Abstract
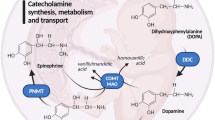
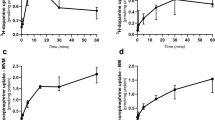
Serotonin (5-hydroxytryptamine or 5-HT) is thought to regulate neurodevelopmental processes through maternal–fetal interactions that have long-term mental health implications. It is thought that beyond fetal 5-HT neurons there are significant maternal contributions to fetal 5-HT during pregnancy1,2 but this has not been tested empirically. To examine putative central and peripheral sources of embryonic brain 5-HT, we used Pet1−/− (also called Fev) mice in which most dorsal raphe neurons lack 5-HT3. We detected previously unknown differences in accumulation of 5-HT between the forebrain and hindbrain during early and late fetal stages, through an exogenous source of 5-HT which is not of maternal origin. Using additional genetic strategies, a new technology for studying placental biology ex vivo and direct manipulation of placental neosynthesis, we investigated the nature of this exogenous source. We uncovered a placental 5-HT synthetic pathway from a maternal tryptophan precursor in both mice and humans. This study reveals a new, direct role for placental metabolic pathways in modulating fetal brain development and indicates that maternal–placental–fetal interactions could underlie the pronounced impact of 5-HT on long-lasting mental health outcomes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Cote, F. et al. Maternal serotonin is crucial for murine embryonic development. Proc. Natl Acad. Sci. USA 104, 329–334 (2007)
Yavarone, M. S., Shuey, D. L., Sadler, T. W. & Lauder, J. M. Serotonin uptake in the ectoplacental cone and placenta of the mouse. Placenta 14, 149–161 (1993)
Hendricks, T. J. et al. Pet-1 ETS gene plays a critical role in 5-HT neuron development and is required for normal anxiety-like and aggressive behavior. Neuron 37, 233–247 (2003)
Bonnin, A., Torii, M., Wang, L., Rakic, P. & Levitt, P. Serotonin modulates the response of embryonic thalamocortical axons to netrin-1. Nature Neurosci. 10, 588–597 (2007)
Ansorge, M. S., Zhou, M., Lira, A., Hen, R. & Gingrich, J. A. Early-life blockade of the 5-HT transporter alters emotional behavior in adult mice. Science 306, 879–881 (2004)
Gross, C. et al. Serotonin1A receptor acts during development to establish normal anxiety-like behaviour in the adult. Nature 416, 396–400 (2002)
Taylor, S. E. et al. Early family environment, current adversity, the serotonin transporter promoter polymorphism, and depressive symptomatology. Biol. Psychiatry 60, 671–676 (2006)
Bonnin, A., Peng, W., Hewlett, W. & Levitt, P. Expression mapping of 5–HT1 serotonin receptor subtypes during fetal and early postnatal mouse forebrain development. Neuroscience 141, 781–794 (2006)
Anderson, G. M. et al. Serotonin transporter promoter variants in autism: functional effects and relationship to platelet hyperserotonemia. Mol. Psychiatry 7, 831–836 (2002)
Gaspar, P., Cases, O. & Maroteaux, L. The developmental role of serotonin: news from mouse molecular genetics. Nature Rev. Neurosci. 4, 1002–1012 (2003)
Buznikov, G. A., Lambert, H. W. & Lauder, J. M. Serotonin and serotonin-like substances as regulators of early embryogenesis and morphogenesis. Cell Tissue Res. 305, 177–186 (2001)
Aitken, A. R. & Tork, I. Early development of serotonin-containing neurons and pathways as seen in wholemount preparations of the fetal rat brain. J. Comp. Neurol. 274, 32–47 (1988)
Lidov, H. G. & Molliver, M. E. Immunohistochemical study of the development of serotonergic neurons in the rat CNS. Brain Res. Bull. 9, 559–604 (1982)
Lebrand, C. et al. Transient uptake and storage of serotonin in developing thalamic neurons. Neuron 17, 823–835 (1996)
Lebrand, C. et al. Transient developmental expression of monoamine transporters in the rodent forebrain. J. Comp. Neurol. 401, 506–524 (1998)
Scott, A. L., Bortolato, M., Chen, K. & Shih, J. C. Novel monoamine oxidase A knock out mice with human-like spontaneous mutation. Neuroreport 19, 739–743 (2008)
Lynn-Bullock, C. P., Welshhans, K., Pallas, S. L. & Katz, P. S. The effect of oral 5-HTP administration on 5-HTP and 5-HT immunoreactivity in monoaminergic brain regions of rats. J. Chem. Neuroanat. 27, 129–138 (2004)
Branchek, T. A. & Gershon, M. D. Time course of expression of neuropeptide Y, calcitonin gene-related peptide, and NADPH diaphorase activity in neurons of the developing murine bowel and the appearance of 5-hydroxytryptamine in mucosal enterochromaffin cells. J. Comp. Neurol. 285, 262–273 (1989)
Chen, J. J. et al. Maintenance of serotonin in the intestinal mucosa and ganglia of mice that lack the high-affinity serotonin transporter: Abnormal intestinal motility and the expression of cation transporters. J. Neurosci. 21, 6348–6361 (2001)
Howd, R. A., Nelson, M. F. & Lytle, L. D. L-tryptophan and rat fetal brain serotonin. Life Sci. 17, 803–811 (1975)
Balkovetz, D. F., Tiruppathi, C., Leibach, F. H., Mahesh, V. B. & Ganapathy, V. Evidence for an imipramine-sensitive serotonin transporter in human placental brush-border membranes. J. Biol. Chem. 264, 2195–2198 (1989)
Robson, J. M. & Senior, J. B. The 5-hydroxytryptamine content of the placenta and foetus during pregnancy in mice. Br. J. Pharmacol. Chemother. 22, 380–391 (1964)
Kim, H. et al. Serotonin regulates pancreatic beta cell mass during pregnancy. Nature Med. 16, 804–808 (2010)
Jawerbaum, A. & White, V. Animal models in diabetes and pregnancy. Endocr. Rev. 31, 680–701 (2010)
Suzuki, S. et al. Expression of indoleamine 2,3-dioxygenase and tryptophan 2,3-dioxygenase in early concepti. Biochem. J. 355, 425–429 (2001)
Kanai, M. et al. Tryptophan 2,3-dioxygenase is a key modulator of physiological neurogenesis and anxiety-related behavior in mice. Mol. Brain 2 8 10.1186/1756-6606-2-8 (2009)
Miller, C. L. et al. Two complex genotypes relevant to the kynurenine pathway and melanotropin function show association with schizophrenia and bipolar disorder. Schizophr. Res. 113, 259–267 (2009)
Nabi, R., Serajee, F. J., Chugani, D. C., Zhong, H. & Huq, A. H. Association of tryptophan 2,3 dioxygenase gene polymorphism with autism. Am. J. Med. Genet. B. Neuropsychiatr. Genet. 125B, 63–68 (2004)
Brown, A. S. & Derkits, E. J. Prenatal infection and schizophrenia: a review of epidemiologic and translational studies. Am. J. Psychiatry 167, 261–280 (2010)
Oberlander, T. F., Gingrich, J. A. & Ansorge, M. S. Sustained neurobehavioral effects of exposure to SSRI antidepressants during development: molecular to clinical evidence. Clin. Pharmacol. Ther. 86, 672–677 (2009)
Johansen, P. A., Jennings, I., Cotton, R. G. & Kuhn, D. M. Tryptophan hydroxylase is phosphorylated by protein kinase A. J. Neurochem. 65, 882–888 (1995)
Kuhn, D. M., Ruskin, B. & Lovenberg, W. Tryptophan hydroxylase. The role of oxygen, iron, and sulfhydryl groups as determinants of stability and catalytic activity. J. Biol. Chem. 255, 4137–4143 (1980)
Chen, K. et al. Forebrain-specific expression of monoamine oxidase A reduces neurotransmitter levels, restores the brain structure, and rescues aggressive behavior in monoamine oxidase A-deficient mice. J. Biol. Chem. 282, 115–123 (2007)
Nagai, A., Takebe, K., Nio-Kobayashi, J., Takahashi-Iwanaga, H. & Iwanaga, T. Cellular expression of the monocarboxylate transporter (MCT) family in the placenta of mice. Placenta 31, 126–133 (2010)
Koe, B. K. & Weissman, A. p-Chlorophenylalanine: a specific depletor of brain serotonin. J. Pharmacol. Exp. Ther. 154, 499–516 (1966)
Acknowledgements
We thank H.H. Wu and K. Eagleson for discussions and comments on the manuscript. We thank R. Johnson (Vanderbilt HPLC core facility), L. Zhang for technical help and E. Meng for discussions. This work was supported by the NICHD (grant 5R21HD065287 to A.B.), NARSAD (A.B.) and the NIMH (grant R01MH39085 to J.C.S. and 1P50MH078280A1 to R.D.B. and P.L.).
Author information
Authors and Affiliations
Contributions
A.B. conducted the experiments with assistance from N.G. in placenta studies, K.C. and J.C.S. in providing mutant mouse strains and in MAOA enzymatic assays. R.D.B. and E.S.D. provided mutant mouse strains and M.L.W. and J.K. provided human tissue. A.B. and P.L. conceived this study, interpreted the data and wrote the manuscript. All authors commented on the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Tables 1-2, a Supplementary Discussion with additional references and Supplementary Figures 1-5 with legends. (PDF 2230 kb)
Rights and permissions
About this article
Cite this article
Bonnin, A., Goeden, N., Chen, K. et al. A transient placental source of serotonin for the fetal forebrain. Nature 472, 347–350 (2011). https://doi.org/10.1038/nature09972
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature09972
This article is cited by
-
Preeclampsia as a possible risk factor for memory impairment, anxiety- and depressive-like behavior in offspring
Psychopharmacology (2024)
-
PIBF1 regulates trophoblast syncytialization and promotes cardiovascular development
Nature Communications (2024)
-
Identification and prediction model of placenta-brain axis genes associated with neurodevelopmental delay in moderate and late preterm children
BMC Medicine (2023)
-
Characterization of a human placental clearance system to regulate serotonin levels in the fetoplacental unit
Reproductive Biology and Endocrinology (2023)
-
The Serotonergic System and Bone Metabolism During Pregnancy and Lactation and the Implications of SSRI Use on the Maternal-Offspring Dyad
Journal of Mammary Gland Biology and Neoplasia (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.