Abstract
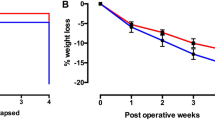
This study investigated the anatomical integrity of the vagal innervation to the gastrointestinal tract following Roux-en-Y gastric bypass (RYGB) in the mouse. Specifically, the surgical procedure was performed in high-fat-fed reporter mice (Phox2b-Cre-tdTomato), in which the entire vagal innervation of the gastrointestinal tract was fluorescently labeled. As a result, our anatomical observations revealed both qualitative and quantitative changes of the vagal supply to the gut after RYGB. This included the extensive denervation of the glandular and distal stomach, and sites of surgical interventions (clipping and anastomosis). Furthermore, the stomach wall after RYGB frequently contained dystrophic axons and endings, suggestive of vagal neurodegeneration. In contrast, RYGB did not significantly modify the innervation to the rest of the intestines and glucostatic organs. In summary, the present study describes a previously unrecognized pattern of vagal remodeling and denervation following RYGB. Our findings may serve as a guideline for future investigations on the role of gut–brain communication in bariatric surgery.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Buchwald H, Williams SE . Bariatric surgery worldwide 2003. Obes Surg 2004; 14: 1157–1164.
Bueter M, Lowenstein C, Ashrafian H, Hillebrand J, Bloom SR, Olbers T et al. Vagal sparing surgical technique but not stoma size affects body weight loss in rodent model of gastric bypass. Obes Surg 2010; 20: 616–622.
Troy S, Soty M, Ribeiro L, Laval L, Migrenne S, Fioramonti X et al. Intestinal gluconeogenesis is a key factor for early metabolic changes after gastric bypass but not after gastric lap-band in mice. Cell Metab 2008; 8: 201–211.
Breen DM, Rasmussen BA, Kokorovic A, Wang R, Cheung GW, Lam TK . Jejunal nutrient sensing is required for duodenal-jejunal bypass surgery to rapidly lower glucose concentrations in uncontrolled diabetes. Nat Med 2012; 18: 950–955.
Zechner JF, Mirshahi UL, Satapati S, Berglund ED, Rossi J, Scott MM et al. Weight-independent effects of Roux-en-Y gastric bypass on glucose homeostasis via melanocortin-4 receptors in mice and humans. Gastroenterology 2012; 144: 580–590.
Yamada H, Kojima K, Inokuchi M, Kawano T, Sugihara K . Efficacy of celiac branch preservation in Roux-en-y reconstruction after laparoscopy-assisted distal gastrectomy. Surgery 2011; 149: 22–28.
Berthoud HR, Shin AC, Zheng H . Obesity surgery and gut-brain communication. Physiol Behav 2011; 105: 106–119.
Nestoridi E, Kvas S, Kucharczyk J, Stylopoulos N. . Resting energy expenditure and energetic cost of feeding are augmented after Roux-en-Y gastric bypass in obese mice. Endocrinology 2012; 153: 2234–2244.
Scott MM, Williams KW, Rossi J, Lee CE, Elmquist JK. . Leptin receptor expression in hindbrain Glp-1 neurons regulates food intake and energy balance in mice. J Clin Invest 2011; 121: 2413–2421.
Wang FB, Powley TL . Vagal innervation of intestines: afferent pathways mapped with new en bloc horseradish peroxidase adaptation. Cell Tissue Res 2007; 329: 221–230.
Berthoud HR . The vagus nerve, food intake and obesity. Regul Pept 2008; 149: 15–25.
Ardila-Hani A, Soffer EE . Review article: the impact of bariatric surgery on gastrointestinal motility. Aliment Pharmacol Ther 2011; 34: 825–831.
Zhang T, Uchida T, Gomez G, Lluis F, Thompson JC, Greeley GH Jr . Neural regulation of peptide YY secretion. Regul Pept 1993; 48: 321–328.
Mizrahi M, Ya'acov AB, Ilan Y . Gastric stimulation for weight loss. World J Gastroenterol 2012; 18: 2309–2319.
Kral JG . Vagotomy for treatment of severe obesity. Lancet 1978; 1: 307–308.
Shin AC, Zheng H, Berthoud HR . Vagal innervation of the hepatic portal vein and liver is not necessary for Roux-en-Y gastric bypass surgery-induced hypophagia, weight loss, and hypermetabolism. Ann Surg 2012; 255: 294–301.
Acknowledgements
We are grateful to Matthew Harper for his technical assistance and Joel K Elmquist (UT Southwestern Medical Center at Dallas) for his comments on the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Gautron, L., Zechner, J. & Aguirre, V. Vagal innervation patterns following Roux-en-Y gastric bypass in the mouse. Int J Obes 37, 1603–1607 (2013). https://doi.org/10.1038/ijo.2013.48
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2013.48
Keywords
This article is cited by
-
Novel Insights into the Physiology of Nutrient Sensing and Gut-Brain Communication in Surgical and Experimental Obesity Therapy
Obesity Surgery (2023)
-
Shortened-Interval Dual-Session EDGE Reduces the Risk of LAMS Dislodgement While Facilitating Timely ERCP
Digestive Diseases and Sciences (2021)
-
Surgical Mouse Models of Vertical Sleeve Gastrectomy and Roux-en Y Gastric Bypass: a Review
Obesity Surgery (2019)
-
Gut vagal sensory signaling regulates hippocampus function through multi-order pathways
Nature Communications (2018)
-
Signalling from the periphery to the brain that regulates energy homeostasis
Nature Reviews Neuroscience (2018)