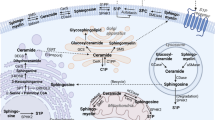
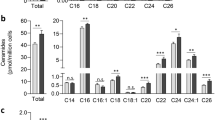
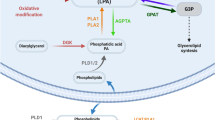
Abstract
Sphingolipids comprise a complex family of naturally occurring molecules that are enriched in lipid rafts and contribute to their unique biochemical properties. Membrane sphingolipids also serve as a reservoir for bioactive metabolites including sphingosine, ceramide, sphingosine-1-phosphate and ceramide-1-phosphate. Among these, sphingosine-1-phosphate has emerged as a central regulator of mammalian biology. Sphingosine-1-phosphate is essential for mammalian brain and cardiac development and for maturation of the systemic circulatory system and lymphatics. In addition, sphingosine-1-phosphate contributes to trafficking and effector functions of lymphocytes and other hematopoietic cells and protects against various forms of tissue injury. However, sphingosine-1-phosphate is also an oncogenic lipid that promotes tumor growth and progression. Recent preclinical and clinical investigations using pharmacological agents that target sphingosine-1-phosphate, its receptors and the enzymes required for its biosynthesis and degradation demonstrate the promise and potential risks of modulating sphingosine-1-phosphate signaling in treatment strategies for autoimmunity, cancer, cardiovascular disease and other pathological conditions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
20 July 2010
In the version of this article initially published, the DNA molecule and the SphK2 protein shape were missing from Figure 3. The errors have been corrected in the HTML and PDF versions of the article.
References
Holthuis, J.C., Pomorski, T., Raggers, R.J., Sprong, H. & Van Meer, G. The organizing potential of sphingolipids in intracellular membrane transport. Physiol. Rev. 81, 1689–1723 (2001).
Merrill, A.H.J. & Sandhoff, K. Sphingolipids: metabolism and cell signaling. in Biochemistry of Lipids, Lipoproteins and Membranes (eds. D.E. Vance & J.E. Vance) 373–407 (Elsevier, 2002).
Mandala, S. et al. Alteration of lymphocyte trafficking by sphingosine-1-phosphate receptor agonists. Science 296, 346–349 (2002).
Brinkmann, V. et al. The immune modulator FTY720 targets sphingosine 1-phosphate receptors. J. Biol. Chem. 277, 21453–21457 (2002).
Lahiri, S. & Futerman, A. The metabolism and function of sphingolipids and glycosphingolipids. Cell. Mol. Life Sci. 64, 2270–2284 (2007).
Hannun, Y.A. & Obeid, L.M. Principles of bioactive lipid signalling: lessons from sphingolipids. Nat. Rev. Mol. Cell Biol. 9, 139–150 (2008).
Zheng, W. et al. Ceramides and other bioactive sphingolipid backbones in health and disease: lipidomic analysis, metabolism and roles in membrane structure, dynamics, signaling and autophagy. Biochim. Biophys. Acta 1758, 1864–1884 (2006).
Han, G. et al. Identification of small subunits of mammalian serine palmitoyltransferase that confer distinct acyl-CoA substrate specificities. Proc. Natl. Acad. Sci. USA 106, 8186–8191 (2009).
Breslow, D.K. et al. Orm family proteins mediate sphingolipid homeostasis. Nature 463, 1048–1053 (2010).
Chalfant, C.E. & Spiegel, S. Sphingosine 1-phosphate and ceramide 1-phosphate: expanding roles in cell signaling. J. Cell Sci. 118, 4605–4612 (2005).
Shida, D., Takabe, K., Kapitonov, D., Milstien, S. & Spiegel, S. Targeting SphK1 as a new strategy against cancer. Curr. Drug Targets 9, 662–673 (2008).
Pyne, S., Lee, S.C., Long, J. & Pyne, N.J. Role of sphingosine kinases and lipid phosphate phosphatases in regulating spatial sphingosine 1-phosphate signalling in health and disease. Cell. Signal. 21, 14–21 (2009).
Alemany, R., van Koppen, C.J., Danneberg, K., Ter Braak, M. & Meyer Zu Heringdorf, D. Regulation and functional roles of sphingosine kinases. Naunyn Schmiedebergs Arch. Pharmacol. 374, 413–428 (2007).
Mizugishi, K. et al. Essential role for sphingosine kinases in neural and vascular development. Mol. Cell. Biol. 25, 11113–11121 (2005).
Gillies, L. et al. The sphingosine 1-phosphate receptor 5 and sphingosine kinases 1 and 2 are localised in centrosomes: possible role in regulating cell division. Cell. Signal. 21, 675–684 (2009).
Jarman, K.E., Moretti, P.A., Zebol, J.R. & Pitson, S.M. Translocation of sphingosine kinase 1 to the plasma membrane is mediated by calcium- and integrin-binding protein 1. J. Biol. Chem. 285, 483–492 (2010).
Mizugishi, K. et al. Maternal disturbance in activated sphingolipid metabolism causes pregnancy loss in mice. J. Clin. Invest. 117, 2993–3006 (2007).
Hait, N.C. et al. Regulation of histone acetylation in the nucleus by sphingosine-1-phosphate. Science 325, 1254–1257 (2009).
Weigert, A. et al. Sphingosine kinase 2 deficient tumor xenografts show impaired growth and fail to polarize macrophages towards an anti-inflammatory phenotype. Int. J. Cancer 125, 2114–2121 (2009).
Lai, W.Q. et al. Distinct roles of sphingosine kinase 1 and 2 in murine collagen-induced arthritis. J. Immunol. 183, 2097–2103 (2009).
Wacker, B.K., Park, T.S. & Gidday, J.M. Hypoxic preconditioning-induced cerebral ischemic tolerance: role of microvascular sphingosine kinase 2. Stroke 40, 3342–3348 (2009).
Liu, H. et al. Sphingosine kinase type 2 is a putative BH3-Only protein that induces apoptosis. J. Biol. Chem. 278, 40330–40336 (2003).
Weigert, A. et al. Cleavage of sphingosine kinase 2 by caspase-1 provokes its release from apoptotic cells. Blood 115, 3531–3540 (2010).
Don, A.S. & Rosen, H. A lipid binding domain in sphingosine kinase 2. Biochem. Biophys. Res. Commun. 380, 87–92 (2009).
Samy, E.T. et al. Cutting edge: Modulation of intestinal autoimmunity and IL-2 signaling by sphingosine kinase 2 independent of sphingosine 1-phosphate. J. Immunol. 179, 5644–5648 (2007).
Pappu, R. et al. Promotion of lymphocyte egress into blood and lymph by distinct sources of sphingosine-1-phosphate. Science 316, 295–298 (2007).
Sigal, Y.J., McDermott, M.I. & Morris, A.J. Integral membrane lipid phosphatases/phosphotransferases: common structure and diverse functions. Biochem. J. 387, 281–293 (2005).
Brindley, D.N. & Pilquil, C. Lipid phosphate phosphatases and signaling. J. Lipid Res. 50 Suppl, S225–S230 (2009).
Kirby, R.J. et al. Dynamic regulation of sphingosine-1-phosphate homeostasis during development of mouse metanephric kidney. Am. J. Physiol. Renal Physiol. 296, F634–F641 (2009).
Giussani, P. et al. Sphingosine-1-phosphate phosphohydrolase regulates endoplasmic reticulum-to-golgi trafficking of ceramide. Mol. Cell. Biol. 26, 5055–5069 (2006).
Peter, B.F. et al. Role of sphingosine-1-phosphate phosphohydrolase 1 in the regulation of resistance artery tone. Circ. Res. 103, 315–324 (2008).
Mechtcheriakova, D. et al. FTY720-phosphate is dephosphorylated by lipid phosphate phosphatase 3. FEBS Lett. 581, 3063–3068 (2007).
Serra, M. & Saba, J.D. Sphingosine 1-phosphate lyase, a key regulator of sphingosine 1-phosphate signaling and function. Adv. Enzyme Regul. 50, 349–362 (2010).
Zhan, X. & Desiderio, D.M. Nitroproteins from a human pituitary adenoma tissue discovered with a nitrotyrosine affinity column and tandem mass spectrometry. Anal. Biochem. 354, 279–289 (2006).
Mukhopadhyay, D., Howell, K.S., Riezman, H. & Capitani, G. Identifying key residues of sphinganine-1-phosphate lyase for function in vivo and in vitro. J. Biol. Chem. 283, 20159–20169 (2008).
Vogel, P. et al. Incomplete inhibition of sphingosine 1-phosphate lyase modulates immune system function yet prevents early lethality and non-lymphoid lesions. PLoS ONE 4, e4112 (2009).
Hagen, N. et al. Subcellular origin of sphingosine-1-phosphate is essential for its toxic effect in lyase deficient neurons. J. Biol. Chem. 284, 11346–11353 (2009).
Bektas, M. et al. Sphingosine-1-phosphate lyase deficiency disrupts lipid homeostasis in liver. J. Biol. Chem. 285, 10880–10889 (2010).
Dobrosotskaya, I.Y., Seegmiller, A., Brown, M., Goldstein, J. & Rawson, R. Regulation of SREBP processing and membrane lipid production by phospholipids in Drosophila. Science 296, 879–883 (2002).
Stratford, S., Hoehn, K., Liu, F. & Summers, S. Regulation of insulin action by ceramide: dual mechanisms linking ceramide accumulation to the inhibition of Akt/protein kinase B. J. Biol. Chem. 279, 36608–36615 (2004).
Zhang, K. et al. Redirection of sphingolipid metabolism towards de novo synthesis of ethanolamine in Leishmania. EMBO J. 26, 1094–1104 (2007).
Bourquin, F., Riezman, H., Capitani, G. & Gerhard, M. Structure (in press).
Kono, M., Allende, M.L. & Proia, R.L. Sphingosine-1-phosphate regulation of mammalian development. Biochim. Biophys. Acta 1781, 435–441 (2008).
Choi, J.W., Lee, C.W. & Chun, J. Biological roles of lysophospholipid receptors revealed by genetic null mice: an update. Biochim. Biophys. Acta 1781, 531–539 (2008).
Skoura, A. & Hla, T. Lysophospholipid receptors in vertebrate development, physiology, and pathology. J. Lipid Res. 50 Suppl, S293–S298 (2009).
Strochlic, L., Dwivedy, A., van Horck, F.P., Falk, J. & Holt, C.E. A role for S1P signalling in axon guidance in the Xenopus visual system. Development 135, 333–342 (2008).
Miron, V.E. et al. Fingolimod (FTY720) enhances remyelination following demyelination of organotypic cerebellar slices. Am. J. Pathol. doi:10.2353/ajpath.2010.091234 (22 April 2010).
Olivera, A. et al. Sphingosine kinase 1 and sphingosine-1-phosphate receptor 2 are vital to recovery from anaphylactic shock in mice. J. Clin. Invest. 120, 1429–1440 (2010).
Pébay, A. et al. Essential roles of sphingosine-1-phosphate and platelet-derived growth factor in the maintenance of human embryonic stem cells. Stem Cells 23, 1541–1548 (2005).
Bai, A., Hu, H., Yeung, M. & Chen, J. Kruppel-like factor 2 controls T cell trafficking by activating L-selectin (CD62L) and sphingosine-1-phosphate receptor 1 transcription. J. Immunol. 178, 7632–7639 (2007).
Jenne, C.N. et al. T-bet-dependent S1P5 expression in NK cells promotes egress from lymph nodes and bone marrow. J. Exp. Med. 206, 2469–2481 (2009).
Sanchez, T. & Hla, T. Structural and functional characteristics of S1P receptors. J. Cell. Biochem. 92, 913–922 (2004).
Rosen, H., Gonzalez-Cabrera, P.J., Sanna, M.G. & Brown, S. Sphingosine 1-phosphate receptor signaling. Annu. Rev. Biochem. 78, 743–768 (2009).
Mitra, P. et al. Role of ABCC1 in export of sphingosine-1-phosphate from mast cells. Proc. Natl. Acad. Sci. USA 103, 16394–16399 (2006).
Kobayashi, N., Yamaguchi, A. & Nishi, T. Characterization of the ATP-dependent sphingosine 1-phosphate transporter in rat erythrocytes. J. Biol. Chem. 284, 21192–21200 (2009).
Kupperman, E., An, S., Osborne, N., Waldron, S. & Stainier, D.Y. A sphingosine-1-phosphate receptor regulates cell migration during vertebrate heart development. Nature 406, 192–195 (2000).
Kawahara, A. et al. The sphingolipid transporter spns2 functions in migration of zebrafish myocardial precursors. Science 323, 524–527 (2009).
Nakano, Y. et al. Mutations in the novel membrane protein spinster interfere with programmed cell death and cause neural degeneration in Drosophila melanogaster. Mol. Cell. Biol. 21, 3775–3788 (2001).
Hinkovska-Galcheva, V., VanWay, S.M., Shanley, T.P. & Kunkel, R.G. The role of sphingosine-1-phosphate and ceramide-1-phosphate in calcium homeostasis. Curr. Opin. Investig. Drugs 9, 1192–1205 (2008).
Estrada, R. et al. Ligand-induced nuclear translocation of S1P(1) receptors mediates Cyr61 and CTGF transcription in endothelial cells. Histochem. Cell Biol. 131, 239–249 (2009).
Liao, J.J. et al. Distinctive T cell-suppressive signals from nuclearized type 1 sphingosine 1-phosphate G protein-coupled receptors. J. Biol. Chem. 282, 1964–1972 (2007).
Adachi-Yamada, T. et al. De novo synthesis of sphingolipids is required for cell survival by down-regulating c-Jun N-terminal kinase in Drosophila imaginal discs. Mol. Cell. Biol. 19, 7276–7286 (1999).
Dasgupta, U. et al. Ceramide kinase regulates phospholipase C and phosphatidylinositol 4, 5, bisphosphate in phototransduction. Proc. Natl. Acad. Sci. USA 106, 20063–20068 (2009).
Schmahl, J., Rizzolo, K. & Soriano, P. The PDGF signaling pathway controls multiple steroid-producing lineages. Genes Dev. 22, 3255–3267 (2008).
Zammit, P.S., Partridge, T.A. & Yablonka-Reuveni, Z. The skeletal muscle satellite cell: the stem cell that came in from the cold. J. Histochem. Cytochem. 54, 1177–1191 (2006).
Matloubian, M. et al. Lymphocyte egress from thymus and peripheral lymphoid organs is dependent on S1P receptor 1. Nature 427, 355–360 (2004).
Kharel, Y. et al. Sphingosine kinase 2 is required for modulation of lympocyte traffic by FTY720 J Biol. Chem. 280, 36865–36872 (2005).
Rivera, J., Proia, R.L. & Olivera, A. The alliance of sphingosine-1-phosphate and its receptors in immunity. Nat. Rev. Immunol. 8, 753–763 (2008).
Shimizu, T. Lipid mediators in health and disease: enzymes and receptors as therapeutic targets for the regulation of immunity and inflammation. Annu. Rev. Pharmacol. Toxicol. 49, 123–150 (2009).
Schwab, S.R. & Cyster, J.G. Finding a way out: lymphocyte egress from lymphoid organs. Nat. Immunol. 8, 1295–1301 (2007).
Zachariah, M.A. & Cyster, J.G. Neural crest-derived pericytes promote egress of mature thymocytes at the corticomedullary junction. Science doi:10.1126/science.1188222 (22 April 2010).
Kappos, L. et al. A placebo-controlled trial of oral fingolimod in relapsing multiple sclerosis. N. Engl. J. Med. 362, 387–401 (2010).
Cohen, J.A. et al. Oral fingolimod or intramuscular interferon for relapsing multiple sclerosis. N. Engl. J. Med. 362, 402–415 (2010).
Ratajczak, M.Z. et al. Novel insight into stem cell mobilization-Plasma sphingosine-1-phosphate is a major chemoattractant that directs the egress of hematopoietic stem progenitor cells from the bone marrow and its level in peripheral blood increases during mobilization due to activation of complement cascade/membrane attack complex. Leukemia 24, 976–985 (2010).
Gossens, K. et al. Thymic progenitor homing and lymphocyte homeostasis are linked via S1P-controlled expression of thymic P-selectin/CCL25. J. Exp. Med. 206, 761–778 (2009).
Weber, C. K.A., Münk, A., Bode, C., Van Veldhoven, P.P. & Gräler, M.H. Discontinued postnatal thymocyte development in sphingosine 1-phosphate-lyase-deficient mice. J. Immunol. 183, 4292–4301 (2009).
Ishii, M. et al. Sphingosine-1-phosphate mobilizes osteoclast precursors and regulates bone homeostasis. Nature 458, 524–528 (2009).
Pereira, J.P., Xu, Y. & Cyster, J.G. A role for S1P and S1P1 in immature-B cell egress from mouse bone marrow. PLoS ONE 5, e9277 (2010).
Allende, M.L. et al. S1P1 receptor directs the release of immature B cells from bone marrow into blood. J. Exp. Med. doi: 10.1084/jem.20092210 (19 April 2010).
Wolf, A.M. et al. The sphingosine 1-phosphate receptor agonist FTY720 potently inhibits regulatory T cell proliferation in vitro and in vivo. J. Immunol. 183, 3751–3760 (2009).
Michaud, J., Im, D. & Hla, T. Inhibitory role of sphingosine 1-phosphate receptor 2 in macrophage recruitment during inflammation. J. Immunol. 184, 1475–1483 (2010).
Means, C.K. et al. Sphingosine 1-phosphate S1P2 and S1P3 receptor-mediated Akt activation protects against in vivo myocardial ischemia-reperfusion injury. Am. J. Physiol. Heart Circ. Physiol. 292, H2944–H2951 (2007).
Hasegawa, Y., Suzuki, H., Sozen, T., Rolland, W. & Zhang, J.H. Activation of sphingosine 1-phosphate receptor-1 by FTY720 is neuroprotective after ischemic stroke in rats. Stroke 41, 368–374 (2010).
Morita, Y. et al. Oocyte apoptosis is suppressed by disruption of the acid sphingomyelinase gene or by sphingosine-1-phosphate therapy. Nat. Med. 6, 1109–1114 (2000).
Sattler, K. & Levkau, B. Sphingosine-1-phosphate as a mediator of high-density lipoprotein effects in cardiovascular protection. Cardiovasc. Res. 82, 201–211 (2009).
Xia, P. et al. An oncogenic role of sphingosine kinase. Curr. Biol. 10, 1527–1530 (2000).
Visentin, B. et al. Validation of an anti-sphingosine-1-phosphate antibody as a potential therapeutic in reducing growth, invasion, and angiogenesis in multiple tumor lineages. Cancer Cell 9, 225–238 (2006).
Oskouian, B. et al. Sphingosine-1-phosphate lyase potentiates apoptosis via p53- and p38-dependent pathways and is downregulated in colon cancer. Proc. Natl. Acad. Sci. USA 103, 17384–17389 (2006).
Colié, S. et al. Disruption of sphingosine 1-phosphate lyase confers resistance to chemotherapy and promotes oncogenesis through Bcl-2/Bcl-xL upregulation. Cancer Res. 69, 9346–9353 (2009).
Cattoretti, G. et al. Targeted disruption of the S1P2 sphingosine 1-phosphate receptor gene leads to diffuse large B-cell lymphoma formation. Cancer Res. 69, 8686–8692 (2009).
Levi, M., Meijler, M.M., Gomez-Munoz, A. & Zor, T. Distinct receptor-mediated activities in macrophages for natural ceramide-1-phosphate (C1P) and for phospho-ceramide analogue-1 (PCERA-1). Mol. Cell. Endocrinol. 314, 248–255 (2010).
Wijesinghe, D.S. et al. Substrate specificity of human ceramide kinase. J. Lipid Res. 46, 2706–2716 (2005).
Granado, M.H. et al. Ceramide 1-phosphate (C1P) promotes cell migration Involvement of a specific C1P receptor. Cell. Signal. 21, 405–412 (2009).
Lankalapalli, R.S., Ouro, A., Arana, L., Gomez-Munoz, A. & Bittman, R. Caged ceramide 1-phosphate analogues: synthesis and properties. J. Org. Chem. 74, 8844–8847 (2009).
Vacaru, A.M. et al. Sphingomyelin synthase-related protein SMSr controls ceramide homeostasis in the ER. J. Cell Biol. 185, 1013–1027 (2009).
Ternes, P., Brouwers, J.F., van den Dikkenberg, J. & Holthuis, J.C. Sphingomyelin synthase SMS2 displays dual activity as ceramide phosphoethanolamine synthase. J. Lipid Res. 50, 2270–2277 (2009).
Eichler, F.S. et al. Overexpression of the wild-type SPT1 subunit lowers desoxysphingolipid levels and rescues the phenotype of HSAN1. J. Neurosci. 29, 14646–14651 (2009).
Symolon, H., Schmelz, E., Dillehay, D. & Merrill, A.J. Dietary soy sphingolipids suppress tumorigenesis and gene expression in 1,2-dimethylhydrazine-treated CF1 mice and ApcMin/+ mice. J. Nutr. 134, 1157–1161 (2004).
Fyrst, H. et al. Natural sphingadienes inhibit Akt-dependent signaling and prevent intestinal tumorigenesis. Cancer Res. 69, 9457–9464 (2009).
Yamaji, T., Kumagai, K., Tomishige, N. & Hanada, K. Two sphingolipid transfer proteins, CERT and FAPP2: their roles in sphingolipid metabolism. IUBMB Life 60, 511–518 (2008).
Mao, C. & Obeid, L.M. Ceramidases: regulators of cellular responses mediated by ceramide, sphingosine, and sphingosine-1-phosphate. Biochim. Biophys. Acta 1781, 424–434 (2008).
Acknowledgements
This work was supported by US National Institutes of Health Public Health Service grants CA77528, CA129438, RAT005336 and GM66954 (J.D.S.). We thank B. Oskouian for careful review of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Fyrst, H., Saba, J. An update on sphingosine-1-phosphate and other sphingolipid mediators. Nat Chem Biol 6, 489–497 (2010). https://doi.org/10.1038/nchembio.392
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.392
This article is cited by
-
A comprehensive multiomics approach reveals that high levels of sphingolipids in cardiac cachexia adipose tissue are associated with inflammatory and fibrotic changes
Lipids in Health and Disease (2023)
-
Pathogenic sphingosine 1-phosphate pathway in psoriasis: a critical review of its pathogenic significance and potential as a therapeutic target
Lipids in Health and Disease (2023)
-
Targeting SPHK1/S1PR3-regulated S-1-P metabolic disorder triggers autophagic cell death in pulmonary lymphangiomyomatosis (LAM)
Cell Death & Disease (2022)
-
The Golgi-localized sphingosine-1-phosphate phosphatase is indispensable for Leishmania major
Scientific Reports (2022)
-
Lipidomics Analysis Reveals a Protective Effect of Myriocin on Cerebral Ischemia/Reperfusion Model Rats
Journal of Molecular Neuroscience (2022)