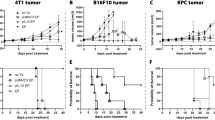
Abstract
Gene therapy with Plasmid AMEP (antiangiogenic metargidin peptide) has recently been studied as a potential targeted therapy for melanoma. This plasmid is designed to downregulate α5β1 and αvβ3 integrins. In our study, electroporation was used as a nonviral delivery system. We investigated the antiangiogenic and direct antitumor effectiveness of this gene therapy on low and highly metastatic B16 melanoma variants. In vitro, the antiangiogenic effectiveness as determined by tube formation assay on endothelial cells was predominantly dependent on AMEP expression levels. In vivo, antitumor effectiveness was mediated by the inhibition of proliferation, migration and invasion of melanoma cells and correlated with the expression of integrins on tumor cells after intratumor delivery. In addition, reduced metastatic potential was shown. Intramuscular gene electrotransfer of Plasmid AMEP, for AMEP systemic distribution, had no antitumor effect with this specific preventive treatment protocol, confirming that direct tumor delivery was more effective. This study confirms our previous in vitro data that the expression levels of integrins on melanoma cells could be used as a biomarker for antitumor effectiveness in integrin-targeted therapies, whereas the expression levels of AMEP peptide could be a predictive factor for antiangiogenic effectiveness of Plasmid AMEP in the treatment of melanoma.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout










Similar content being viewed by others
References
Heller LC, Heller R . Electroporation gene therapy preclinical and clinical trials for melanoma. Curr Gene Ther 2010; 10: 312–317.
Daud AI, DeConti RC, Andrews S, Urbas P, Riker AI, Sondak VK et al. Phase I trial of interleukin-12 plasmid electroporation in patients with metastatic melanoma. J Clin Oncol 2008; 26: 5896–5903.
Cemazar M, Jarm T, Sersa G . Cancer electrogene therapy with interleukin-12. Curr Gene Ther 2010; 10: 300–311.
Mizejewski GJ . Role of integrins in cancer: survey of expression patterns. Proc Soc Exp Biol Med 1999; 222: 124–138.
Trochon-Joseph V, Martel-Renoir D, Mir LM, Thomaidis A, Opolon P, Connault E et al. Evidence of antiangiogenic and antimetastatic activities of the recombinant disintegrin domain of metargidin. Cancer Res 2004; 64: 2062–2069.
Daugimont L, Vandermeulen G, Defresne F, Bouzin C, Mir LM, Bouquet C et al. Antitumoral and antimetastatic effect of antiangiogenic plasmids in B16 melanoma: higher efficiency of the recombinant disintegrin domain of ADAM 15. Eur J Pharm Biopharm 2011; 78: 314–319.
Bosnjak M, Prosen L, Dolinsek T, Blagus T, Markelc B, Cemazar M et al. Biological properties of melanoma and endothelial cells after plasmid AMEP gene electrotransfer depend on integrin quantity on cells. J Membr Biol 2013; 246: 803–819.
Spanggaard I, Snoj M, Cavalcanti A, Bouquet C, Sersa G, Robert C et al. Gene electrotransfer of plasmid antiangiogenic metargidin peptide (AMEP) in disseminated melanoma: safety and efficacy results of a phase I first-in-man study. Hum Gene Ther Clin Dev 2013; 24: 99–107.
Tesic N, Cemazar M . In vitro targeted gene electrotransfer to endothelial cells with plasmid DNA containing human endothelin-1 promoter. J Membr Biol 2013; 246: 783–791.
Bosnjak M, Lorente BC, Pogacar Z, Makovsek V, Cemazar M . Different incubation times of cells after gene electrotransfer in fetal bovine serum affect cell viability, but not transfection efficiency. J Membr Biol 2014; 247: 421–428.
Tevz G, Kranjc S, Cemazar M, Kamensek U, Coer A, Krzan M et al. Controlled systemic release of interleukin-12 after gene electrotransfer to muscle for cancer gene therapy alone or in combination with ionizing radiation in murine sarcomas. J Gene Med 2009; 11: 1125–1137.
Mir LM, Banoun H, Paoletti C . Introduction of definite amounts of nonpermeant molecules into living cells after electropermeabilization: direct access to the cytosol. Exp Cell Res 1988; 175: 15–25.
Mir LM, Orlowski S, Belehradek J Jr., Paoletti C . Electrochemotherapy potentiation of antitumour effect of bleomycin by local electric pulses. Eur J Cancer 1991; 27: 68–72.
Sersa G, Cemazar M, Miklavcic D . Antitumor effectiveness of electrochemotherapy with cis-diamminedichloroplatinum(II) in mice. Cancer Res 1995; 55: 3450–3455.
Cemazar M, Miklavcic D, Sersa G . Intrinsic sensitivity of tumor cells to bleomycin as an indicator of tumor response to electrochemotherapy. Jpn J Cancer Res 1998; 89: 328–333.
Jaroszeski MJ, Dang V, Pottinger C, Hickey J, Gilbert R, Heller R . Toxicity of anticancer agents mediated by electroporation in vitro. Anticancer Drugs 2000; 11: 201–208.
Sersa G, Miklavcic D . Electrochemotherapy of tumours. J Vis Exp 2008; 15: 1038.
Sedlar A, Dolinsek T, Markelc B, Prosen L, Kranjc S, Bosnjak M et al. Potentiation of electrochemotherapy by intramuscular IL-12 gene electrotransfer in murine sarcoma and carcinoma with different immunogenicity. Radiol Oncol 2012; 46: 302–311.
Teissie J, Escoffre JM, Paganin A, Chabot S, Bellard E, Wasungu L et al. Drug delivery by electropulsation: Recent developments in oncology. Int J Pharm 2012; 423: 3–6.
Edhemovic I, Brecelj E, Gasljevic G, Marolt Music M, Gorjup V, Mali B et al. Intraoperative electrochemotherapy of colorectal liver metastases. J Surg Oncol 2014; 110: 320–327.
Miklavcic D, Mali B, Kos B, Heller R, Sersa G . Electrochemotherapy: from the drawing board into medical practice. Biomed Eng Online 2014; 13: 29.
Mali B, Miklavcic D, Campana LG, Cemazar M, Sersa G, Snoj M et al. Tumor size and effectiveness of electrochemotherapy. Radiol Oncol 2013; 47: 32–41.
Rols MP, Delteil C, Golzio M, Dumond P, Cros S, Teissie J . In vivo electrically mediated protein and gene transfer in murine melanoma. Nat Biotechnol 1998; 16: 168–171.
Jaroszeski MJ, Gilbert R, Nicolau C, Heller R . In vivo gene delivery by electroporation. Adv Drug Deliv Rev 1999; 35: 131–137.
Golzio M, Rols MP, Teissie J . In vitro and in vivo electric field-mediated permeabilization, gene transfer, and expression. Methods 2004; 33: 126–135.
Vidic S, Markelc B, Sersa G, Coer A, Kamensek U, Tevz G et al. MicroRNAs targeting mutant K-ras by electrotransfer inhibit human colorectal adenocarcinoma cell growth in vitro and in vivo. Cancer Gene Ther 2010; 17: 409–419.
Chabot S, Pelofy S, Paganin-Gioanni A, Teissie J, Golzio M . Electrotransfer of RNAi-based oligonucleotides for oncology. Anticancer Res 2011; 31: 4083–4089.
Dolinsek T, Markelc B, Sersa G, Coer A, Stimac M, Lavrencak J et al. Multiple delivery of siRNA against endoglin into murine mammary adenocarcinoma prevents angiogenesis and delays tumor growth. PLoS One 2013; 8: e58723.
Kamensek U, Sersa G, Cemazar M . Evaluation of p21 promoter for interleukin 12 radiation induced transcriptional targeting in a mouse tumor model. Mol Cancer 2013; 12: 136.
Prosen L, Markelc B, Dolinsek T, Music B, Cemazar M, Sersa G . Mcam silencing with RNA interference using magnetofection has antitumor effect in murine melanoma. Mol Ther Nucleic Acids 2014; 3: e205.
Crokart N, Danhier F, Daugimont L, Goncalves N, Jordan BF, Gregoire V et al. Potentiation of radiotherapy by a localized antiangiogenic gene therapy. Radiother Oncol 2013; 107: 252–258.
Robinson SD, Hodivala-Dilke KM . The role of beta3-integrins in tumor angiogenesis: context is everything. Curr Opin Cell Biol 2011; 23: 630–637.
Niemisto A, Dunmire V, Yli-Harja O, Zhang W, Shmulevich I . Robust quantification of in vitro angiogenesis through image analysis. IEEE Trans Med Imaging 2005; 24: 549–553.
Livak KJ, Schmittgen TD . Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001; 25: 402–408.
Acknowledgements
We acknowledge the financial support from the state budget by the Slovenian Research Agency (programs no. P3-0003 and P1-0140 and projects no. J3-4211, J3-4259). The research was conducted in the scope of LEA EBAM (French-Slovenian European Associated Laboratory: Pulsed Electric Fields Applications in Biology and Medicine) and is a result of networking efforts within COST TD1104 Action. We thank Mira Lavric, Ursa Lampreht and Natasa Tesic (Institute of Oncology Ljubljana, Ljubljana, Slovenia) and Miha Butinar (Jozef Stefan Institute) for all the valuable work they contributed to this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
CB is a stockholder and an employee at ONXEO. The remaining authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on Gene Therapy website
Supplementary information
Rights and permissions
About this article
Cite this article
Bosnjak, M., Dolinsek, T., Cemazar, M. et al. Gene electrotransfer of plasmid AMEP, an integrin-targeted therapy, has antitumor and antiangiogenic action in murine B16 melanoma. Gene Ther 22, 578–590 (2015). https://doi.org/10.1038/gt.2015.26
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2015.26
This article is cited by
-
Experimental Study of Antitumor Activity of Pefagtal Addressed to αvβ3 Integrins
Bulletin of Experimental Biology and Medicine (2022)
-
Pre-clinical investigation of the synergy effect of interleukin-12 gene-electro-transfer during partially irreversible electropermeabilization against melanoma
Journal for ImmunoTherapy of Cancer (2019)
-
Cycloartane triterpenoid (23R, 24E)-23-acetoxymangiferonic acid inhibited proliferation and migration in B16-F10 melanoma via MITF downregulation caused by inhibition of both β-catenin and c-Raf–MEK1–ERK signaling axis
Journal of Natural Medicines (2019)
-
Antitumor in situ vaccination effect of TNFα and IL-12 plasmid DNA electrotransfer in a murine melanoma model
Cancer Immunology, Immunotherapy (2018)
-
Electrotransfer parameters as a tool for controlled and targeted gene expression in skin
Molecular Therapy - Nucleic Acids (2016)