Abstract
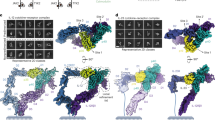
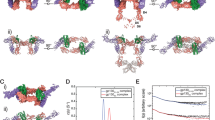
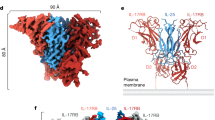
Inflammation, regardless of whether it is provoked by infection or by tissue damage, starts with the activation of macrophages which initiate a cascade of inflammatory responses by producing the cytokines interleukin-1 (IL-1) and tumour necrosis factor-α (ref. 1). Three naturally occurring ligands for the IL-1 receptor (IL1R) exist: the agonists IL-1α and IL-lβ and the IL-1-receptor antagonist IL1RA (ref. 2). IL-1 is the only cytokine for which a naturally occurring antagonist is known. Here we describe the crystal structure at 2.7 Å resolution of the soluble extracellular part of type-I IL1R complexed with IL1RA. The receptor consists of three immunoglobulin-like domains. Domains 1 and 2 are tightly linked, but domain three is completely separate and connected by a flexible linker. Residues of all three domains contact the antagonist and include the five critical IL1RA residues which were identified by site-directed mutagenesis3. A region that is important for biological function in IL-1β, the 'receptor trigger site', is not in direct contact with the receptor in the IL1RA complex. Modelling studies suggest that this IL-1β trigger site might induce a movement of domain 3.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Baumann, H. & Gauldie, J. The acute phase response. Immunol. Today 15, 74–80 (1994).
Dinarello, C. A. The interleukin-1 family: 10 years of discovery. FASEB J. 8, 1314–1325 (1994).
Evans, R. J. et al. Mapping receptor binding sites in interleukin (IL)-l receptor antagonist and IL-1β by site-directed mutagenesis. Identificaiton of a single site in IL-lra and two sites in IL-1β. J. Biol. Chem. 270, 11477–11483 (1995).
Schreuder, H. A. et al. Crystals of soluble interleukin-1 receptor complexed with its natural antagonist reveal a 1:1 receptor-ligand complex. FEBS Lett. 373, 39–40 (1995).
Schreuder, H. A. et al. Refined crystal structure of the interleukin-1 receptor antagonist. Presence of a disulfide link and a cis proline. Eur. J. Biochem. 227, 838–847 (1995).
Sims, J. E. et al. cDNA expression cloning of the IL-1 receptor, a member of the immunoglobulin superfamily. Science 241, 585–589 (1988).
Harpaz, Y. & Chothia, C. Many of the immunoglobulin superfamily domains in cell adhesion molecules and surface receptors belong to a new structural set which is close to that containing variable domains. J. Mol. Biol. 238, 528–539 (1994).
Hemmingsen, J. M., Gernert, K. M., Richardson, J. S. & Richardson, D. C. The tyrosine corner: A feature of most greek key β-barrel proteins. Prot. Sci. 3, 1927–1937 (1994).
Kabsch, W. & Sander, C. Dictionary of protein secondary structure: Pattern recognition of hydrogen-bonded and geometrical features. Biopolymers 22, 2577–2637 (1983).
Clackson, T. & Wells, J. A. A hot spot of binding energy in a hormone-receptor interface. Science 267, 383–386 (1995).
Yanofksy, S. D. et al. High affinity type I interleukin 1 receptor antagonists discovered by screening recombinant peptide libraries. Proc. Natl Acad. Sci. USA 93, 7381–7386 (1996).
Banner, D. W. et al. Crystal structure of the soluble human 55 kd TNF receptor-human TNFβ complex: Implications for TNF receptor activation. Cell 73, 431–445 (1993).
Vos, A. M., Ultsch, M. & Kossiakoff, A. A. Human growth hormone and extracellular domain of its receptor: Crystal structure of the complex. Science 255, 306–312 (1992).
Walter, M. R. et al. Crystal structure of a complex between interferon-γ and its soluble high-affinity receptor. Nature 376, 230–235 (1995).
Livnah, O. et al. Functional mimicry of a protein hormone by a peptide agonist: The EPO receptor complex at 2.8 Å. Science 273, 464–471 (1996).
Greenfeder, S. A. et al. Molecular cloning and characterization of a second subunit of the interleukin 1 receptor complex. J. Biol. Chem. 270, 13757–13765 (1995).
Priestle, J. P., Schär, H.-P. & Grütter, M. G. Crystal structure of the cytokine interleukin-1β. EMBOJ. 7, 339–343 (1988).
Ju, G. et al. Conversion of the interleukin 1 receptor antagonist into an agonist by site-specific mutagenesis. Proc. Natl Acad. Sci. USA 88, 2658–2662 (1991).
Kabsch, W. Automatic processing of rotation diffraction data from crystals of initially unknown symmetry and cell constants. J. Appl. Crystallogr. 26, 795–800 (1993).
Navaza, J. AMoRe: an automated package for molecular replacement. Acta Crystallogr. A 50, 157–163 (1994).
Wang, J. et al. Atomic structure of a fragment of human CD4 containing two immunoglobulin-like domains. Nature 348, 411–418 (1990).
Collaborative Computational Project Number 4. The CCP4 suite: Programs for protein crystallography. Acta Crystallogr. D 50, 760–763 (1994).
Terwilliger, T. C . & Eisenberg, D. Unbiased three-dimensional refinement of heavy-atom parameters by correlation of origin-removed Patterson functions. Acta Crystallogr. A 39, 813–817 (1983).
Otwinowsky, W. Maximum likelihood refinement of heavy atom parameters. In Isomorphous Replacement and Anomalous Scattering Proc. CCP4 Study Weekend, 25-26 January 1991 (compiled by Wolf, W., Evans, P. R. & Lesly, A. G. W.) 80–86 (1991).
Read, R.J. & Schierbeek, A. J. A phased translation function. J. Appl Crystallogr. 13, 490–495 (1988).
Zhang, K. Y. J. SQUASH-combining constraints for macromolecular phase refinement and extension. Acta Crystallogr. D 49, 213–222 (1993).
Jones, T. A., Zou, J.-Y., Cowan, S. W. & Kjeldgaard, M. Improved methods for building protein models in electron density maps and the location of errors in these models. Acta Crystallogr. A 47, 110–119 (1991).
Brünger, A. X-PLOR, version 3.1. A system for X-ray crystallography and NMR. (Yale University Press, New Haven, Connecticut, 1992).
Kraulis, P. MOLSCRIPT: A program to produce both detailed and schematic plots of protein structures. J. Appl. Crystallogr. 24, 946–950 (1991).
Nicholls, A., Sharp, K. A. & Honig, B. Protein folding and association: Insights from the interfacial and thermodynamic properties of hydrocarbons. Proteins 11, 281–296 (1991).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schreuder, H., Tardif, C., Trump-Kallmeyer, S. et al. A new cytokine-receptor binding mode revealed by the crystal structure of the IL-1 receptor with an antagonist. Nature 386, 194–200 (1997). https://doi.org/10.1038/386194a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/386194a0
This article is cited by
-
Discovery of a selective and biologically active low-molecular weight antagonist of human interleukin-1β
Nature Communications (2023)
-
Targeted therapeutics and novel signaling pathways in non-alcohol-associated fatty liver/steatohepatitis (NAFL/NASH)
Signal Transduction and Targeted Therapy (2022)
-
Comparative Analyses of the Conformational Dynamics Between the Soluble and Membrane-Bound Cytokine Receptors
Scientific Reports (2020)
-
Novel Functions of Interleukin-1 Beta in Spotted Halibut (Verasper variegatus) at Its Embryonic and Metamorphic Stages
Journal of Ocean University of China (2018)
-
The Role of Interleukin-1 in the Pathogenesis of Cancer and its Potential as a Therapeutic Target in Clinical Practice
Oncology and Therapy (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.