Abstract
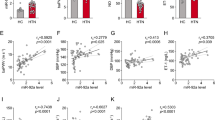
Vascular smooth muscle cell (VSMC) phenotypic plasticity has a critical role in the pathophysiology of arterial remodeling in essential hypertension. MicroRNAs are emerging as potential biomarkers and therapeutic targets in cardiovascular disease. We assessed the expression levels of the microRNAs miR-143, miR-145, miR-21, miR-133 and miR-1, which are implicated in VSMC phenotypic modulation, in 60 patients with essential hypertension and 29 healthy individuals. All patients underwent 24-h ambulatory blood pressure (BP) monitoring. MicroRNA levels in peripheral blood mononuclear cells were quantified by real-time reverse transcription polymerase chain reaction. Hypertensive patients showed lower miR-143 (2.20±0.25 versus 4.19±0.57, P<0.001), miR-145 (13.51±1.73 versus 22.38±3.31, P=0.010) and miR-133 (8.15±1.32 versus 37.03±8.18, P<0.001) and higher miR-21 (3.08±0.32 versus 2.06±0.31, P=0.048) and miR-1 (33.94±5.19 versus 12.35±2.13 P=0.006) expression levels compared with controls. In hypertensive patients, we observed correlations of miR-143 (r=−0.380, P=0.003), miR-145 (r=−0.405, P=0.001), miR-21 (r=−0.486, P<0.001) and miR-133 (r=0.479, P<0.001) expression levels with 24-h diastolic BP. Furthermore, we observed correlations of miR-21 (r=−0.291, P=0.024), miR-1 (r=−0.312, P=0.015) and miR-133 (r=0.310, P=0.016) levels with the dipping status. Associations of miR-143 (r=−0.292, P=0.025), miR-145 (r=−0.399, P=0.002), miR-21 (r=−0.343, P=0.008) and miR-133 (r=0.370, P=0.004) levels with 24-h mean pulse pressure were also found. Our data provide important evidence that VSMC-modulating microRNAs are closely related to essential hypertension in humans and they may represent potential therapeutic targets in essential hypertension.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Chobanian AV, Bakris GL, Black HR, Cushman WC, Green LA, Izzo JL et al. Seventh report of the Joint National Committee on prevention, detection, evaluation, and treatment of high blood pressure. Hypertension 2003; 42 (6): 1206–1252.
Small EM, Olson EN . Pervasive roles of microRNAs in cardiovascular biology. Nature 2011; 469 (7330): 336–342.
Abdellatif M . Differential expression of microRNAs in different disease states. Circ Res 2012; 110 (4): 638–650.
Boettger T, Braun T . A New level of complexity: the role of microRNAs in cardiovascular development. Circ Res 2012; 110 (7): 1000–1013.
Fiedler J, Thum T . MicroRNAs in myocardial infarction. Arterioscler Thromb Vasc Biol 2013; 33 (2): 201–205.
Da Costa Martins PA, De Windt LJ . MicroRNAs in control of cardiac hypertrophy. Cardiovasc Res 2012; 93 (4): 563–572.
Bauersachs J, Thum T . Biogenesis and regulation of cardiovascular microRNAs. Circ Res 2011; 109 (3): 334–347.
Topkara V, Mann D . Role of microRNAs in cardiac remodeling and heart failure. Cardiovasc Drugs Ther 2011; 25 (2): 171–182.
Thum T . MicroRNA therapeutics in cardiovascular medicine. EMBO Mol Med 2012; 4 (1): 3–14.
Heggermont WA, Heymans S . MicroRNAs are involved in end-organ damage during hypertension. Hypertension 2012; 60 (5): 1088–1093.
Batkai S, Thum T . MicroRNAs in hypertension: mechanisms and therapeutic targets. Curr Hypertens Rep 2012; 14 (1): 79–87.
Lacolley P, Regnault V, Nicoletti A, Li Z, Michel J-B . The vascular smooth muscle cell in arterial pathology: a cell that can take on multiple roles. Cardiovasc Res 2012; 95 (2): 194–204.
Feihl F, Liaudet L, Levy BI, Waeber B . Hypertension and microvascular remodelling. Cardiovasc Res 2008; 78 (2): 274–285.
Davis-Dusenbery BN, Wu C, Hata A . Micromanaging vascular smooth muscle cell differentiation and phenotypic modulation. Arterioscler Thromb Vasc Biol 2011; 31 (11): 2370–2377.
Cordes KR, Sheehy NT, White MP, Berry EC, Morton SU, Muth AN et al. miR-145 and miR-143 regulate smooth muscle cell fate and plasticity. Nature 2009; 460 (7256): 705–710.
Boettger T, Beetz N, Kostin S, Schneider J, Kruger M, Hein L et al. Acquisition of the contractile phenotype by murine arterial smooth muscle cells depends on the Mir143/145 gene cluster. J Clin Invest 2009; 119 (9): 2634–2647.
Xin M, Small EM, Sutherland LB, Qi X, McAnally J, Plato CF et al. MicroRNAs miR-143 and miR-145 modulate cytoskeletal dynamics and responsiveness of smooth muscle cells to injury. Genes Dev 2009; 23 (18): 2166–2178.
Elia L, Quintavalle M, Zhang J, Contu R, Cossu L, Latronico MV et al. The knockout of miR-143 and -145 alters smooth muscle cell maintenance and vascular homeostasis in mice: correlates with human disease. Cell Death Differ 2009; 16 (12): 1590–1598.
Rangrez AY, Massy ZA, Metzinger-Le Meuth V, Metzinger L . miR-143 and miR-145: molecular keys to switch the phenotype of vascular smooth muscle cells. Circ Cardiovasc Genet 2011; 4 (2): 197–205.
Ji R, Cheng Y, Yue J, Yang J, Liu X, Chen H et al. MicroRNA expression signature and antisense-mediated depletion reveal an essential role of microRNA in vascular neointimal lesion formation. Circ Res 2007; 100 (11): 1579–1588.
Torella D, Iaconetti C, Catalucci D, Ellison GM, Leone A, Waring CD et al. MicroRNA-133 controls vascular smooth muscle cell phenotypic switch in vitro and vascular remodeling in vivo. Circ Res 2011; 109 (8): 880–893.
Chen J, Yin H, Jiang Y, Radhakrishnan SK, Huang Z-P, Li J et al. Induction of microRNA-1 by myocardin in smooth muscle cells inhibits cell proliferation. Arterioscler Thromb Vasc Biol 2011; 31 (2): 368–375.
Mancia G, De Backer G, Dominiczak A, Cifkova R, Fagard R, Germano G et al. 2007 guidelines for the management of arterial hypertension: the task force for the management of arterial hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). J Hypertens 2007; 25 (6): 1105–1187.
Pickering TG, Shimbo D, Haas D . Ambulatory blood-pressure monitoring. N Engl J Med 2006; 354 (22): 2368–2374.
White WB, Maraka S . Is it possible to manage hypertension and evaluate therapy without ambulatory blood pressure monitoring? Curr Hypertens Rep 2012; 14 (4): 366–373.
Hodgkinson J, Mant J, Martin U, Guo B, Hobbs FD, Deeks JJ et al. Relative effectiveness of clinic and home blood pressure monitoring compared with ambulatory blood pressure monitoring in diagnosis of hypertension: systematic review. BMJ 2011; 342: d3621.
Hansen TW, Li Y, Boggia J, Thijs L, Richart T, Staessen JA . Predictive role of the nighttime blood pressure. Hypertension 2011; 57 (1): 3–10.
Yano Y, Kario K . Nocturnal blood pressure and cardiovascular disease: a review of recent advances. Hypertens Res 2012; 35 (7): 695–701.
Safar ME, Nilsson PM, Blacher J, Mimran A . Pulse pressure, arterial stiffness, and end-organ damage. Curr Hypertens Rep 2012; 14 (4): 339–344.
Mendell Joshua T, Olson Eric N . MicroRNAs in stress signaling and human disease. Cell 2012; 148 (6): 1172–1187.
Li S, Liang Z, Xu L, Zou F . MicroRNA-21: a ubiquitously expressed pro-survival factor in cancer and other diseases. Mol Cell Biochem 2012; 360 (1–2): 147–158.
O'Sullivan JF, Martin K, Caplice NM . Microribonucleic acids for prevention of plaque rupture and in-stent restenosis: "A Finger in the Dam". J Am Coll Cardiol 2011; 57 (4): 383–389.
Albinsson S, Sward K . Targeting smooth muscle microRNAs for therapeutic benefit in vascular disease. Pharmacol Res 2013; 75: 28–36.
Kontaraki JE, Marketou ME, Zacharis EA, Parthenakis FI, Vardas PE . Early cardiac gene transcript levels in peripheral blood mononuclear cells in patients with untreated essential hypertension. J Hypertens 2011; 29 (4): 791–797.
Cuspidi C, Meani S, Valerio C, Sala C, Fusi V, Masaidi M et al. Reproducibility of dipping/nondipping pattern in untreated essential hypertensive patients: impact of sex and age. Blood Press. Monit 2007; 12 (2): 101–106.
Ben-Dov IZ, Ben-Arieh L, Mekler J, Bursztyn M . Blood pressure dipping is reproducible in clinical practice. Blood Press Monit 2005; 10 (2): 79–84.
Fernández-Hernando C, Ramírez CM, Goedeke L, Suárez Y . MicroRNAs in Metabolic Disease. Arterioscler Thromb Vasc Biol 2013; 33 (2): 178–185.
Acknowledgements
The study was supported by the European Commission (EC) support program Translational Potential (TransPOT; EC contract number 285948).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Kontaraki, J., Marketou, M., Zacharis, E. et al. Differential expression of vascular smooth muscle-modulating microRNAs in human peripheral blood mononuclear cells: novel targets in essential hypertension. J Hum Hypertens 28, 510–516 (2014). https://doi.org/10.1038/jhh.2013.117
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jhh.2013.117
Keywords
This article is cited by
-
Arterial Stiffness and the Canonical WNT/β-catenin Pathway
Current Hypertension Reports (2022)
-
MitomiRs: new emerging microRNAs in mitochondrial dysfunction and cardiovascular disease
Hypertension Research (2020)
-
Screening of differentially expressed microRNAs of essential hypertension in Uyghur population
Lipids in Health and Disease (2019)
-
Epigenetic modification: a regulatory mechanism in essential hypertension
Hypertension Research (2019)
-
miR-1 induces endothelial dysfunction in rat pulmonary arteries
Journal of Physiology and Biochemistry (2019)