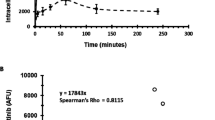
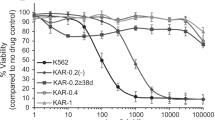
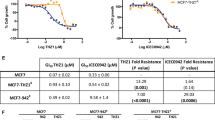
Abstract
Imatinib mesylate and nilotinib are highly effective at eradicating the majority of chronic myeloid leukemia (CML) cells; however, neither agent induces apoptosis of primitive CML CD34+ cells. One possible explanation is that CD34+ cells do not accumulate sufficient intracellular drug levels because of either inadequate active uptake or increased efflux. To determine the interaction of nilotinib with major clinically implicated drug transporters, we analyzed their interactions with MDR1 (ABCB1), MRP1 (ABCC1), ABCG2 (BCRP) and human organic cation transporter (hOCT)1 in CML cell lines and primitive (CD34+) primary CML cells. Nilotinib is neither dependent on active import by hOCT1, nor effluxed through the ATP-binding cassette transporters analyzed. Indeed, we found nilotinib to be an inhibitor of hOCT1, MDR1 and ABCG2. The efflux transporters MDR1, MRP1 and ABCG2 are expressed on CML CD34+ cells at 13.5, 108 and 291% of control, respectively, although hOCT1 expression was absent; however, inhibition of efflux transporter activity did not potentiate the effect of nilotinib on apoptosis, Bcr–Abl inhibition or CML CD34+ cell proliferation. Therefore, we have found no evidence for either active uptake of nilotinib through hOCT1 or efflux through MDR1, MRP1 or ABCG2, and it is therefore unlikely that these transporters will have any effect on the clinical response to this drug.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Druker BJ, Talpaz M, Resta DJ, Peng B, Buchdunger E, Ford JM et al. Efficacy and safety of a specific inhibitor of the BCR-ABL tyrosine kinase in chronic myeloid leukemia. N Engl J Med 2001; 344: 1031–1037.
Druker BJ, Guilhot F, O'Brien SG, Gathmann I, Kantarjian H, Gattermann N et al. Five-year follow-up of patients receiving imatinib for chronic myeloid leukemia. N Engl J Med 2006; 355: 2408–2417.
Kantarjian HM, Giles F, Gattermann N, Bhalla K, Alimena G, Palandri F et al. Nilotinib (formerly AMN107), a highly selective BCR-ABL tyrosine kinase inhibitor, is effective in patients with Philadelphia chromosome-positive chronic myelogenous leukemia in chronic phase following imatinib resistance and intolerance. Blood 2007; 110: 3540–3546.
Ottmann O, Dombret H, Martinelli G, Simonsson B, Guilhot F, Larson RA et al. Dasatinib induces rapid hematologic and cytogenetic responses in adult patients with Philadelphia chromosome positive acute lymphoblastic leukemia with resistance or intolerance to imatinib: interim results of a phase 2 study. Blood 2007; 110: 2309–2315.
Jorgensen HG, Allan EK, Jordanides NE, Mountford JC, Holyoake TL . Nilotinib exerts equipotent antiproliferative effects to imatinib and does not induce apoptosis in CD34+ CML cells. Blood 2007; 109: 4016–4019.
Elrick LJ, Jorgensen HG, Mountford JC, Holyoake TL . Punish the parent not the progeny. Blood 2005; 105: 1862–1866.
Bhatia R, Holtz M, Niu N, Gray R, Snyder DS, Sawyers CL et al. Persistence of malignant hematopoietic progenitors in chronic myelogenous leukemia patients in complete cytogenetic remission following imatinib mesylate treatment. Blood 2003; 101: 4701–4707.
Jordanides NE, Jorgensen HG, Holyoake TL, Mountford JC . Functional ABCG2 is overexpressed on primary CML CD34+ cells and is inhibited by imatinib mesylate. Blood 2006; 108: 1370–1373.
Burger H, van Tol H, Boersma AW, Brok M, Wiemer EA, Stoter G et al. Imatinib mesylate (STI571) is a substrate for the breast cancer resistance protein (BCRP)/ABCG2 drug pump. Blood 2004; 104: 2940–2942.
Houghton PJ, Germain GS, Harwood FC, Schuetz JD, Stewart CF, Buchdunger E et al. Imatinib mesylate is a potent inhibitor of the ABCG2 (BCRP) transporter and reverses resistance to topotecan and SN-38 in vitro. Cancer Res 2004; 64: 2333–2337.
Nakanishi T, Shiozawa K, Hassel BA, Ross DD . Complex interaction of BCRP/ABCG2 and imatinib in BCR-ABL-expressing cells: BCRP-mediated resistance to imatinib is attenuated by imatinib-induced reduction of BCRP expression. Blood 2006; 108: 678–684.
Burger H, van Tol H, Brok M, Wiemer EA, de Bruijn EA, Guetens G et al. Chronic imatinib mesylate exposure leads to reduced intracellular drug accumulation by induction of the ABCG2 (BCRP) and ABCB1 (MDR1) drug transport pumps. Cancer Biol Ther 2005; 4: 747–752.
Hamada A, Miyano H, Watanabe H, Saito H . Interaction of imatinib mesilate with human P-glycoprotein. J Pharmacol Exp Ther 2003; 307: 824–828.
Illmer T, Schaich M, Platzbecker U, Freiberg-Richter J, Oelschlagel U, von Bonin M et al. P-glycoprotein-mediated drug efflux is a resistance mechanism of chronic myelogenous leukemia cells to treatment with imatinib mesylate. Leukemia 2004; 18: 401–408.
Hatziieremia S, Jordanides NE, Holyoake TL, Mountford JC, Jorgensen HG . Inhibition of MDR1 does not sensitise primitive chronic myeloid leukaemia CD34+ cell to imatinib. Exp Hematol 2009; 37: 692–700.
Thomas J, Wang L, Clark RE, Pirmohamed M . Active transport of imatinib into and out of cells: implications for drug resistance. Blood 2004; 104: 3739–3745.
Wang L, Giannoudis A, Lane S, Williamson P, Pirmohamed M, Clark RE . Expression of the uptake drug transporter hOCT1 is an important clinical determinant of the response to imatinib in chronic myeloid leukemia. Clin Pharmacol Ther 2008; 83: 258–264.
Brendel C, Scharenberg C, Dohse M, Robey RW, Bates SE, Shukla S . et al. Imatinib mesylate and nilotinib (AMN107) exhibit high-affinity interaction with ABCG2 on primitive hematopoietic stem cells. Leukemia 2007; 21: 1267–1275.
White DL, Saunders VA, Dang P, Engler J, Zannettino AC, Cambareri AC et al. OCT-1-mediated influx is a key determinant of the intracellular uptake of imatinib but not nilotinib (AMN107): reduced OCT-1 activity is the cause of low in vitro sensitivity to imatinib. Blood 2006; 108: 697–704.
Wang DS, Jonker JW, Kato Y, Kusuhara H, Schinkel AH, Sugiyama Y . Involvement of organic cation transporter 1 in hepatic and intestinal distribution of metformin. J Pharmacol Exp Ther 2002; 302: 510–515.
Southgate TD, Garside E, Margison GP, Fairbairn LJ . Dual agent chemoprotection by retroviral co-expression of either MDR1 or MRP1 with the P140K mutant of O6-methylguanine-DNA-methyl transferase. J Gene Med 2006; 8: 972–979.
Graham SM, Jorgensen HG, Allan E, Pearson C, Alcorn MJ, Richmond L et al. Primitive, quiescent, Philadelphia-positive stem cells from patients with chronic myeloid leukemia are insensitive to STI571 in vitro. Blood 2002; 99: 319–325.
Hamilton A, Elrick L, Myssina S, Copland M, Jorgensen H, Melo JV et al. BCR-ABL activity and its response to drugs can be determined in CD34+ CML stem cells by CrkL phosphorylation status using flow cytometry. Leukemia 2006; 20: 1035–1039.
Robey RW, Honjo Y, van de Laar A, Miyake K, Regis JT, Litman T et al. A functional assay for detection of the mitoxantrone resistance protein, MXR (ABCG2). Biochim Biophys Acta 2001; 1512: 171–182.
Dai H, Marbach P, Lemaire M, Hayes M, Elmquist WF . Distribution of STI-571 to the brain is limited by P-glycoprotein-mediated efflux. J Pharmacol Exp Ther 2003; 304: 1085–1092.
Yunger LM, Cramer III RD . Measurement of correlation of partition coefficients of polar amino acids. Mol Pharmacol 1981; 20: 602–608.
Davies A, Hayes A, Giannoudis A, Lucas C, Knight K, Watmough S et al. A study of plasma levels of imatinib and its bioactive metabolite CGP-74588 reveals no correlation with subsequent clinical outcome in imatinib-treated chronic myeloid leukaemia. Haematologica 2009; 94 (s2): 259 (abstract 0637).
White DL, Saunders VA, Quinn SR, Manley PW, Hughes TP . Imatinib increases the intracellular concentration of nilotinib, which may explain the observed synergy between these drugs. Blood 2007; 109: 3609–3610.
Mahon FX, Belloc F, Lagarde V, Chollet C, Moreau-Gaudry F, Reiffers J et al. MDR1 gene overexpression confers resistance to imatinib mesylate in leukemia cell line models. Blood 2003; 101: 2368–2373.
Kock K, Grube M, Jedlitschky G, Oevermann L, Siegmund W, Ritter CA et al. Expression of adenosine triphosphate-binding cassette (ABC) drug transporters in peripheral blood cells: relevance for physiology and pharmacotherapy. Clin Pharmacokinet 2007; 46: 449–470.
Norgaard JM, Olesen LH, Hokland P . Changing picture of cellular drug resistance in human leukemia. Crit Rev Oncol Hematol 2004; 50: 39–49.
Galimberti S, Cervetti G, Guerrini F, Testi R, Pacini S, Fazzi R et al. Quantitative molecular monitoring of BCR-ABL and MDR1 transcripts in patients with chronic myeloid leukemia during imatinib treatment. Cancer Genet Cytogenet 2005; 162: 57–62.
Mahon F-X, Hayette S, Lagarde V, Belloc F, Turcq B, Nicolini F et al. Evidence that resistance to nilotinib may be due to BCR-ABL, Pgp, or Src kinase overexpression. Cancer Res 2008; 68: 9809–9816.
Baer MR, George SL, Dodge RK, O'Loughlin KL, Minderman H, Caligiuri MA et al. Phase 3 study of the multidrug resistance modulator PSC-833 in previously untreated patients 60 years of age and older with acute myeloid leukemia: cancer and Leukemia Group B Study 9720. Blood 2002; 100: 1224–1232.
Acknowledgements
We are grateful to Novartis Pharma for the supply of imatinib, nilotinib, radio-labelled nilotinib and PSC833. This work was supported by Leukaemia Research Great Britain (Grant #04034). AD, NEJ, AG, CML, SH and RJH conducted the experiments; AD, NEJ, AG, JCM and HGJ analyzed the results, compiled the figures and edited the manuscript; JCM, REC, TLH and MP designed and supervised the research; all authors contributed to the critical review of the data and preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Leukemia website (http://www.nature.com/leu)
Rights and permissions
About this article
Cite this article
Davies, A., Jordanides, N., Giannoudis, A. et al. Nilotinib concentration in cell lines and primary CD34+ chronic myeloid leukemia cells is not mediated by active uptake or efflux by major drug transporters. Leukemia 23, 1999–2006 (2009). https://doi.org/10.1038/leu.2009.166
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2009.166
Keywords
This article is cited by
-
CML Resistant to 2nd-Generation TKIs: Mechanisms, Next Steps, and New Directions
Current Hematologic Malignancy Reports (2022)
-
Efficiency of nilotinib to target chronic phase-chronic myeloid leukaemia primary mature CD34− and immature CD34+ cells
Scientific Reports (2021)
-
Past, present, and future of Bcr-Abl inhibitors: from chemical development to clinical efficacy
Journal of Hematology & Oncology (2018)
-
Chronic myeloid leukemia: the paradigm of targeting oncogenic tyrosine kinase signaling and counteracting resistance for successful cancer therapy
Molecular Cancer (2018)
-
Effect of bosutinib on the absorption of dabigatran etexilate mesylate, a P-glycoprotein substrate, in healthy subjects
European Journal of Clinical Pharmacology (2017)