Abstract
Objective:
To determine the effects of sodium bicarbonate (NaHCO3) correction of metabolic acidosis on cardiopulmonary, laboratory, and cerebral, renal and splanchnic regional oxygen saturation (rSO2) and fractional tissue oxygen extraction (FTOE) in extremely premature neonates during the first postnatal week.
Study Design:
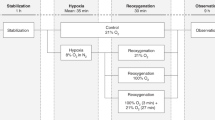
Observational cohort data were collected from 500 to 1250 g neonates who received NaHCO3 'half' corrections (0.3 * Weight (kg) * Base Deficit (mmol l−1)) for presumed renal losses.
Result:
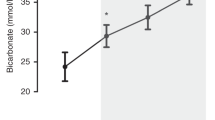
Twelve subjects with normal blood pressure and heart rate received 17 NaHCO3 corrections. Mean (±s.d.) gestational age was 27±2 week and birth weight was 912±157 g. NaHCO3 corrections provided a mean (±s.d.) 4.5±1.0 ml kg−1 fluid bolus, shifted mean (±s.d.) base deficit from 7.6±1.8 to 3.4±2.1 mmol l−1 (P<0.05), and increased median (±s.d.) pH from 7.23±0.06 to 7.31±0.05 (P<0.05). No significant changes in blood pressure, pulse oximetry, PCO2, lactate, sodium, blood urea nitrogen, creatinine or hematocrit were observed. Cerebral, renal and splanchnic rSO2 (74%, 66% and 44%, respectively, at baseline) and FTOE (0.21, 0.29 and 0.52, respectively, at baseline) were unchanged following NaHCO3 correction.
Conclusion:
NaHCO3 infusions decreased base deficits and increased pH though produced no discernible effects or benefits on cardiopulmonary parameters including rSO2 and FTOE. These findings warrant further prospective evaluation in larger populations with more significant metabolic acidosis to determine the utility of tissue oxygenation monitoring in differentiating metabolic acidosis due to oxygen delivery/consumption imbalance versus renal bicarbonate losses.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Ammari AN, Schulze KF . Uses and abuses of sodium bicarbonate in the neonatal intensive care unit. Curr Opin Pediatr 2002; 14: 151–156.
Aschner JL, Poland RL . Sodium bicarbonate: basically useless therapy. Pediatrics 2008; 122: 831–835.
Berg CS, Barnette AR, Myers BJ, Shimony MK, Barton AW, Inder TE . Sodium bicarbonate administration and outcome in preterm infants. J Pediatr 2010; 157: 684–687.
Bourchier D . Bicarbonate therapy for infants. J Pediatr 2011; 158: 518.
Kecskes ZB, Davies MW . Rapid correction of early metabolic acidaemia in comparison with placebo, no intervention or slow correction in LBW infants. Cochrane Database Syst Rev 2002; CD002976.
Lawn CJ, Weir FJ, McGuire W . Base administration or fluid bolus for preventing morbidity and mortality in preterm infants with metabolic acidosis. Cochrane Database Syst Rev 2005; CD003215.
Ramiro-Tolentino SB, Markarian K, Kleinman LI . Renal bicarbonate excretion in extremely low birth weight infants. Pediatrics 1996; 98: 256–261.
Sato T, Takahashi N, Komatsu Y, Wada M, Matsunaga M, Ito K et al. Urinary acidification in extremely low birth weight infants. Early Hum Dev 2002; 70: 15–24.
Johnson PJ . Sodium bicarbonate use in the treatment of acute neonatal lactic acidosis: benefit or harm? Neonatal Netw 2011; 30: 199–205.
Noori S, Wu TW, Seri I . pH effects on cardiac function and systemic vascular resistance in preterm infants. J Pediatr 2013; 162: 958–963.
Bonestroo HJ, Lemmers PM, Baerts W, van Bel F . Effect of antihypotensive treatment on cerebral oxygenation of preterm infants without PDA. Pediatrics 2011; 128: e1502–e1510.
La Gamma EF, Krauss A, Auld PAM . Effects of increased red cell mass on subclinical tissue acidosis in hyaline membrane disease. Arch Dis Child Fetal Neonatal Ed 1996; 75: F87–F93.
Mintzer JP, Parvez B, Chelala M, Alpan G, LaGamma EF . Monitoring regional tissue oxygen extraction in neonates <1250 g helps identify transfusion thresholds independent of hematocrit. J Neonatal Perinatal Med 2014; 7: 89–100.
Bailey SM, Hendricks-Munoz KD, Mally P . Splanchnic-cerebral oxygenation ratio as a marker of preterm infant blood transfusion needs. Transfusion 2012; 52: 252–260.
Dani C, Pratesi S, Fontanelli G, Barp J, Bertini G . Blood transfusions increase cerebral, splanchnic, and renal oxygenation in anemic preterm infants. Transfusion 2010; 50: 1220–1226.
Petrova A, Bhatt M, Mehta R . Regional tissue oxygenation in preterm born infants in association with echocardiographically significant patent ductus arteriosus. J Perinatol 2011; 31: 460–464.
Wardle SP, Garr R, Yoxall CW, Weindling AM . A pilot randomised controlled trial of peripheral fractional oxygen extraction to guide blood transfusions in preterm infants. Arch Dis Child Fetal Neonatal Ed 2002; 86: F22–F27.
Bailey S, Hendricks-Muñoz K, Wells J, Mally P . Packed red blood cell transfusion increases regional cerebral and splanchnic tissue oxygen saturation in anemic symptomatic preterm infants. Am J Perinatol 2010; 27: 445–453.
Lemmers PM, Toet MC, van Bel F . Impact of patent ductus arteriosus and subsequent therapy with indomethacin on cerebral oxygenation in preterm infants. Pediatrics 2008; 121: 142–147.
van Bel F, Lemmers P, Naulaers G . Monitoring neonatal regional cerebral oxygen saturation in clinical practice: value and pitfalls. Neonatology 2008; 94: 237–244.
Chock VY, Ramamoorthy C, Van Meurs KP . Cerebral oxygenation during different treatment strategies for a patent ductus arteriosus. Neonatology 2011; 100: 233–240.
Cortez J, Gupta M, Amaram A, Pizzino J, Sawhney M, Sood BG . Noninvasive evaluation of splanchnic tissue oxygenation using near-infrared spectroscopy in preterm neonates. J Matern Fetal Neonatal Med 2010; 24: 574–582.
Kaufman J, Almodovar MC, Zuk J, Friesen RH . Correlation of abdominal site near-infrared spectroscopy with gastric tonometry in infants following surgery for congenital heart disease. Pediatr Crit Care Med 2008; 9: 62–68.
McNeill S, Gatenby JC, McElroy S, Engelhardt B . Normal cerebral, renal and abdominal regional oxygen saturations using near-infrared spectroscopy in preterm infants. J Perinatol 2010; 31: 51–57.
Mintzer JP, Parvez B, Chelala M, Alpan G, LaGamma EF . Quiescent variability of cerebral, renal, and splanchnic regional tissue oxygenation in very low birth weight neonates. J Neonatal Perinatal Med 2014; 7: 199–206.
Uebing A, Furck AK, Hansen JH, Nufer E, Scheewe J, Dutschke P et al. Perioperative cerebral and somatic oxygenation in neonates with hypoplastic left heart syndrome or transposition of the great arteries. J Thorac Cardiovasc Surg 2011; 142: 523–530.
Adan D, La Gamma EF, Browne L . Nutritional management and the multisystem organ failure/systemic inflammatory response syndrome in critically ill preterm neonates. Crit Care Clin 1995; 11: 751–784.
Haleem A, Zia M, Mishra R, Parton L, Stringel G, La Gamma EF . Bedside lung mechanics predict survival in hypoplastic lung disease. Am J Perinatol 2011; 28: 305–314.
La Gamma EF, Browne L . Feeding practices for infants weighing less than 1500 G at birth and the pathogenesis of necrotizing enterocolitis. Clin Perinatol 1994; 21: 271–306.
van Alfen-van der Velden AA, Hopman JC, Klaessens JH, Feuth T, Sengers RC, Liem KD . Effects of rapid versus slow infusion of sodium bicarbonate on cerebral hemodynamics and oxygenation in preterm infants. Biol Neonate 2006; 90: 122–127.
Binder C, Urlesberger B, Avian A, Pocivalnik M, Muller W, Pichler G . Cerebral and peripheral regional oxygen saturation during postnatal transition in preterm neonates. J Pediatr 2013; 163: 394–399.
Pichler G, Binder C, Avian A, Beckenbach E, Schmolzer GM, Urlesberger B . Reference ranges for regional cerebral tissue oxygen saturation and fractional oxygen extraction in neonates during immediate transition after birth. J Pediatr 2013; 163: 1558–1563.
Sandal G, Oguz SS, Erdeve O, Akar M, Uras N, Dilmen U . Assessment of red blood cell transfusion and transfusion duration on cerebral and mesenteric oxygenation using near-infrared spectroscopy in preterm infants with symptomatic anemia. Transfusion 2014; 54: 1100–1105.
van Hoften JCR, Verhagen EA, Keating P, ter Horst HJ, Bos AF . Cerebral tissue oxygen saturation and extraction in preterm infants before and after blood transfusion. Arch Dis Child Fetal Neonatal Ed 2010; 95: F352–F358.
Ancora G, Maranella E, Locatelli C, Pierantoni L, Faldella G . Changes in cerebral hemodynamics and amplitude integrated EEG in an asphyxiated newborn during and after cool cap treatment. Brain Dev 2009; 31: 442–444.
ter Horst HJ, Verhagen EA, Keating P, Bos AF . The relationship between electrocerebral activity and cerebral fractional tissue oxygen extraction in preterm infants. Pediatr Res 2011; 70: 384–388.
Zhang Y, Chan GS, Tracy MB, Lee QY, Hinder M, Savkin AV et al. Cerebral near-infrared spectroscopy analysis in preterm infants with intraventricular hemorrhage. Conf Proc IEEE Eng Med Biol Soc 2011; 2011: 1937–1940.
Ricci Z, Garisto C, Favia I, Schloderer U, Giorni C, Fragasso T et al. Cerebral NIRS as a marker of superior vena cava oxygen saturation in neonates with congenital heart disease. Paediatr Anaesth 2010; 20: 1040–1045.
Zulueta JL, Vida VL, Perisinotto E, Pittarello D, Stellin G . Role of intraoperative regional oxygen saturation using near infrared spectroscopy in the prediction of low output syndrome after pediatric heart surgery. J Card Surg 2013; 28: 446–452.
Rodig NM, McDermott KC, Schneider MF, Hotchkiss HM, Yadin O, Seikaly MG et al. Growth in children with chronic kidney disease: a report from the chronic kidney disease in children study. Pediatr Nephrol 2014; 29: 1987–1995.
Acknowledgements
We would like to thank the families of our patients for allowing us to perform NIRS monitoring to study the effects of NaHCO3 corrections. We also want to acknowledge the cooperation of our nursing and medical staff during the conduct of this work. The research in this report was supported in part by an unrestricted equipment use grant from Somanetics (Troy, MI); now Covidien (Mansfield, MA).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Mintzer, J., Parvez, B., Alpan, G. et al. Effects of sodium bicarbonate correction of metabolic acidosis on regional tissue oxygenation in very low birth weight neonates. J Perinatol 35, 601–606 (2015). https://doi.org/10.1038/jp.2015.37
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jp.2015.37
This article is cited by
-
Changes in regional oxygen saturation of the kidney and brain of infants during hospitalization
Journal of Clinical Monitoring and Computing (2022)
-
Cerebral oxygenation during umbilical arterial blood sampling in very low birth weight neonates
Journal of Perinatology (2018)
-
Hemodynamic effects of sodium bicarbonate administration
Journal of Perinatology (2017)