Abstract
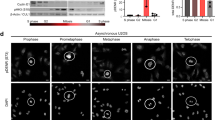
The cell cycle is exquisitely controlled by multiple sequential regulatory inputs to ensure fidelity. Here we demonstrate that the final step in division, the physical separation of daughter cells, is controlled by a member of the PKC gene superfamily. Specifically, we have identified three phosphorylation sites within PKCɛ that control its association with 14-3-3. These phosphorylations are executed by p38 MAP kinase (Ser 350), GSK3 (Ser 346) and PKC itself (Ser 368). Integration of these signals is essential during mitosis because mutations that prevent phosphorylation of PKCɛ and/or PKCɛ binding to 14-3-3 also cause defects in the completion of cytokinesis. Using chemical genetic and dominant-negative approaches it is shown that selective inhibition of PKCɛ halts cells at the final stages of separation. This arrest is associated with persistent RhoA activation at the midbody and a delay in actomyosin ring dissociation. This study therefore identifies a new regulatory mechanism that controls exit from cytokinesis, which has implications for carcinogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Mellor, H. & Parker, P. J. The extended protein kinase C superfamily. Biochem. J. 332, 281–292 (1998).
Newton, A. C. Protein kinase C: structure, function, and regulation. J. Biol. Chem. 270, 28495–28498 (1995).
Bridges, D. & Moorhead, G. B. 14–3-3 proteins: a number of functions for a numbered protein. Sci. STKE 2005, re10 (2005).
Yaffe, M. B. et al. The structural basis for 14–3-3:phosphopeptide binding specificity. Cell 91, 961–971 (1997).
Lo, M. C., Gay, F., Odom, R., Shi, Y. & Lin, R. Phosphorylation by the β-catenin/MAPK complex promotes 14–3-3-mediated nuclear export of TCF/POP-1 in signal-responsive cells in C. elegans. Cell 117, 95–106 (2004).
LeBron, C., Chen, L., Gilkes, D. M. & Chen, J. Regulation of MDMX nuclear import and degradation by Chk2 and 14–3-3. EMBO J. 25, 1196–1206 (2006).
Obsil, T., Ghirlando, R., Klein, D. C., Ganguly, S. & Dyda, F. Crystal structure of the 14–3-3ζ :serotonin N-acetyltransferase complex. A role for scaffolding in enzyme regulation. Cell 105, 257–267 (2001).
Han, D. C., Rodriguez, L. G. & Guan, J. L. Identification of a novel interaction between integrin beta1 and 14–3-3β. Oncogene 20, 346–357 (2001).
Benton, R. & St Johnston, D. Drosophila PAR-1 and 14–3-3 inhibit Bazooka/PAR-3 to establish complementary cortical domains in polarized cells. Cell 115, 691–704 (2003).
Peng, C. Y. et al. Mitotic and G2 checkpoint control: regulation of 14–3-3 protein binding by phosphorylation of Cdc25C on serine-216. Science 277, 1501–1505 (1997).
Eggert, U. S., Mitchison, T. J. & Field, C. M. Animal cytokinesis: from parts list to mechanisms. Annu. Rev. Biochem. 75, 543–566 (2006).
Glotzer, M. The molecular requirements for cytokinesis. Science 307, 1735–1739 (2005).
Fujiwara, T. et al. Cytokinesis failure generating tetraploids promotes tumorigenesis in p53-null cells. Nature 437, 1043–1047 (2005).
Nguyen, T. A., Takemoto, L. J. & Takemoto, D. J. Inhibition of gap junction activity through the release of the C1B domain of protein kinase Cgamma (PKCγ) from 14–3-3: identification of PKCγ-binding sites. J. Biol. Chem. 279, 52714–52725 (2004).
Dai, J. G. & Murakami, K. Constitutively and autonomously active protein kinase C associated with 14–3-3 ζ in the rodent brain. J. Neurochem. 84, 23–34 (2003).
Durgan, J. et al. The identification and characterisation of novel PKCɛ phosphorylation sites provides evidence for functional cross-talk within the PKC superfamily. Biochem. J. 411, 319–331 (2008).
Aitken, A. 14–3-3 proteins: a historic overview. Semin. Cancer Biol. 16, 162–172 (2006).
Eyers, P. A., van den, I. P., Quinlan, R. A., Goedert, M. & Cohen, P. Use of a drug-resistant mutant of stress-activated protein kinase 2a/p38 to validate the in vivo specificity of SB 203580. FEBS Lett. 451, 191–196 (1999).
Fiol, C. J., Wang, A., Roeske, R. W. & Roach, P. J. Ordered multisite protein phosphorylation. Analysis of glycogen synthase kinase 3 action using model peptide substrates. J. Biol. Chem. 265, 6061–6065 (1990).
Wilker, E. W. et al. 14–3-3σ controls mitotic translation to facilitate cytokinesis. Nature 446, 329–332 (2007).
Thorson, J. A. et al. 14–3-3 proteins are required for maintenance of Raf-1 phosphorylation and kinase activity. Mol. Cell Biol. 18, 5229–5238 (1998).
Wilker, E. W., Grant, R. A., Artim, S. C. & Yaffe, M. B. A structural basis for 14–3-3σ functional specificity. J. Biol. Chem. 280, 18891–18898 (2005).
Bishop, A. C. et al. A chemical switch for inhibitor-sensitive alleles of any protein kinase. Nature 407, 395–401 (2000).
Emoto, K., Inadome, H., Kanaho, Y., Narumiya, S. & Umeda, M. Local change in phospholipid composition at the cleavage furrow is essential for completion of cytokinesis. J. Biol. Chem. 280, 37901–37907 (2005).
Emoto, K. & Umeda, M. An essential role for a membrane lipid in cytokinesis. Regulation of contractile ring disassembly by redistribution of phosphatidylethanolamine. J. Cell Biol. 149, 1215–1224 (2000).
Dadke, D., Jarnik, M., Pugacheva, E. N., Singh, M. K. & Golemis, E. A. Deregulation of HEF1 impairs M-phase progression by disrupting the RhoA activation cycle. Mol. Biol. Cell 17, 1204–1217 (2006).
Yuce, O., Piekny, A. & Glotzer, M. An ECT2-centralspindlin complex regulates the localization and function of RhoA. J. Cell Biol. 170, 571–582 (2005).
Minoshima, Y. et al. Phosphorylation by aurora B converts MgcRacGAP to a RhoGAP during cytokinesis. Dev. Cell 4, 549–560 (2003).
Shi, Q. & King, R. W. Chromosome nondisjunction yields tetraploid rather than aneuploid cells in human cell lines. Nature 437, 1038–1042 (2005).
Li, F., Wang, X., Bunger, P. C. & Gerdes, A. M. Formation of binucleated cardiac myocytes in rat heart: I. Role of actin-myosin contractile ring. J. Mol. Cell. Cardiol. 29, 1541–1551 (1997).
Geddis, A. E., Fox, N. E., Tkachenko, E. & Kaushansky, K. Endomitotic megakaryocytes that form a bipolar spindle exhibit cleavage furrow ingression followed by furrow regression. Cell Cycle 6, 455–460 (2007).
Faisal, A., Saurin, A., Gregory, B., Foxwell, B. & Parker, P. J. The scaffold MYD88 acts to couple protein kinase C ɛ (PKCɛ) to toll-like receptors. J. Biol. Chem. advance online publication, doi 10:1074/jbc.M710330200 (5 May 2008).
Saurin, A. T. et al. Targeted disruption of the protein kinase C ɛ gene abolishes the infarct size reduction that follows ischaemic preconditioning of isolated buffer-perfused mouse hearts. Cardiovasc. Res. 55, 672–680 (2002).
Griner, E. M. & Kazanietz, M. G. Protein kinase C and other diacylglycerol effectors in cancer. Nature Rev. 7, 281–294 (2007).
Kim, L. & Kimmel, A. R. GSK3, a master switch regulating cell-fate specification and tumorigenesis. Curr. Opin. Genet. Dev. 10, 508–514 (2000).
Olson, J. M. & Hallahan, A. R. p38 MAP kinase: a convergence point in cancer therapy. Trends Mol. Med. 10, 125–129 (2004).
Ivaska, J., Whelan, R. D., Watson, R. & Parker, P. J. PKCɛ controls the traffic of β1 integrins in motile cells. EMBO J. 21, 3608–3619 (2002).
Acknowledgements
We thank Robert Messing for the initial characterization and constructs required to develop the chemical genetic approach for specific PKCɛ inhibition. We thank David Barford for providing purified GSK3β, Erik Sahai for cherry-RhoA, Andrey Shaw for DN-14-3-3, Alastair Aitken for GST–14-3-3-β and Myc–14-3-3-β, and Brenda Kostelecky for 14.3.3ζ. We would also like to acknowledge staff at the CRUK London Research Institute FACS laboratory and light microscopy facility. This work was supported by a British Heart Foundation Intermediate Research Fellowship, British Heart Foundation Project Grant and Cancer Research UK.
Author information
Authors and Affiliations
Contributions
A.T.S. carried out all experiments (except those depicted in Fig. 2b and Supplementary Information, Fig. S1c, which were performed by A.F.); A.T.S. jointly conceived the project (with P.J.P.) and wrote the manuscript with the help of P.J.P.; J.D. identified the Ser 368 autophosphorylation site and made the relevant antibodies and mutant constructs. A.J.C. helped in generating PKCɛ knockout MEFs expressing various PKCɛ mutants. M.S.M. provided the SR-p38 constructs and helpful advice. All authors contributed to editing of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures S1, S2, S3, S4, S5, S6, S7 and S8 (PDF 1750 kb)
Rights and permissions
About this article
Cite this article
Saurin, A., Durgan, J., Cameron, A. et al. The regulated assembly of a PKCɛ complex controls the completion of cytokinesis. Nat Cell Biol 10, 891–901 (2008). https://doi.org/10.1038/ncb1749
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1749
This article is cited by
-
The Cardiac Sarcomere and Cell Cycle
Current Cardiology Reports (2022)
-
A genetically-encoded crosslinker screen identifies SERBP1 as a PKCε substrate influencing translation and cell division
Nature Communications (2021)
-
Equivocal, explicit and emergent actions of PKC isoforms in cancer
Nature Reviews Cancer (2021)
-
The Aurora B specificity switch is required to protect from non-disjunction at the metaphase/anaphase transition
Nature Communications (2020)
-
Building bridges between chromosomes: novel insights into the abscission checkpoint
Cellular and Molecular Life Sciences (2019)