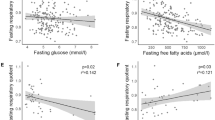
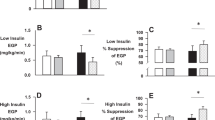
Abstract
Objective:
To determine insulin sensitivity and skeletal muscle fatty acid (FA) handling at baseline and after a high-fat mixed meal in impaired fasting glucose (IFG), impaired glucose tolerance (IGT), IFG/IGT and normal glucose tolerance (NGT) subjects.
Design:
In this multi-center study, insulin sensitivity and β-cell function were assessed (n=102), using a euglycemic-hyperinsulinemic and hyperglycemic clamp with additional arginine stimulation and a 75 g oral glucose tolerance test. Fasting and postprandial skeletal muscle FA handling was examined in a substudy using the forearm balance technique (n=35).
Subjects:
A total of 102 subjects with IFG (n=48), IGT (n=12), IFG/IGT (n=26) and NGT (n=16).
Results:
IFG, IGT and IFG/IGT subjects had lower insulin sensitivity with no differences between groups, and lower impaired β-cell function compared with NGT controls. The early postprandial increase in triacylglycerol (TAG) concentration was higher (iAUC0−2 h IFG: 238.4±26.5, IGT: 234.0±41.0 and NGT: 82.6±13.8 μmol l−1 min−1, both P<0.05) and early TAG extraction was increased (AUC0−2 h IFG: 56.8±9.0, IGT: 52.2±12.0 and NGT: 3.8±15.4 nmol·100 ml−1 min−1, P<0.05 and P=0.057, respectively) in both IFG and IGT subjects.
Conclusion:
IFG, IGT and IFG/IGT subjects have lower insulin sensitivity and impaired β-cell function compared with age- and BMI-matched NGT controls. The increased postprandial TAG response and higher muscle TAG extraction in both IFG and IGT compared with NGT may lead to ectopic fat accumulation in the skeletal muscle, thereby contributing to insulin resistance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Dinneen SF, Maldonado III D, Leibson CL, Klee GG, Li H, Melton III LJ et al. Effects of changing diagnostic criteria on the risk of developing diabetes. Diabetes Care 1998; 21: 1408–1413.
Meigs JB, Muller DC, Nathan DM, Blake DR, Andres R . The natural history of progression from normal glucose tolerance to type 2 diabetes in the Baltimore Longitudinal Study of Aging. Diabetes 2003; 52: 1475–1484.
Shaw JE, Zimmet PZ, de Courten M, Dowse GK, Chitson P, Gareeboo H et al. Impaired fasting glucose or impaired glucose tolerance. What best predicts future diabetes in Mauritius? Diabetes Care 1999; 22: 399–402.
Tirosh A, Shai I, Tekes-Manova D, Israeli E, Pereg D, Shochat T et al. Normal fasting plasma glucose levels and type 2 diabetes in young men. N Engl J Med 2005; 353: 1454–1462.
Faerch K, Borch-Johnsen K, Holst JJ, Vaag A . Pathophysiology and aetiology of impaired fasting glycaemia and impaired glucose tolerance: does it matter for prevention and treatment of type 2 diabetes? Diabetologia 2009; 52: 1714–1723.
The DECODE study group. European Diabetes Epidemiology Group. Glucose tolerance and mortality: comparison of WHO and American Diabetes Association diagnostic criteria. Diabetes epidemiology: collaborative analysis of diagnostic criteria in Europe. Lancet 1999; 354: 617–621.
Meigs JB, Nathan DM, D’Agostino Sr RB, Wilson PW . Fasting and postchallenge glycemia and cardiovascular disease risk: the Framingham Offspring Study. Diabetes Care 2002; 25: 1845–1850.
Abdul-Ghani MA, Jenkinson CP, Richardson DK, Tripathy D, DeFronzo RA . Insulin secretion and action in subjects with impaired fasting glucose and impaired glucose tolerance: results from the Veterans Administration Genetic Epidemiology Study. Diabetes 2006; 55: 1430–1435.
Bock G, Dalla Man C, Campioni M, Chittilapilly E, Basu R, Toffolo G et al. Pathogenesis of pre-diabetes: mechanisms of fasting and postprandial hyperglycemia in people with impaired fasting glucose and/or impaired glucose tolerance. Diabetes 2006; 55: 3536–3549.
Meyer C, Pimenta W, Woerle HJ, Van Haeften T, Szoke E, Mitrakou A et al. Different mechanisms for impaired fasting glucose and impaired postprandial glucose tolerance in humans. Diabetes Care 2006; 29: 1909–1914.
Faerch K, Vaag A, Holst JJ, Glumer C, Pedersen O, Borch-Johnsen K . Impaired fasting glycaemia vs impaired glucose tolerance: similar impairment of pancreatic alpha and beta cell function but differential roles of incretin hormones and insulin action. Diabetologia 2008; 51: 853–861.
Tripathy D, Almgren P, Tuomi T, Groop L . Contribution of insulin-stimulated glucose uptake and basal hepatic insulin sensitivity to surrogate measures of insulin sensitivity. Diabetes Care 2004; 27: 2204–2210.
Wasada T, Kuroki H, Katsumori K, Arii H, Sato A, Aoki K . Who are more insulin resistant, people with IFG or people with IGT? Diabetologia 2004; 47: 758–759.
Weyer C, Bogardus C, Pratley RE . Metabolic characteristics of individuals with impaired fasting glucose and/or impaired glucose tolerance. Diabetes 1999; 48: 2197–2203.
Abdul-Ghani MA, Sabbah M, Kher J, Minuchin O, Vardi P, Raz I . Different contributions of insulin resistance and beta-cell dysfunction in overweight Israeli Arabs with IFG and IGT. Diabetes Metab Res Rev 2006; 22: 126–130.
Faerch K, Vaag A, Holst JJ, Hansen T, Jorgensen T, Borch-Johnsen K . Natural history of insulin sensitivity and insulin secretion in the progression from normal glucose tolerance to impaired fasting glycemia and impaired glucose tolerance: the Inter99 study. Diabetes Care 2009; 32: 439–444.
Novoa FJ, Boronat M, Saavedra P, Diaz-Cremades JM, Varillas VF, La Roche F et al. Differences in cardiovascular risk factors, insulin resistance, and insulin secretion in individuals with normal glucose tolerance and in subjects with impaired glucose regulation: the Telde Study. Diabetes Care 2005; 28: 2388–2393.
Corpeleijn E, Saris WH, Blaak EE . Metabolic flexibility in the development of insulin resistance and type 2 diabetes: effects of lifestyle. Obes Rev 2009; 10: 178–193.
Goossens GH . The role of adipose tissue dysfunction in the pathogenesis of obesity-related insulin resistance. Physiol Behav 2008; 94: 206–218.
Krssak M, Falk Petersen K, Dresner A, DiPietro L, Vogel SM, Rothman DL et al. Intramyocellular lipid concentrations are correlated with insulin sensitivity in humans: a 1H NMR spectroscopy study. Diabetologia 1999; 42: 113–116.
Sinha R, Dufour S, Petersen KF, LeBon V, Enoksson S, Ma YZ et al. Assessment of skeletal muscle triglyceride content by (1)H nuclear magnetic resonance spectroscopy in lean and obese adolescents: relationships to insulin sensitivity, total body fat, and central adiposity. Diabetes 2002; 51: 1022–1027.
Blaak EE, van Aggel-Leijssen DP, Wagenmakers AJ, Saris WH, van Baak MA . Impaired oxidation of plasma-derived fatty acids in type 2 diabetic subjects during moderate-intensity exercise. Diabetes 2000; 49: 2102–2107.
Blaak EE, Wagenmakers AJ, Glatz JF, Wolffenbuttel BH, Kemerink GJ, Langenberg CJ et al. Plasma FFA utilization and fatty acid-binding protein content are diminished in type 2 diabetic muscle. Am J Physiol Endocrinol Metab 2000; 279: E146–E154.
Mensink M, Blaak EE, van Baak MA, Wagenmakers AJ, Saris WH . Plasma free fatty acid uptake and oxidation are already diminished in subjects at high risk for developing type 2 diabetes. Diabetes 2001; 50: 2548–2554.
DeFronzo RA, Tobin JD, Andres R . Glucose clamp technique: a method for quantifying insulin secretion and resistance. Am J Physiol 1979; 237: E214–E223.
Bunck MC, Diamant M, Corner A, Eliasson B, Malloy JL, Shaginian RM et al. One-year treatment with exenatide improves beta-cell function, compared with insulin glargine, in metformin-treated type 2 diabetic patients: a randomized, controlled trial. Diabetes Care 2009; 32: 762–768.
Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC . Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985; 28: 412–419.
Abdul-Ghani MA, Tripathy D, DeFronzo RA . Contributions of beta-cell dysfunction and insulin resistance to the pathogenesis of impaired glucose tolerance and impaired fasting glucose. Diabetes Care 2006; 29: 1130–1139.
Mari A, Pacini G, Murphy E, Ludvik B, Nolan JJ . A model-based method for assessing insulin sensitivity from the oral glucose tolerance test. Diabetes Care 2001; 24: 539–548.
Carnevale Schianca GP, Rossi A, Sainaghi PP, Maduli E, Bartoli E . The significance of impaired fasting glucose versus impaired glucose tolerance: importance of insulin secretion and resistance. Diabetes Care 2003; 26: 1333–1337.
Kim SH, Reaven GM . Isolated impaired fasting glucose and peripheral insulin sensitivity: not a simple relationship. Diabetes Care 2008; 31: 347–352.
Festa A, D’Agostino Jr R, Hanley AJ, Karter AJ, Saad MF, Haffner SM . Differences in insulin resistance in nondiabetic subjects with isolated impaired glucose tolerance or isolated impaired fasting glucose. Diabetes 2004; 53: 1549–1555.
Laakso M, Zilinskaite J, Hansen T, Boesgaard TW, Vanttinen M, Stancakova A et al. Insulin sensitivity, insulin release and glucagon-like peptide-1 levels in persons with impaired fasting glucose and/or impaired glucose tolerance in the EUGENE2 study. Diabetologia 2008; 51: 502–511.
Dagogo-Jack S, Askari H, Tykodi G . Glucoregulatory physiology in subjects with low-normal, high-normal, or impaired fasting glucose. J Clin Endocrinol Metab 2009; 94: 2031–2036.
van Hees AM, Jans A, Hul GB, Roche HM, Saris WH, Blaak EE . Skeletal muscle fatty acid handling in insulin resistant men. Obesity (Silver Spring) 2011; e-pub ahead of print 17 February 2011; doi:10.1038/oby.2011.10.
Corpeleijn E, Pelsers MM, Soenen S, Mensink M, Bouwman FG, Kooi ME et al. Insulin acutely upregulates protein expression of the fatty acid transporter CD36 in human skeletal muscle in vivo. J Physiol Pharmacol 2008; 59: 77–83.
Cali AM, Bonadonna RC, Trombetta M, Weiss R, Caprio S . Metabolic abnormalities underlying the different prediabetic phenotypes in obese adolescents. J Clin Endocrinol Metab 2008; 93: 1767–1773.
Acknowledgements
The authors would like to thank Anneke Jans and Ellen Konings for their excellent practical support, and Jos Stegen and Wendy Sluijsmans for technical assistance. An Investigator-Initiated Study Grant from Novartis Pharma supported this study. Clinical Trial Registration Number: ISRCTN42786336
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Moors, C., van der Zijl, N., Diamant, M. et al. Impaired insulin sensitivity is accompanied by disturbances in skeletal muscle fatty acid handling in subjects with impaired glucose metabolism. Int J Obes 36, 709–717 (2012). https://doi.org/10.1038/ijo.2011.123
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2011.123
Keywords
This article is cited by
-
Tuning fatty acid oxidation in skeletal muscle with dietary fat and exercise
Nature Reviews Endocrinology (2020)
-
Altered skeletal muscle fatty acid handling is associated with the degree of insulin resistance in overweight and obese humans
Diabetologia (2016)
-
Obesity-related insulin resistance: implications for the surgical patient
International Journal of Obesity (2015)
-
Comprehensive Biomarker Testing of Glycemia, Insulin Resistance, and Beta Cell Function Has Greater Sensitivity to Detect Diabetes Risk Than Fasting Glucose and HbA1c and Is Associated with Improved Glycemic Control in Clinical Practice
Journal of Cardiovascular Translational Research (2014)
-
In vivo postprandial lipid partitioning in liver and skeletal muscle in prediabetic and diabetic rats
Diabetologia (2013)