Abstract
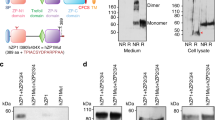
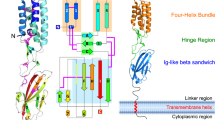
Many eukaryotic extracellular proteins share a sequence of unknown function, called the zona pellucida (ZP) domain1. Among these proteins are the mammalian sperm receptors ZP2 and ZP3, non-mammalian egg coat proteins, Tamm-Horsfall protein (THP), glycoprotein-2 (GP-2), α- and β-tectorins, transforming growth factor (TGF)-β receptor III and endoglin, DMBT-1 (deletd in malignant brain tumour-1), NompA (no-mechanoreceptor-potential-A), Dumpy and cuticlin-1 (refs 1,2). Here, we report that the ZP domain of ZP2, ZP3 and THP is responsible for polymerization of these proteins into filaments of similar supramolecular structure. Most ZP domain proteins are synthesized as precursors with carboxy-terminal transmembrane domains or glycosyl phosphatidylinositol (GPI) anchors1,2. Our results demonstrate that the C-terminal transmembrane domain and short cytoplasmic tail of ZP2 and ZP3 are not required for secretion, but are essential for assembly. Finally, we suggest a molecular basis for dominant human hearing disorders caused by point mutations within the ZP domain of α-tectorin3,4,5.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Bork, P. & Sander, C. FEBS Lett. 300, 237–240 (1992).
Wassarman, P. M., Jovine, L. & Litscher, E. S. Nature Cell Biol. 3, E59–E64 (2001).
Moreno-Pelayo, M. A. et al. J. Med. Genet. 38, E13–E16 (2001).
Verhoeven, K. et al. Nature Genet. 19, 60–62 (1998).
Kirschhofer, K. et al. Cytogenet. Cell Genet. 82, 126–130 (1998).
Wassarman, P. M. Annu. Rev. Biochem. 57, 415–442 (1988).
Wassarman, P. M. & Mortillo, S. Int. Rev. Cytol. 130, 85–110 (1991).
Tsubamoto, H. et al. Biol. Reprod. 61, 1649–1654 (1999).
Qi, H., Williams, Z. & Wassarman, P. M. Mol. Biol. Cell 13, 530–541 (2002).
Ponting, C. P., Schultz, J., Milpetz, F. & Bork, P. Nucleic Acids Res. 27, 229–232 (1999).
Tamm, I. & Horsfall, F. L. Jr. Proc. Soc. Exp. Biol. Med. 74, 108–114 (1950).
Kokot, F. & Dulawa, J. Nephron 85, 97–102 (2000).
Maxfield, M. & Davis, M. S. Ann. NY Acad. Sci. 106, 288–297 (1963).
Easton, R. L., Patankar, M. S., Clark, G. F., Morris, H. R. & Dell, A. J. Biol. Chem. 275, 21928–21938 (2000).
Bayer, M. E. J. Cell Biol. 21, 265–274 (1964).
Sawada, H. et al. Proc. Natl Acad. Sci. USA 99, 1223–1228 (2002).
Litscher, E. S., Qi, H. & Wassarman, P. M. Biochemistry 38, 12280–12287 (1999).
Cavallone, D., Malagolini, N. & Serafini-Cessi, F. Biochem. Biophys. Res. Commun. 280, 110–114 (2001).
Killick, R., Legan, P. K., Malenczak, C. & Richardson, G. P. J. Cell Biol. 129, 535–547 (1995).
Harris, J., Seid, C., Fontenot, G. & Liu, H. Protein Expr. Purif. 16, 298–307 (1999).
Harrison, P. T. Mol. Membr. Biol. 13, 67–79 (1996).
Sugiyama, H., Murata, K., Iuchi, I., Nomura, K. & Yamagami, K. J. Biochem. (Tokyo) 125, 469–475 (1999).
Hyllner, S. J., Westerlund, L., Olsson, P. E. & Schopen, A. Biol. Reprod. 64, 805–811 (2001).
Steel, K. P. & Kros, C. J. Nature Genet. 27, 143–149 (2001).
Legan, P. K., Rau, A., Keen, J. N. & Richardson, G. P. J. Biol. Chem. 272, 8791–8801 (1997).
Legan, P. K. et al. Neuron 28, 273–285 (2000).
Knipper, M. et al. J. Biol. Chem. 276, 39046–39052 (2001).
Serafini-Cessi, F., Bellabarba, G., Malagolini, N. & Dall'Olio, F. J. Immunol. Methods 120, 185–189 (1989).
Karp, D. R., Atkinson, J. P. & Shreffler, D. C. J. Biol. Chem. 257, 7330–7335 (1982).
Heuser, J. J. Electron Microsc. Tech. 13, 244–263 (1989).
Acknowledgements
We thank F. Serafini-Cessi for advice on THP purification, W.G.M. Janssen for help with EM analysis of THP and J.E. Heuser for imaging of mouse ZP filaments. We are also grateful to R. De Santis for communicating results before publication and to F. Cotelli, M. Paterlini and V. Gusarova for discussion. Confocal laser scanning microscopy was performed at the MSSM-Microscopy Shared Resource Facility; the N-terminal sequence of THP-ZPD was determined at the HHMI/Columbia University Protein Chemistry Core Facility. L.J. is supported by a Human Frontier Science Program long-term fellowship. This research was supported in part by National Institutes of Health grant HD35105.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Jovine, L., Qi, H., Williams, Z. et al. The ZP domain is a conserved module for polymerization of extracellular proteins. Nat Cell Biol 4, 457–461 (2002). https://doi.org/10.1038/ncb802
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb802
This article is cited by
-
UMOD gene mutations in Chinese patients with autosomal dominant tubulointerstitial kidney disease: a pediatric case report and literature review
BMC Pediatrics (2019)
-
Hepsin-mediated Processing of Uromodulin is Crucial for Salt-sensitivity and Thick Ascending Limb Homeostasis
Scientific Reports (2019)
-
Efficient production of human zona pellucida-3 in a prokaryotic expression system
World Journal of Microbiology and Biotechnology (2018)
-
Uromodulin: from physiology to rare and complex kidney disorders
Nature Reviews Nephrology (2017)
-
The scavenging capacity of DMBT1 is impaired by germline deletions
Immunogenetics (2017)