Abstract
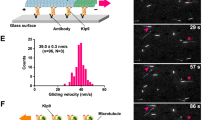
The budding yeast protein Kip3p is a member of the conserved kinesin-8 family of microtubule motors, which are required for microtubule–cortical interactions, normal spindle assembly and kinetochore dynamics. Here, we demonstrate that Kip3p is both a plus end-directed motor and a plus end-specific depolymerase — a unique combination of activities not found in other kinesins. The ATPase activity of Kip3p was activated by both microtubules and unpolymerized tubulin. Furthermore, Kip3p in the ATP-bound state formed a complex with unpolymerized tubulin. Thus, motile kinesin-8s may depolymerize microtubules by a mechanism that is similar to that used by non-motile kinesin-13 proteins. Fluorescent speckle analysis established that, in vivo, Kip3p moved toward and accumulated on the plus ends of growing microtubules, suggesting that motor activity brings Kip3p to its site of action. Globally, and more dramatically on cortical contact, Kip3p promoted catastrophes and pausing, and inhibited microtubule growth. These findings explain the role of Kip3p in positioning the mitotic spindle in budding yeast and potentially other processes controlled by kinesin-8 family members.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Nogales, E. Structural insights into microtubule function. Annu. Rev. Biochem. 69, 277–302 (2000).
Sharp, D. J., Rogers, G. C. & Scholey, J. M. Roles of motor proteins in building microtubule-based structures: a basic principle of cellular design. Biochim. Biophys. Acta 1496, 128–141 (2000).
Busch, K. E., Hayles, J., Nurse, P. & Brunner, D. Tea2p kinesin is involved in spatial microtubule organization by transporting tip1p on microtubules. Dev. Cell 6, 831–843 (2004).
Carvalho, P., Gupta, M. L., Jr., Hoyt, M. A. & Pellman, D. Cell cycle control of kinesin-mediated transport of Bik1 (CLIP-170) regulates microtubule stability and dynein activation. Dev. Cell 6, 815–829 (2004).
Moore, A. & Wordeman, L. The mechanism, function and regulation of depolymerizing kinesins during mitosis. Trends Cell Biol. 14, 537–546 (2004).
Endow, S. A. et al. Yeast Kar3 is a minus-end microtubule motor protein that destabilizes microtubules preferentially at the minus ends. EMBO J. 13, 2708–2713 (1994).
Sproul, L. R., Anderson, D. J., Mackey, A. T., Saunders, W. S. & Gilbert, S. P. Cik1 targets the minus-end kinesin depolymerase kar3 to microtubule plus ends. Curr. Biol. 15, 1420–1427 (2005).
Cottingham, F. R. & Hoyt, M. A. Mitotic spindle positioning in Saccharomyces cerevisiae is accomplished by antagonistically acting microtubule motor proteins. J. Cell. Biol. 138, 1041–1053 (1997).
DeZwaan, T. M., Ellingson, E., Pellman, D. & Roof, D. M. Kinesin-related KIP3 of Saccharomyces cerevisiae is required for a distinct step in nuclear migration. J. Cell. Biol. 138, 1023–1040 (1997).
Miller, R. K. et al. The kinesin-related proteins, Kip2p and Kip3p, function differently in nuclear migration in yeast. Mol. Biol. Cell 9, 2051–2068 (1998).
West, R. R., Malmstrom, T., Troxell, C. L. & McIntosh, J. R. Two related kinesins, klp5+ and klp6+, foster microtubule disassembly and are required for meiosis in fission yeast. Mol. Biol. Cell 12, 3919–3932 (2001).
Garcia, M. A., Koonrugsa, N. & Toda, T. Two kinesin-like Kin I family proteins in fission yeast regulate the establishment of metaphase and the onset of anaphase A. Curr. Biol. 12, 610–621 (2002).
Gatt, M. K. et al. Klp67A destabilises pre-anaphase microtubules but subsequently is required to stabilise the central spindle. J. Cell Sci. 118, 2671–2682 (2005).
Goshima, G. & Vale, R. D. The roles of microtubule-based motor proteins in mitosis: comprehensive RNAi analysis in the Drosophila S2 cell line. J. Cell Biol. 162, 1003–1016 (2003).
Goshima, G., Wollman, R., Stuurman, N., Scholey, J. M. & Vale, R. D. Length control of the metaphase spindle. Curr. Biol. 15, 1979–1988 (2005).
Pereira, A. J., Dalby, B., Stewart, R. J., Doxsey, S. J. & Goldstein, L. S. Mitochondrial association of a plus end-directed microtubule motor expressed during mitosis in Drosophila. J. Cell Biol. 136, 1081–1090 (1997).
Rischitor, P. E., Konzack, S. & Fischer, R. The Kip3-like kinesin KipB moves along microtubules and determines spindle position during synchronized mitoses in Aspergillus nidulans hyphae. Eukaryot. Cell 3, 632–645 (2004).
Tytell, J. D. & Sorger, P. K. Analysis of kinesin motor function at budding yeast kinetochores. J. Cell Biol. 172, 861–874 (2006).
Zhu, C. et al. Functional analysis of human microtubule-based motor proteins, the kinesins and dyneins, in mitosis/cytokinesis using RNA interference. Mol. Biol. Cell 16, 3187–3199 (2005).
Miki, H., Okada, Y. & Hirokawa, N. Analysis of the kinesin superfamily: insights into structure and function. Trends Cell Biol. 15, 467–476 (2005).
Severin, F., Habermann, B., Huffaker, T. & Hyman, T. Stu2 promotes mitotic spindle elongation in anaphase. J. Cell Biol. 153, 435–442 (2001).
Ogawa, T., Nitta, R., Okada, Y. & Hirokawa, N. A common mechanism for microtubule destabilizers-M type kinesins stabilize curling of the protofilament using the class-specific neck and loops. Cell 116, 591–602 (2004).
Shipley, K. et al. Structure of a kinesin microtubule depolymerization machine. EMBO J. 23, 1422–1432 (2004).
Cottingham, F. R., Gheber, L., Miller, D. L. & Hoyt, M. A. Novel roles for Saccharomyces cerevisiae mitotic spindle motors. J. Cell Biol. 147, 335–350 (1999).
Straight, A. F., Sedat, J. W. & Murray, A. W. Time-lapse microscopy reveals unique roles for kinesins during anaphase in budding yeast. J. Cell Biol. 143, 687–694 (1998).
Waterman-Storer, C. M., Desai, A., Bulinski, J. C. & Salmon, E. D. Fluorescent speckle microscopy, a method to visualize the dynamics of protein assemblies in living cells. Curr. Biol. 8, 1227–1230 (1998).
Akhmanova, A. & Hoogenraad, C. C. Microtubule plus-end-tracking proteins: mechanisms and functions. Curr. Opin. Cell Biol. 17, 47–54 (2005).
Howard, J. & Hyman, A. A. Preparation of marked microtubules for the assay of the polarity of microtubule-based motors by fluorescence microscopy. Methods Cell Biol. 39, 105–113 (1993).
Hyman, A. A., Salser, S., Drechsel, D. N., Unwin, N. & Mitchison, T. J. Role of GTP hydrolysis in microtubule dynamics: information from a slowly hydrolyzable analogue, GMPCPP. Mol. Biol. Cell 3, 1155–1167 (1992).
Desai, A., Verma, S., Mitchison, T. J. & Walczak, C. E. Kin I kinesins are microtubule-destabilizing enzymes. Cell 96, 69–78 (1999).
Hunter, A. W. et al. The kinesin-related protein MCAK is a microtubule depolymerase that forms an ATP-hydrolyzing complex at microtubule ends. Mol. Cell 11, 445–457 (2003).
Liakopoulos, D., Kusch, J., Grava, S., Vogel, J. & Barral, Y. Asymmetric loading of Kar9 onto spindle poles and microtubules ensures proper spindle alignment. Cell 112, 561–574 (2003).
Pearson, C. G. & Bloom, K. Dynamic microtubules lead the way for spindle positioning. Nature Rev. Mol. Cell Biol. 5, 481–492 (2004).
Adames, N. R. & Cooper, J. A. Microtubule interactions with the cell cortex causing nuclear movements in Saccharomyces cerevisiae. J. Cell Biol. 149, 863–874 (2000).
Maddox, P. S., Stemple, J. K., Satterwhite, L., Salmon, E. D. & Bloom, K. The minus end-directed motor Kar3 is required for coupling dynamic microtubule plus ends to the cortical shmoo tip in budding yeast. Curr. Biol. 13, 1423–1428 (2003).
Brunner, D. & Nurse, P. CLIP170-like tip1p spatially organizes microtubular dynamics in fission yeast. Cell 102, 695–704 (2000).
Tran, P. T., Marsh, L., Doye, V., Inoue, S. & Chang, F. A mechanism for nuclear positioning in fission yeast based on microtubule pushing. J. Cell Biol. 153, 397–411 (2001).
Komarova, Y. A., Vorobjev, I. A. & Borisy, G. G. Life cycle of MTs: persistent growth in the cell interior, asymmetric transition frequencies and effects of the cell boundary. J. Cell Sci. 115, 3527–3539 (2002).
Bringmann, H. et al. A kinesin-like motor inhibits microtubule dynamic instability. Science 303, 1519–1522 (2004).
Mennella, V. et al. Functionally distinct kinesin-13 family members cooperate to regulate microtubule dynamics during interphase. Nature Cell Biol. 7, 235–245 (2005).
Moore, A. T. et al. MCAK associates with the tips of polymerizing microtubules. J. Cell Biol. 169, 391–397 (2005).
Wadsworth, P. Regional regulation of microtubule dynamics in polarized, motile cells. Cell Motil. Cytoskeleton 42, 48–59 (1999).
Wittmann, T., Bokoch, G. M. & Waterman-Storer, C. M. Regulation of leading edge microtubule and actin dynamics downstream of Rac1. J. Cell Biol. 161, 845–851 (2003).
Labbe, J. C., Maddox, P. S., Salmon, E. D. & Goldstein, B. PAR proteins regulate microtubule dynamics at the cell cortex in C. elegans. Curr. Biol. 13, 707–714 (2003).
Kline-Smith, S. L. & Walczak, C. E. The microtubule-destabilizing kinesin XKCM1 regulates microtubule dynamic instability in cells. Mol. Biol. Cell 13, 2718–2731 (2002).
Rose, M. D., Winston, F. & Hieter, P. Methods in Yeast Genetics (Cold Spring Harbor Laboratory Press, 1990).
Hyman, A. et al. Preparation of modified tubulins. Methods Enzymol. 196, 478–485 (1991).
Paschal, B. M. & Vallee, R. B. Microtubule and axoneme gliding assays for force production by microtubule motor proteins. Methods Cell Biol. 39, 65–74 (1993).
Huang, T. G. & Hackney, D. D. Drosophila kinesin minimal motor domain expressed in Escherichia coli. Purification and kinetic characterization. J. Biol. Chem. 269, 16493–16501 (1994).
Tirnauer, J. S., O'Toole, E., Berrueta, L., Bierer, B. E. & Pellman, D. Yeast Bim1p promotes the G1-specific dynamics of microtubules. J. Cell Biol. 145, 993–1007 (1999).
Acknowledgements
We are grateful to K. Bloom, J. Huff, A. Murray, R. Ohi and A. Straight for providing reagents. We thank S. Gilbert, T. Mitchison, H. Sosa and members of the Pellman lab for discussion, and S. Buttery, N. Chandhok, S. Gilbert, M. Guillet and S. Yoshida for comments on the manuscript. M. Gupta was supported by a postdoctoral fellowship from the American Cancer Society (ACS; PF-05-025-01-CCG). D. Pellman was supported by a National Institutes of Health (NIH) grant (GM R0161345).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures S1, S2, S3 and Supplementary Methods (PDF 1194 kb)
Supplementary Information
Supplementary Movie 1 (MOV 230 kb)
Supplementary Information
Supplementary Movie 2 (MOV 1683 kb)
Supplementary Information
Supplementary Movie 3 (MOV 184 kb)
Supplementary Information
Supplementary Movie 4 (MOV 363 kb)
Supplementary Information
Supplementary Movie 5 (MOV 842 kb)
Supplementary Information
Supplementary Movie 6 (MOV 790 kb)
Supplementary Information
Supplementary Movie 7 (MOV 273 kb)
Supplementary Information
Supplementary Movie 8 (MOV 509 kb)
Supplementary Information
Supplementary Movie 9 (MOV 368 kb)
Supplementary Information
Supplementary Movie 10 (MOV 659 kb)
Rights and permissions
About this article
Cite this article
Gupta, M., Carvalho, P., Roof, D. et al. Plus end-specific depolymerase activity of Kip3, a kinesin-8 protein, explains its role in positioning the yeast mitotic spindle. Nat Cell Biol 8, 913–923 (2006). https://doi.org/10.1038/ncb1457
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1457
This article is cited by
-
Regulation of ciliary homeostasis by intraflagellar transport-independent kinesins
Cell Death & Disease (2024)
-
Determinant factors for residence time of kinesin motors at microtubule ends
Journal of Biological Physics (2023)
-
Spindle positioning and its impact on vertebrate tissue architecture and cell fate
Nature Reviews Molecular Cell Biology (2021)
-
A model of processive walking and slipping of kinesin-8 molecular motors
Scientific Reports (2021)
-
Kinesin-8 motors: regulation of microtubule dynamics and chromosome movements
Chromosoma (2020)