells for the SV hemagglutinin-neuraminidase (HN) and fusion (F) glycoproteins. Virology 128, 366–376 (1983).
Yewdell, J. W. & Gerhard, W. Delineation of four antigenic sites on a paramyxovirus glycoprotein via which antibodies mediate distinct antiviral activities. J. Immunol. 128, 2670–2675 (1982).
Yewdell, J. W., Gerhard, W. & Bachi, T. Monoclonal anti-hemagglutinin antibodies detect irreversible antigenic alterations that coincide with the acid activation of influenza virus A/PR/8/34-mediated hemolysis. J. Virol. 48, 239–248 (1983).
Porgador, A., Mandelboim, O., Restifo, N. P., Strominger, J. L. Natural killer cell lines kill autologous beta2-microglobulin-deficient melanoma cells: implications for cancer immunotherapy. Proc. Natl Acad. Sci. USA 94, 13140–13145 (1997).
Brand, C. M. & Skehel, J. J. Crystalline antigen from the influenza virus envelope. Nature 238, 145–147 (1972).
Irving, B. A. & Weiss, A. The cytoplasmic domain of the T cell receptor zeta chain is sufficient to couple to receptor-associated signal transduction pathways. Cell 64, 891–901 (1991).
Acknowledgements
We thank N. P. Restifo for the 1106mel cells; A. Portner from St. Jude Children's Research Hospital for providing the pca-svhn plasmid; and J. Skehel and R. Gonsalves from National Institute of Medical Research for the purified HA. O.M. is supported by the Israel Science Foundation and The Charles H. Revson Foundation. A.P. is supported by the Israel Science Foundation and by the USA–Israel Binational Science Foundation. O.M. and A.P. are supported by the Israel Cancer Research Foundation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mandelboim, O., Lieberman, N., Lev, M. et al. Recognition of haemagglutinins on virus-infected cells by NKp46 activates lysis by human NK cells. Nature 409, 1055–1060 (2001). https://doi.org/10.1038/35059110
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/35059110
This article is cited by
-
Unwinding the Role of the CMG Helicase in Inborn Errors of Immunity
Journal of Clinical Immunology (2023)
-
Natural killer cells: a promising immunotherapy for cancer
Journal of Translational Medicine (2022)
-
Candida albicans evades NK cell elimination via binding of Agglutinin-Like Sequence proteins to the checkpoint receptor TIGIT
Nature Communications (2022)
-
Enhanced generation of influenza-specific tissue resident memory CD8 T cells in NK-depleted mice
Scientific Reports (2021)
-
Maternal natural killer cells at the intersection between reproduction and mucosal immunity
Mucosal Immunology (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.