Abstract
BACKGROUND/OBJECTIVES:
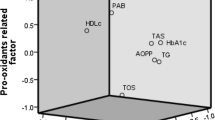
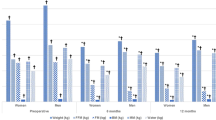
This study examined the effect of weight loss after 3, 6 and 12 months of Roux-en-Y Gastric Bypass (RYGB) on energy intake and on several biomarkers of oxidative stress such as levels of vitamin C, beta-carotene, vitamin E (diet/blood), nitric oxide metabolites (NOx), myeloperoxidase (MPO), thiobarbituric acid reactive substances (TBARS), reduced glutathione (GSH) and activity of catalase (CAT).
SUBJECTS/METHODS:
Study with a control group (CG), assessed once, and a bariatric group (BG) assessed at the basal period as well as at 3, 6 and 12 months post-surgery; both groups were composed of 5 men and 31 women (n=36). Age was 38.7±9.4 and 39.6±9.2 years old and body mass index (BMI) was 22.2±2.1 and 47.6±9.1 kg/m2, respectively. The variance measure quoted was SEM.
RESULTS:
The body weight at 12 months was 35.8±1.0% (P<0.001) lower than that of the basal period. At the basal period BG showed higher levels of NOx (P=0.007) and TBARS (P<0.001) and lower levels of vitamins C and E (P<0.001) compared with CG. After 3 months the activity of MPO was decreased (P<0.001). Six months after surgery GSH levels were decreased (P=0.037), whereas CAT activity was increased (P=0.029). After 12 months levels of NOx (P=0.004), TBARS (P<0.001), beta-carotene (P<0.001) and vitamin E (P<0.001) were decreased, whereas those of vitamin C (P<0.001) were increased compared with controls.
CONCLUSION:
RYGB followed by a daily vitamin supplement apparently attenuated pro-inflammatory and oxidative stress markers 1 year after surgery, but additional antioxidant supplementation appears necessary.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
WHO (World Health Organization). Obesity: Preventing and Managing the Global Epidemic. Report of a WHO consultation on obesity: Geneva, 2000, 894p.
Serra A, Granada ML, Romero R, Bayés B, Cantón A, Bonet J et al. The effect of bariatric surgery on adipocytokines, renal parameters and other cardiovascular risk factors in severe and very severe obesity: 1-year follow-up. Clin Nutr 2006; 25, 400–408.
Uzun H, Zengin K, Taskin M, Aydin S, Simsek G, Dariyerli N . Changes in leptin, plasminogen activator factor and oxidative stress in morbidly obese patients following open and laparoscopic Swedish adjustable gastric banding. Obes Surg 2004; 14, 659–665.
Higdon JV, Frei B . Obesity and oxidative stress: a direct link to CVD? Arterioscler Thromb Vasc Biol 2003; 23, 365–367.
Choi JW, Pai SH, Kim SK . Increases in nitric oxide concentrations correlate strongly with body fat in obese humans. Clin Chem 2001; 47, 1106–1109.
Vincent HK, Taylor AG . Biomarkers and potential mechanisms of obesity-induced oxidant stress in humans. Int J Obes 2006; 30, 400–418.
Olusi SO . Obesity is an independent risk factor for plasma lipid peroxidation and depletion of erythrocyte cytoprotective enzymes in humans. Int J Obes Relat Metab Disord 2002; 26, 1159–1164.
Kisakol G, Guney E, Bayraktar F, Yilmaz C, Kabalak T, Ozmen D . Effect of surgical weight loss on free radical and antioxidant balance: a preliminary report. Obes Surg 2002; 12, 795–800.
Viroonudomphol D, Pongpaew P, Tungtrongchitr R, Changbumrung S, Tungtrongchitr A, Phonrat B et al. The relationships between anthropometric measurements, serum vitamin A and E concentrations and lipid profiles in overweight and obese subjects. Asia Pac J Clin Nutr 2003; 12, 73–79.
Brown LA, Kerr CJ, Whiting P, Finer N, McEneny J, Ashton T . Oxidant stress in healthy normal-weight, overweight, and obese individuals. Obesity 2009; 17, 460–466.
Suzuki K, Inoue T, Hioki R, Ochiai J, Kusuhara Y, Ichino N et al. Association of abdominal obesity with decreased serum levels of carotenoids in a healthy Japanese population. Clin Nutr 2006; 25, 780–789.
World Medical Association. Declaration of Helsinki 2008, Seoul, 59th. General Assembly-WMA, viewed 15 May 2011. http://www.wma.net/en/30publications/10policies/b3/index.html.
WHO (World Health Organization). Physical Status: the Use and Interpretation of Anthropometry. WHO technical report series 854. WHO: Geneva, 1995, 453p.
Sichieri R, Everhart JE . Validity of a Brazilian frequency questionnaire against dietary recalls and estimated energy intake. Nutr Res 2003; 18, 1649–1659.
Willet W, Stampfer MJ . Total energy intake: implications for epidemiologic analyses. Am J Epidemiology 1986; 124, 17–27.
United States Department of Agriculture (USDA). Available at http://www.nal.usda.gov/fnic/foodcomp/search/. (accessed on 6 June 2008).
Bessey OA . Ascorbic acid microchemical methods. In: Bessey OA (ed). Vitamin Methods. Academic Press: New York 1960, p 303.
Arnaud J, Fortis I, Blachier S, Kia D, Favier A . Simultaneous determination of retinol, alpha-tocopherol and beta-carotene in serum by isocratic high-performance liquid chromatography. J Chromatogr 1991; 572, 103–116.
Nagaya T, Nakaya K, Yoshida I, Okamoto Y . Comparison of indices for serum vitamin E status in healthy subjects. Clin Chim Acta 1998; 276, 103–108.
Beutler E, Duron O, Kelly BM . Improved method for the determination of blood glutathione. J Lab Clin Med 1963; 61, 882–888.
Aebi H . Catalase in vitro. Methods Enzymol 1984; 105, 121–126.
Rao TS, Currie JL, Shaffer AF, Isakson PC . Comparative evaluation of arachidonic acid (AA)- and tetradecanoylphorbol acetate (TPA)- induced dermal inflammation. Inflammation 1993; 17, 723–741.
Green LC, Wagner DA, Glogowski J, Skipper PL, Wishnok JS, Tannenbaum SR . Analysis of nitrate, nitrite, and [15N] nitrate in biological fluids. Anal Biochem 1982; 126, 131–138.
Ohkawa H, Ohishi N, Yagi K . Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 1979; 95, 351–358.
Bird RP, Draper HH . Comparative studies on different methods of malondialdehyde determination. Methods Enzymol 1984; 105, 299–305.
van Dielen FM, Buurman WA, Hadfoune M, Nijhuis J, Greve JW . Macrophage inhibitory factor, plasminogen activator inhibitor-1, other acute phase proteins, and inflammatory mediators normalize as a result of weight loss in morbidly obese subjects treated with gastric restrictive surgery. J Clin Endocrinol Metab 2004; 89, 4062–4068.
Riess KP, Farnen JP, Lambert PJ, Mathiason MA, Kothari SN . Ascorbic acid deficiency in bariatric surgical population. Surg Obes Relat Dis 2009; 5, 81–86.
Bernet CP, Ciangura C, Coupaye M, Czernichow S, Bouillot JL, Basdevant A . Nutritional deficiency after gastric bypass: diagnosis, prevention and treatment. Diabetes Metab 2007; 33, 13–24.
Young DS . Implementation of SI units for clinical laboratory data. Style specifications and conversion tables. Ann Intern Med 1987; 106, 114–129.
Padayatt SJ, Katz A, Wang Y, Eck P, kwon O, Lee J et al. Vitamin C as an antioxidant: evaluation of its role in disease prevention. J Am Coll Nutr 2003; 22, 18–35.
Granado-Lorencio F, Herrero-Barbudo C, Olmedilla-Alonso B, Blanco-Navarro I, Pérez-Sacristán B . Hypocarotenemia after bariatric surgery: a preliminary study. Obes Surg 2008; 19, 879–882.
Clements RH, Katasani VG, Palepu R, Leeth RR, Leath TD, Roy BP et al. Incidence of vitamin deficiency after laparoscopic Roux-en-Y gastric bypass in a university hospital setting. Am Surg 2006; 72, 1196–1202.
de Luis DA, Pacheco D, Izaola O, Terroba MC, Cuellar L, Martin T . Clinical results and nutritional consequences of biliopancreatic diversion: Three years of follow-up. Ann Nutr Metab 2008; 53, 234–239.
Decker GA, Swain JM, Crowell MD, Scolapio JS . Gastrointestinal and nutritional complications after bariatric surgery. Am J Gastroenterol 2007; 102, 2571–2580.
Strohmayer E, Via MA, Yanagisawa R . Metabolic management following bariatric surgery. Mt Sinai J Med 2010; 77, 431–445.
Scopinaro N, Marinari GM, Pretolesi F, Papadia F, Murelli F, Marini P et al. Energy and nitrogen absorption after biliopancreatic diversion. Obes Surg 2000; 10, 436–441.
Traber MG, Arai H . Molecular mechanisms of vitamin E transport. Annu Rev Nutr 1999; 19, 343–355.
Boesing F, Moreira EA, Wilhelm-Filho D, Vigil SV, Parizottto EB, Inácio DB et al. Roux-en-Y bypass gastroplasty: markers of oxidative stress six months after surgery. Obes Surg 2010; 20, 1236–1244.
Cabrera EJ, Valezi AC, Delfino VDA, Lavado EL, Barbosa DS . Reduction in plasma levels of inflammatory and oxidative stress indicators after Roux-en-Y gastric bypass. Obes Surg 2010; 20, 42–49.
Faber P, Johnstone AM, Gibney ER, Elia M, Stubbs RJ, Duthie GG et al. The effect of rate of weight loss on erythrocyte glutathione concentration and synthesis in healthy obese men. Clin Sci 2002; 102, 569–577.
Sigfrid LA, Cunningham JM, Beeharry N, Lortz S, Tiedge M, Lenzen S et al. Cytokines and nitric oxide inhibit the enzyme activity of catalase but not its protein or mRNA expression in insulin-producing cells. J Mol Endoc 2003; 31, 509–518.
Kirkman HN, Gaetani GF . Mammalian catalase: a venerable enzyme with new mysteries. Trends Biochem Sc 2007; 32, 44–50.
Okuno Y, Matsuda M, Kobayashi H, Morita K, Suzuki E, Fukuhara A et al. Adipose expression of catalase is regulated via a novel remote PPARgamma-responsive region. Biochem Biophys Res Commun 2008; 366, 698–704.
Murri M, García-Fuentes E, García-Almenida JM, Garrido-Sánchez L, Mayas MD, Bernal R et al. Changes in oxidative stress and insulin resistance in morbidly obese patients after bariatric surgery. Obes Surg 2010; 20, 363–368.
Roberts CK, Won D, Pruthi S, Kurtovic S, Sindhu RK, Vaziri ND et al. Effect of a short-term diet and exercise intervention on oxidative stress, inflammation, MMP-9, and monocyte chemotactic activity in men with metabolic syndrome factors. J Appl Physiol 2006; 100, 1657–1665.
Elgazar-Carmon V, Rudich A, Hadad N, Levy R . Neutrophils transiently infiltrate intra-abdominal fat early in the course of high-fat feeding. J Lipid Res 2008; 49, 1894–1903.
Olszanecka-Glinianowicz M, Zahorska-Markiewicz B, Janowska J, Zurakowski A . Serum concentrations of nitric oxide, tumor necrosis factor (tnf-alpha) and tnf soluble receptors in women with overweight and obesity. Metabolism 2004; 53, 1268–1273.
Lin LY, Lee WJ, Shen HN, Yang WS, Pai NH, Su TC et al. Nitric oxide production is paradoxically decreased after weight reduction surgery in morbid obesity patients. Atherosclerosis 2007; 190, 436–442.
Xu J, Yunshi Z, Li R . Immunonutrition in surgical patients. Curr Drug Targets 2009; 10, 771–777.
Kücükakin B, Gögenur I, Reiter RJ, Rosenberg J . Oxidative stress in relation to surgery: is there a role for the antioxidant melatonin? J Surg Res 2009; 152, 338–347.
Acknowledgements
We are grateful to the following for financial support: Fundação de Apoio à Pesquisa Científica e Tecnológica do Estado de Santa Catarina (FAPESC): Grant #14191/2007–7, Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq)–Bolsa Produtividade and Programa Institucional de Bolsa de Iniciação Científica (PIBIC), Programa de Pós-Graduação em Nutrição da Universidade Federal de Santa Catarina, Programa de Fomento à Pós-Graduação (PROF)/Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Contributors: EAMM had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Study concept and design: EAMM. Acquisition of data: VRGS, JXM. Analysis and interpretation of data: VRGS, JB, SVGV, AMBM, TRG, MSSM, HV, EAMM, DWF. Drafting of the manuscript: VRGS, EAMM, DWF. Critical revision of the manuscript for important intellectual content: EAMM, DWF, TSF. Statistical analysis: VRGS, EAMM. Final approval of the version to be submitted: EAMM, DWF. Obtained funding: EAMM, DWF, TSF.
Rights and permissions
About this article
Cite this article
da Silva, V., Moreira, E., Wilhelm-Filho, D. et al. Proinflammatory and oxidative stress markers in patients submitted to Roux-en-Y gastric bypass after 1 year of follow-up. Eur J Clin Nutr 66, 891–899 (2012). https://doi.org/10.1038/ejcn.2012.17
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ejcn.2012.17
Keywords
This article is cited by
-
SMP30-mediated synthesis of vitamin C activates the liver PPARα/FGF21 axis to regulate thermogenesis in mice
Experimental & Molecular Medicine (2022)
-
Ascorbic acid inhibits visceral obesity and nonalcoholic fatty liver disease by activating peroxisome proliferator-activated receptor α in high-fat-diet-fed C57BL/6J mice
International Journal of Obesity (2019)
-
The Effects of Bariatric Surgery-Induced Weight Loss on Adipose Tissue in Morbidly Obese Women Depends on the Initial Metabolic Status
Obesity Surgery (2016)
-
Nitric Oxide, Oxidant Status and Antioxidant Response in Morbidly Obese Patients: the Impact of 1-Year Surgical Weight Loss
Obesity Surgery (2013)