Abstract
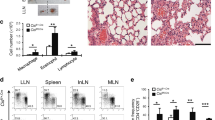
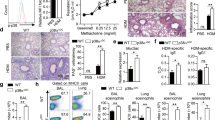
The transcription factor GATA-3 is expressed in T helper 2 (TH2) but not TH1 cells and plays a critical role in TH2 differentiation and allergic airway inflammation in vivo. Mice that lack the p50 subunit of nuclear factor κB (NF-κB) are unable to mount airway eosinophilic inflammation. We show here that this is not due to defects in TH2 cell recruitment but due to the inability of the p50−/− mice to produce interleukin 4 (IL-4), IL-5 and IL-13: cytokines that play distinct roles in asthma pathogenesis. CD4+ T cells from p50−/− mice failed to induce Gata3 expression under TH2-differentiating conditions but showed unimpaired T-bet expression and interferon γ (IFN-γ) production under TH1-differentiating conditions. Inhibition of NF-κB activity prevented GATA-3 expression and TH2 cytokine production in developing, but not committed, TH2 cells. Our studies provide a molecular basis for the need for both T cell receptor and cytokine signaling for GATA-3 expression and, in turn, TH2 differentiation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Walker, C. et al. Allergic and non-allergic asthmatics have distinct patterns of T-cell activation and cytokine production in peripheral blood and bronchoalveolar lavage. Am. Rev. Respir. Dis. 146, 109–115 (1992).
Robinson, D. S. et al. Predominant TH2-like bronchoalveolar T-lymphocyte population in atopic asthma. N. Engl. J. Med. 326, 298–304 (1992).
Wills-Karp, M. Immunologic basis of antigen-induced airway hyperresponsiveness. Annu. Rev. Immunol. 17, 255–281 (1999).
Ray, A. & Cohn, L. Th2 cells and GATA-3 in asthma: new insights into the regulation of airway inflammation. J. Clin. Invest. 104, 985–993. (1999).
Zhang, D.-H., Cohn, L., Ray, P., Bottomly, K. & Ray, A. Transcription factor GATA-3 is differentially expressed in Th1 and Th2 cells and controls Th2-specific expression of the interleukin-5 gene. J. Biol. Chem. 272, 21597–21603 (1997).
Zheng, W.-P. & Flavell, R. A. The transcription factor GATA-3 is necessary and sufficient for Th2 cytokine gene expression in CD4 T cells. Cell 89, 587–596 (1997).
Ting, C.-N., Olson, M. C., Barton, K. P. & Leiden, J. M. Transcription factor GATA-3 is required for development of the T-cell lineage. Nature 384, 474–478. (1996).
Zhang, D. H. et al. Inhibition of allergic inflammation in a murine model of asthma by expression of a dominant-negative mutant of GATA-3. Immunity 11, 473–482. (1999).
Baldwin, A. S. Jr The NF-κB and IκB proteins: new discoveries and insights. Annu. Rev. Immunol. 14, 649–681 (1996).
Sha, W. C. Regulation of immune responses by NF-κB/Rel transcription factors. J. Exp. Med. 187, 143–146 (1998).
Yang, L. et al. Essential role of nuclear factor κB in the induction of eosinophilia in allergic airway inflammation. J. Exp. Med. 188, 1739–1750. (1998).
Cohn, L., Homer, R. J., Marinov, A., Rankin, J. & Bottomly, K. Induction of airway mucus production by T helper 2 (Th2) cells: a critical role for interleukin-4 in cell recruitment but not mucus production. J. Exp. Med. 186, 1737–1747 (1997).
Cohn, L., Tepper, J. S. & Bottomly, K. IL-4-independent induction of airway hyperresponsiveness by Th2, but not Th1, cells. J. Immunol. 161, 3813–3816. (1998).
Ouyang, W. et al. Inhibition of Th1 development mediated by GATA-3 through an IL-4-independent mechanism. Immunity 9, 745–755. (1998).
Agarwal, S. & Rao, A. Modulation of chromatin structure regulates cytokine gene expression during T cell differentiation. Immunity 9, 765–775. (1998).
Siegel, M. D., Zhang, D.-H., Ray, P. & Ray, A. Activation of the interleukin-5 promoter by cAMP in murine EL-4 cells requires the GATA-3 and CLE0 elements. J. Biol. Chem. 270, 24548–24555 (1995).
Zhang, D.-H., Yang, L. & Ray, A. Differential responsiveness of the interleukin-5 (IL-5) and IL-4 genes to transcription factor GATA-3. J. Immunol. 161, 3817–3821 (1998).
Ranganath, S. et al. GATA-3-dependent enhancer activity in IL-4 gene regulation. J. Immunol. 161, 3822–3826 (1998).
Agarwal, S., Avni, O. & Rao, A. Cell-type-restricted binding of the transcription factor NFAT to a distal IL-4 enhancer in vivo. Immunity 12, 643–652 (2000).
George, K. M. et al. Embryonic expression and cloning of the murine GATA-3 gene. Development 120, 2673–2686. (1994).
Ouyang, W. et al. Stat6-independent GATA-3 autoactivation directs IL-4-independent Th2 development and commitment. Immunity 12, 27–37 (2000).
Ranger, A. M. et al. The transcription factor NF-ATc is essential for cardiac valve formation. Nature 392, 186–190 (1998).
Yoshida, H. et al. The transcription factor NF-ATc1 regulates lymphocyte proliferation and Th2 cytokine production. Immunity 8, 115–124 (1998).
Lin, Y. Z., Yao, S. Y., Veach, R. A., Torgerson, T. R. & Hawiger, J. Inhibition of nuclear translocation of transcription factor NF-κB by a synthetic peptide containing a cell membrane-permeable motif and nuclear localization sequence. J. Biol. Chem. 270, 14255–14258 (1995).
Liu, R. Y., Fan, C., Olashaw, N. E., Wang, X. & Zuckerman, K. S. Tumor necrosis factor-α-induced proliferation of human Mo7e leukemic cells occurs via activation of nuclear factor κB transcription factor. J. Biol. Chem. 274, 13877–13885 (1999).
McKenzie, A. N. et al. Structural comparison and chromosomal localization of the human and mouse IL-13 genes. J. Immunol. 150, 5436–5444 (1993).
Szabo, S. J. et al. A novel transcription factor, T-bet, directs Th1 lineage commitment. Cell 100, 655–669 (2000).
Jain, J., Loh, C. & Rao, A. Transcriptional regulation of the IL-2 gene. Curr. Opin. Immunol. 7, 333–342 (1995).
Kuo, C. T. & Leiden, J. M. Transcriptional regulation of T lymphocyte development and function. Annu. Rev. Immunol. 17, 149–187 (1999).
Jamieson, C., McCaffrey, P. G., Rao, A. & Sen, R. Physiologic activation of T cells via the T cell receptor induces NF-κB. J. Immunol. 147, 416–420 (1991).
Kang, S.-M., Tran, A.-C., Grilli, M. & Lenardo, M. J. NF-κB subunit regulation in nontransformed CD4+ T lymphocytes. Science 256, 1451–1456 (1992).
Robinson, D. et al. IGIF does not drive Th1 development but synergizes with IL-12 for interferon-gamma production and activates IRAK and NF-κB. Immunity 7, 571–581 (1997).
Matsumoto, S. et al. Interleukin-18 activates NF-κB in murine T helper type 1 cells. Biochem. Biophys. Res. Commun. 234, 454–457 (1997).
Sica, A. et al. Interaction of NF-κB and NFAT with the interferon-γ promoter. J. Biol. Chem. 272, 30412–30420 (1997).
Murphy, T. L., Cleveland, M. G., Kulesza, P., Magram, J. & Murphy, K. M. Regulation of interleukin 12 p40 expression through an NF-κB half-site. Mol. Cell. Biol. 15, 5258–5267 (1995).
Shen, C. H. & Stavnezer, J. Interaction of stat6 and NF-κB: direct association and synergistic activation of interleukin-4-induced transcription. Mol. Cell. Biol. 18, 3395–3404 (1998).
Delphin, S. & Stavnezer, J. Characterization of an interleukin 4 (IL-4) responsive region in the immunoglobulin heavy chain germline epsilon promoter: regulation by NF- IL-4, a C/EBP family member and NF-κB/p50. J. Exp. Med. 181, 181–192 (1995).
Mikita, T., Campbell, D., Wu, P., Williamson, K. & Schindler, U. Requirements for interleukin-4-induced gene expression and functional characterization of Stat6. Mol. Cell. Biol. 16, 5811–5820 (1996).
Sha, W. C., Liou, H.-C., Tuomanen, E. I. & Baltimore, D. Targeted disruption of the p50 subunit of NF-κB leads to multifocal defects in immune responses. Cell 80, 321–330 (1995).
Acknowledgements
We thank L. Glimcher for the antibody to T-bet. Supported by grants from the NIH (to A. R.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Das, J., Chen, CH., Yang, L. et al. A critical role for NF-κB in Gata3 expression and TH2 differentiation in allergic airway inflammation. Nat Immunol 2, 45–50 (2001). https://doi.org/10.1038/83158
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/83158
This article is cited by
-
Deficiency of PKCλ/ι alleviates the liver pathologic impairment of Schistosoma japonicum infection by thwarting Th2 response
Parasites & Vectors (2022)
-
Evidence of a genetically driven metabolomic signature in actively inflamed Crohn’s disease
Scientific Reports (2022)
-
Is stress related to the presence and persistence of oncogenic human papillomavirus infection in young women?
BMC Cancer (2021)
-
NF-κB regulation in maternal immunity during normal and IUGR pregnancies
Scientific Reports (2021)
-
IRAK-M alters the polarity of macrophages to facilitate the survival of Mycobacterium tuberculosis
BMC Microbiology (2017)