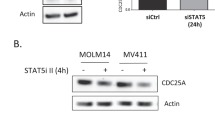
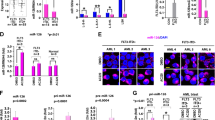
Abstract
Almost 30% of all acute myeloid leukemias (AML) are associated with an internal tandem duplication (ITD) in the juxtamembrane domain of FMS-like tyrosine kinase 3 receptor (FLT3). Patients with FLT3-ITD mutations tend to have a poor prognosis. MicroRNAs (miRNAs) have a pivotal role in myeloid differentiation and leukemia. MiRNA-155 (MiR-155) was found to be upregulated in FLT3-ITD-associated AMLs. In this study, we discovered that FLT3-ITD signaling induces the oncogenic miR-155. We show in vitro and in vivo that miR-155 expression is regulated by FLT3-ITD downstream targets nuclear factor-κB (p65) and signal transducer and activator of transcription 5 (STAT5). Further, we demonstrate that miR-155 targets the myeloid transcription factor PU.1. Knockdown of miR-155 or overexpression of PU.1 blocks proliferation and induces apoptosis of FLT3-ITD-associated leukemic cells. Our data demonstrate a novel network in which FLT3-ITD signaling induces oncogenic miR-155 by p65 and STAT5 in AML, thereby targeting transcription factor PU.1.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Gilliland DG, Griffin JD . The roles of FLT3 in hematopoiesis and leukemia. Blood 2002; 100: 1532–1542.
Zheng R, Small D . Mutant FLT3 signaling contributes to a block in myeloid differentiation. Leuk Lymphoma 2005; 46: 1679–1687.
Bienz M, Ludwig M, Leibundgut EO, Mueller BU, Ratschiller D, Solenthaler M et al. Risk assessment in patients with acute myeloid leukemia and a normal karyotype. Clin Cancer Res 2005; 11: 1416–1424.
Choudhary C, Schwable J, Brandts C, Tickenbrock L, Sargin B, Kindler T et al. AML-associated Flt3 kinase domain mutations show signal transduction differences compared with Flt3 ITD mutations. Blood 2005; 106: 265–273.
Choudhary C, Brandts C, Schwable J, Tickenbrock L, Sargin B, Ueker A et al. Activation mechanisms of STAT5 by oncogenic Flt3-ITD. Blood 2007; 110: 370–374.
Spiekermann K, Bagrintseva K, Schwab R, Schmieja K, Hiddemann W . Overexpression and constitutive activation of FLT3 induces STAT5 activation in primary acute myeloid leukemia blast cells. Clin Cancer Res 2003; 9: 2140–2150.
Hayakawa F, Towatari M, Kiyoi H, Tanimoto M, Kitamura T, Saito H et al. Tandem-duplicated Flt3 constitutively activates STAT5 and MAP kinase and introduces autonomous cell growth in IL-3-dependent cell lines. Oncogene 2000; 19: 624–631.
Tse KF, Mukherjee G, Small D . Constitutive activation of FLT3 stimulates multiple intracellular signal transducers and results in transformation. Leukemia 2000; 14: 1766–1776.
Chalandon Y, Schwaller J . Targeting mutated protein tyrosine kinases and their signaling pathways in hematologic malignancies. Haematologica 2005; 90: 949–968.
Mizuki M, Schwable J, Steur C, Choudhary C, Agrawal S, Sargin B et al. Suppression of myeloid transcription factors and induction of STAT response genes by AML-specific Flt3 mutations. Blood 2003; 101: 3164–3173.
Radomska HS, Basseres DS, Zheng R, Zhang P, Dayaram T, Yamamoto Y et al. Block of C/EBP alpha function by phosphorylation in acute myeloid leukemia with FLT3 activating mutations. J Exp Med 2006; 203: 371–381.
Mizuki M, Fenski R, Halfter H, Matsumura I, Schmidt R, Muller C et al. Flt3 mutations from patients with acute myeloid leukemia induce transformation of 32D cells mediated by the Ras and STAT5 pathways. Blood 2000; 96: 3907–3914.
Bartel DP . MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 2004 23; 116: 281–297.
Ambros V . The functions of animal microRNAs. Nature 2004; 431: 350–355.
Pulikkan JA, Dengler V, Peramangalam PS, Peer Zada AA, Muller-Tidow C, Bohlander SK et al. Cell-cycle regulator E2F1 and microRNA-223 comprise an autoregulatory negative feedback loop in acute myeloid leukemia. Blood 2010; 115: 1768–1778.
Katzerke C, Madan V, Gerloff D, Brauer-Hartmann D, Hartmann JU, Wurm AA et al. Transcription factor C/EBPalpha-induced microRNA-30c inactivates Notch1 during granulopoiesis and is downregulated in acute myeloid leukemia. Blood 2013; 122: 2433–2442.
Pulikkan JA, Peramangalam PS, Dengler V, Ho PA, Preudhomme C, Meshinchi S et al. C/EBPalpha regulated microRNA-34a targets E2F3 during granulopoiesis and is down-regulated in AML with CEBPA mutations. Blood 2010; 116: 5638–5649.
Schotte D, Pieters R, Den Boer ML . MicroRNAs in acute leukemia: from biological players to clinical contributors. Leukemia 2012; 26: 1–12.
Garzon R, Garofalo M, Martelli MP, Briesewitz R, Wang L, Fernandez-Cymering C et al. Distinctive microRNA signature of acute myeloid leukemia bearing cytoplasmic mutated nucleophosmin. Proc Natl Acad Sci USA 2008; 105: 3945–3950.
Garzon R, Volinia S, Liu CG, Fernandez-Cymering C, Palumbo T, Pichiorri F et al. MicroRNA signatures associated with cytogenetics and prognosis in acute myeloid leukemia. Blood 2008; 111: 3183–3189.
Jongen-Lavrencic M, Sun SM, Dijkstra MK, Valk PJ, Lowenberg B . MicroRNA expression profiling in relation to the genetic heterogeneity of acute myeloid leukemia. Blood 2008; 111: 5078–5085.
Cammarata G, Augugliaro L, Salemi D, Agueli C, La Rosa M, Dagnino L et al. Differential expression of specific microRNA and their targets in acute myeloid leukemia. Am J Hematol 2010; 85: 331–339.
Gatto G, Rossi A, Rossi D, Kroening S, Bonatti S, Mallardo M . Epstein–Barr virus latent membrane protein 1 trans-activates miR-155 transcription through the NF-kappaB pathway. Nucleic Acids Res 2008; 36: 6608–6619.
Lu F, Weidmer A, Liu CG, Volinia S, Croce CM, Lieberman PM . Epstein–Barr virus-induced miR-155 attenuates NF-kappaB signaling and stabilizes latent virus persistence. J Virol 2008; 82: 10436–10443.
Rodriguez A, Vigorito E, Clare S, Warren MV, Couttet P, Soond DR et al. Requirement of bic/microRNA-155 for normal immune function. Science 2007; 316: 608–611.
Eis PS, Tam W, Sun L, Chadburn A, Li Z, Gomez MF et al. Accumulation of miR-155 and BIC RNA in human B cell lymphomas. Proc Natl Acad Sci USA 2005; 102: 3627–3632.
Kluiver J, Poppema S, de Jong D, Blokzijl T, Harms G, Jacobs S et al. BIC and miR-155 are highly expressed in Hodgkin, primary mediastinal and diffuse large B cell lymphomas. J Pathol 2005; 207: 243–249.
Wang X, Tang S, Le SY, Lu R, Rader JS, Meyers C et al. Aberrant expression of oncogenic and tumor-suppressive microRNAs in cervical cancer is required for cancer cell growth. PLoS One 2008; 3: e2557.
Lee EJ, Gusev Y, Jiang J, Nuovo GJ, Lerner MR, Frankel WL et al. Expression profiling identifies microRNA signature in pancreatic cancer. Int J Cancer 2007; 120: 1046–1054.
Volinia S, Calin GA, Liu CG, Ambs S, Cimmino A, Petrocca F et al. A microRNA expression signature of human solid tumors defines cancer gene targets. Proc Natl Acad Sci USA 2006; 103: 2257–2261.
Iorio MV, Ferracin M, Liu CG, Veronese A, Spizzo R, Sabbioni S et al. MicroRNA gene expression deregulation in human breast cancer. Cancer Res 2005; 65: 7065–7070.
O'Connell RM, Rao DS, Chaudhuri AA, Boldin MP, Taganov KD, Nicoll J et al. Sustained expression of microRNA-155 in hematopoietic stem cells causes a myeloproliferative disorder. J Exp Med 2008; 205: 585–594.
Marcucci G, Maharry KS, Metzeler KH, Volinia S, Wu YZ, Mrozek K et al. Clinical role of microRNAs in cytogenetically normal acute myeloid leukemia: miR-155 upregulation independently identifies high-risk patients. J Clin Oncol 2013; 31: 2086–2093.
Kawashima T, Murata K, Akira S, Tonozuka Y, Minoshima Y, Feng S et al. STAT5 induces macrophage differentiation of M1 leukemia cells through activation of IL-6 production mediated by NF-kappaB p65. J Immunol 2001; 167: 3652–3660.
Cui Y, Riedlinger G, Miyoshi K, Tang W, Li C, Deng CX et al. Inactivation of Stat5 in mouse mammary epithelium during pregnancy reveals distinct functions in cell proliferation, survival, and differentiation. Mol Cell Biol 2004; 24: 8037–8047.
Weisberg E, Boulton C, Kelly LM, Manley P, Fabbro D, Meyer T et al. Inhibition of mutant FLT3 receptors in leukemia cells by the small molecule tyrosine kinase inhibitor PKC412. Cancer Cell 2002; 1: 433–443.
Yee KW, O'Farrell AM, Smolich BD, Cherrington JM, McMahon G, Wait CL et al. SU5416 and SU5614 inhibit kinase activity of wild-type and mutant FLT3 receptor tyrosine kinase. Blood 2002; 100: 2941–2949.
Spiekermann K, Dirschinger RJ, Schwab R, Bagrintseva K, Faber F, Buske C et al. The protein tyrosine kinase inhibitor SU5614 inhibits FLT3 and induces growth arrest and apoptosis in AML-derived cell lines expressing a constitutively activated FLT3. Blood 2003; 101: 1494–1504.
Levis M, Allebach J, Tse KF, Zheng R, Baldwin BR, Smith BD et al. A FLT3-targeted tyrosine kinase inhibitor is cytotoxic to leukemia cells in vitro and in vivo. Blood 2002; 99: 3885–3891.
Grundler R, Miething C, Thiede C, Peschel C, Duyster J . FLT3-ITD and tyrosine kinase domain mutants induce 2 distinct phenotypes in a murine bone marrow transplantation model. Blood 2005; 105: 4792–4799.
Thompson RC, Herscovitch M, Zhao I, Ford TJ, Gilmore TD . NF-kappaB down-regulates expression of the B-lymphoma marker CD10 through a miR-155/PU.1 pathway. J Biol Chem 2011; 286: 1675–1682.
Takahashi S, Harigae H, Ishii KK, Inomata M, Fujiwara T, Yokoyama H et al. Over-expression of Flt3 induces NF-kappaB pathway and increases the expression of IL-6. Leuk Res 2005; 29: 893–899.
Guzman ML, Neering SJ, Upchurch D, Grimes B, Howard DS, Rizzieri DA et al. Nuclear factor-kappaB is constitutively activated in primitive human acute myelogenous leukemia cells. Blood 2001; 98: 2301–2307.
Bueso-Ramos CE, Rocha FC, Shishodia S, Medeiros LJ, Kantarjian HM, Vadhan-Raj S et al. Expression of constitutively active nuclear-kappa B RelA transcription factor in blasts of acute myeloid leukemia. Hum Pathol 2004; 35: 246–253.
Grandage VL, Gale RE, Linch DC, Khwaja A . PI3-kinase/Akt is constitutively active in primary acute myeloid leukaemia cells and regulates survival and chemoresistance via NF-kappaB, Mapkinase and p53 pathways. Leukemia 2005; 19: 586–594.
Brandts CH, Sargin B, Rode M, Biermann C, Lindtner B, Schwable J et al. Constitutive activation of Akt by Flt3 internal tandem duplications is necessary for increased survival, proliferation, and myeloid transformation. Cancer Res 2005; 65: 9643–9650.
Grosjean-Raillard J, Ades L, Boehrer S, Tailler M, Fabre C, Braun T et al. Flt3 receptor inhibition reduces constitutive NFkappaB activation in high-risk myelodysplastic syndrome and acute myeloid leukemia. Apoptosis 2008; 13: 1148–1161.
Georgantas RW III, Hildreth R, Morisot S, Alder J, Liu CG, Heimfeld S et al. CD34+ hematopoietic stem-progenitor cell microRNA expression and function: a circuit diagram of differentiation control. Proc Natl Acad Sci USA 2007; 104: 2750–2755.
Fisher RC, Scott EW . Role of PU.1 in hematopoiesis. Stem Cells 1998; 16: 25–37.
Baumgartner B, Weber M, Quirling M, Fischer C, Page S, Adam M et al. Increased IkappaB kinase activity is associated with activated NF-kappaB in acute myeloid blasts. Leukemia 2002; 16: 2062–2071.
Kopp KL, Ralfkiaer U, Gjerdrum LM, Helvad R, Pedersen IH, Litman T et al. STAT5-mediated expression of oncogenic miR-155 in cutaneous T-cell lymphoma. Cell Cycle 2013; 12: 1939–1947.
Nakamura T, Ouchida R, Kodama T, Kawashima T, Makino Y, Yoshikawa N et al. Cytokine receptor common beta subunit-mediated STAT5 activation confers NF-kappa B activation in murine proB cell line Ba/F3 cells. J Biol Chem 2002; 277: 6254–6265.
Nagy ZS, LeBaron MJ, Ross JA, Mitra A, Rui H, Kirken RA . STAT5 regulation of BCL10 parallels constitutive NFkappaB activation in lymphoid tumor cells. Mol Cancer 2009; 8: 67.
Martinez-Nunez RT, Louafi F, Friedmann PS, Sanchez-Elsner T . MicroRNA-155 modulates the pathogen binding ability of dendritic cells (DCs) by down-regulation of DC-specific intercellular adhesion molecule-3 grabbing non-integrin (DC-SIGN). J Biol Chem 2009; 284: 16334–16342.
Vigorito E, Perks KL, Abreu-Goodger C, Bunting S, Xiang Z, Kohlhaas S et al. microRNA-155 regulates the generation of immunoglobulin class-switched plasma cells. Immunity 2007; 27: 847–859.
Metcalf D, Dakic A, Mifsud S, Di Rago L, Wu L, Nutt S . Inactivation of PU.1 in adult mice leads to the development of myeloid leukemia. Proc Natl Acad Sci USA 2006; 103: 1486–1491.
Rosenbauer F, Wagner K, Kutok JL, Iwasaki H, Le Beau MM, Okuno Y et al. Acute myeloid leukemia induced by graded reduction of a lineage-specific transcription factor, PU.1. Nat Genet 2004; 36: 624–630.
Voso MT, Burn TC, Wulf G, Lim B, Leone G, Tenen DG . Inhibition of hematopoiesis by competitive binding of transcription factor PU.1. Proc Natl Acad Sci USA 1994; 91: 7932–7936.
Babar IA, Cheng CJ, Booth CJ, Liang X, Weidhaas JB, Saltzman WM et al. Nanoparticle-based therapy in an in vivo microRNA-155 (miR-155)-dependent mouse model of lymphoma. Proc Natl Acad Sci USA 2012; 109: E1695–E1704.
Ueno S, Tatetsu H, Hata H, Iino T, Niiro H, Akashi K et al. PU.1 induces apoptosis in myeloma cells through direct transactivation of TRAIL. Oncogene 2009; 28: 4116–4125.
Zhang Y, Roccaro AM, Rombaoa C, Flores L, Obad S, Fernandes SM et al. LNA-mediated anti-miR-155 silencing in low-grade B-cell lymphomas. Blood 2012; 120: 1678–1686.
Acknowledgements
We thank Professor Dr M Mallardo for providing miR-155 promoter luciferase constructs, Professor Dr T Kitamura for STAT5 and STAT5 1*6 expression constructs and Professor Dr T Fischer for providing FLT3-WT, FLT3-ITD and FLT3-TKD expression constructs. This study was supported by grants from DFG (German Research Foundation, BE 2042/7-1), Deutsche José Carreras Leukämie-Stiftung e.V. (DJCLS R 11/17), Deutsche Krebshilfe and Translational Centre for Regenerative Medicine Leipzig (to GB), Deutsche José Carreras Leukämie-Stiftung e.V. (DJCLS F 08/05) (to DG), Deutsche José Carreras Leukämie-Stiftung e.V. (DJCLS F 12/03) (to AAW) and the National Institute of Health (CA118316) (to DGT).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Gerloff, D., Grundler, R., Wurm, A. et al. NF-κB/STAT5/miR-155 network targets PU.1 in FLT3-ITD-driven acute myeloid leukemia. Leukemia 29, 535–547 (2015). https://doi.org/10.1038/leu.2014.231
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2014.231
This article is cited by
-
A novel therapeutic strategy: the significance of exosomal miRNAs in acute myeloid leukemia
Medical Oncology (2024)
-
Epigenetic regulation in hematopoiesis and its implications in the targeted therapy of hematologic malignancies
Signal Transduction and Targeted Therapy (2023)
-
Roles of the miR-155 in Neuroinflammation and Neurological Disorders: A Potent Biological and Therapeutic Target
Cellular and Molecular Neurobiology (2023)
-
Functional Mutations in the microRNA-155 Promoter Modulate its Transcription Efficiency and Expression
Molecular Biotechnology (2023)
-
CircSPI1 acts as an oncogene in acute myeloid leukemia through antagonizing SPI1 and interacting with microRNAs
Cell Death & Disease (2021)