Abstract
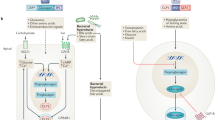
Up to a third of people with diabetes mellitus suffer end-stage renal failure due to diabetic nephropathy. Strategies to delay progression of diabetic nephropathy—including glycemic and blood pressure control, modification of the renin–angiotensin system and management of lipid levels with statins—have been effective, but development of new strategies is essential if the ever-increasing burden of this disease is to be minimized. Thiazolidinediones (TZDs) are a family of compounds used as oral hypoglycemic agents in patients with type 2 diabetes mellitus. The therapeutic effects of TZDs are largely a function of their activity as ligands of peroxisome proliferator-activated receptor gamma (PPARγ), a transcription factor that has a central role in adipogenesis and insulin sensitization. In vitro animal and clinical studies have shown that TZDs ameliorate symptoms and pathogenic mechanisms of diabetic and nondiabetic nephropathy, including proteinuria, excessive deposition of glomerular matrix, cellular proliferation, inflammation and fibrosis. Many of these favorable effects occur under both normal and high-glucose conditions. The mechanisms responsible probably involve both PPARγ-dependent and PPARγ-independent pathways. So, TZDs and other agonists of PPARγ offer promise for treatment of diabetic nephropathy; however, before their putative renoprotective effects can be translated into clinical practice, the complex mechanisms of PPARγ activity and regulation will need to be investigated further.
Key Points
-
Thiazolidinediones (TZDs) are agonists of peroxisome proliferator-activated receptors (PPARs)
-
PPARs are expressed in the kidney, and have prominent roles in insulin sensitization and adipogenesis
-
TZDs (e.g. pioglitazone and rosiglitazone) are used as oral hypoglycemic agents in type 2 diabetes mellitus
-
Agonists of PPARγ, including TZDs, ameliorate tubulointerstitial fibrosis, albuminuria and glomerulosclerosis in models of diabetic and nondiabetic nephropathy
-
Early trials in humans with diabetic nephropathy have shown that TZDs ameliorate albuminuria and confer cardiovascular benefits
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
US Renal Data System (2003) Annual Data Report: Atlas of End-Stage Renal Disease in the United States. Bethesda: National Institute of Diabetes and Digestive and Kidney Diseases
Chuahirun T et al. (2004) Cigarette smoking exacerbates and its cessation ameliorates renal injury in type 2 diabetes. Am J Med Sci 327: 57–67
Harris KP et al. (1990) Lovastatin ameliorates the development of glomerulosclerosis and uremia in experimental nephrotic syndrome. Am J Kidney Dis 15: 16–23
Athyros VG et al. (2004) The effect of statins versus untreated dyslipidaemia on renal function in patients with coronary heart disease. A subgroup analysis of the Greek atorvastatin and coronary heart disease evaluation (GREACE) study. J Clin Pathol 57: 728–734
Collins R et al. (2003) MRC/BHF Heart Protection Study of cholesterol-lowering with simvastatin in 5963 people with diabetes: a randomised placebo-controlled trial. Lancet 361: 2005–2016
Ruggenenti P et al. (2000) Nephropathy of type 1 and type 2 diabetes: diverse pathophysiology, same treatment? Nephrol Dial Transplant 15: 1900–1902
Nath KA (1992) Tubulointerstitial changes as a major determinant in the progression of renal damage. Am J Kidney Dis 20: 1–17
Brownlee M (2001) Biochemistry and molecular cell biology of diabetic complications. Nature 414: 813–820
Qi W et al. (2005) Integrated actions of transforming growth factor-beta1 and connective tissue growth factor in renal fibrosis. Am J Physiol Renal Physiol 288: F800–F809
Hoffman BB et al. (1998) Transcriptional activation of transforming growth factor-beta1 in mesangial cell culture by high glucose concentration. Kidney Int 54: 1107–1116
Ziyadeh FN et al. (1998) Glycated albumin stimulates fibronectin gene expression in glomerular mesangial cells: involvement of the transforming growth factor-beta system. Kidney Int 53: 631–638
Riser BL et al. (1996) Cyclic stretching force selectively up-regulates transforming growth factor-beta isoforms in cultured rat mesangial cells. Am J Pathol 148: 1915–1923
Kolm-Litty V et al. (1998) High glucose-induced transforming growth factor beta1 production is mediated by the hexosamine pathway in porcine glomerular mesangial cells. J Clin Invest 101: 160–169
Wolf G et al. (1997) The role of angiotensin II in diabetic nephropathy: emphasis on nonhemodynamic mechanisms. Am J Kidney Dis 29: 153–163
Nakamura T et al. (1995) Effect of a specific endothelin receptor A antagonist on mRNA levels for extracellular matrix components and growth factors in diabetic glomeruli. Diabetes 44: 895–899
Studer RK et al. (1995) Protein kinase C signals thromboxane induced increases in fibronectin synthesis and TGF-beta bioactivity in mesangial cells. Kidney Int 48: 422–430
Issemann I et al. (1990) Activation of a member of the steroid hormone receptor superfamily by peroxisome proliferators. Nature 347: 645–650
Auboeuf D et al. (1997) Tissue distribution and quantification of the expression of mRNAs of peroxisome proliferator-activated receptors and liver X receptor-alpha in humans: no alteration in adipose tissue of obese and NIDDM patients. Diabetes 46: 1319–1327
Staels B et al. (1998) Mechanism of action of fibrates on lipid and lipoprotein metabolism. Circulation 98: 2088–2093
Fajas L et al. (1998) PPARγ3 mRNA: a distinct PPARγ mRNA subtype transcribed from an independent promoter. FEBS Lett 438: 55–60
Fajas L et al. (1997) The organization, promoter analysis, and expression of the human PPARγ gene. J Biol Chem 272: 18779–18789
Guan Y et al. (1997) Expression of peroxisome proliferator-activated receptors in urinary tract of rabbits and humans. Am J Physiol Renal Physiol 273: F1013–F1022
Escher P et al. (2001) PPARs: Quantitative analysis in adult rat tissues and regulation in fasting and refeeding. Endocrinology 142: 4195–4202
Guan Y and Breyer MD (2001) Peroxisome proliferator-activated receptors (PPARs): Novel therapeutic targets in renal disease. Kidney Int 60: 14–30
Hao C-M et al. (2002) Peroxisome proliferator-activated receptor δ activation promotes cell survival following hypertonic stress. J Biol Chem 277: 21341–21345
Panchapakesan U et al. (2004) The effect of high glucose and PPAR-γ agonists on PPAR-γ expression and function in HK-2 cells. Am J Physiol Renal Physiol 287: F528–F534
Zafiriou S et al. (2004) Pioglitazone increases renal tubular cell albumin uptake but limits proinflammatory and fibrotic responses. Kidney Int 65: 1647–1653
Zafiriou S et al. (2005) Pioglitazone inhibits cell growth and reduces matrix production in the human kidney fibroblasts. J Am Soc Nephrol 16: 638–645
Arici M et al. (2003) Stimulation of proximal tubular cell apoptosis by albumin-bound fatty acids mediated by peroxisome proliferator activated receptor-γ. J Am Soc Nephrol 14: 17–27
Chana RS et al. (2004) Differential effects of peroxisome proliferator activated receptor-γ (PPARγ) ligands in proximal tubular cells: Thiazolidinediones are partial PPARγ agonists. Kidney Int 65: 2081–2090
Miyata KS et al. (1994) The peroxisome proliferator-activated receptor interacts with the retinoid X receptor in vivo. Gene 148: 327–330
Mukherjee R et al. (1997) Sensitization of diabetic and obese mice to insulin by retinoid X receptor agonists. Nature 386: 407–410
Moras D et al. (1998) The nuclear receptor ligand-binding domain: structure and function. Curr Opin Cell Biol 10: 384–391
Forman BM et al. (1995) 15-Deoxy-delta12,14-prostaglandin J2 is a ligand for the adipocyte determination factor PPARγ. Cell 83: 803–812
Davies SS et al. (2001) Oxidized alkyl phospholipids are specific, high affinity peroxisome proliferator-activated receptor γ ligands and agonists. J Biol Chem 276: 16015–16023
Berger J et al. (1999) Novel peroxisome proliferator-activated receptor (PPAR) γ and PPARδ ligands produce distinct biological effects. J Biol Chem 274: 6718–6725
Lehmann JM et al. (1997) Peroxisome proliferator-activated receptors α and γ are activated by indomethacin and other non-steroidal anti-inflammatory drugs. J Biol Chem 272: 3406–3410
Lebovitz HE et al. (2002) Evaluation of liver function in type 2 diabetic patients during clinical trials: evidence that rosiglitazone does not cause hepatic dysfunction. Diabetes Care 25: 815–821
Elbrecht A et al. (1996) Molecular cloning, expression and characterization of human peroxisome proliferator activated receptors γ1 and γ2. Biochem Biophys Res Commun 224: 431–437
Harris PK et al. (1994) Localization of a pioglitazone response element in the adipocyte fatty acid-binding protein gene. Mol Pharmacol 45: 439–445
Tontonoz P et al. (1994) mPPARγ2: tissue-specific regulator of an adipocyte enhancer. Genes Dev 8: 1224–1234
Lehmann JM et al. (1995) An antidiabetic thiazolidinedione is a high affinity ligand for peroxisome proliferator-activated receptor γ (PPARγ). J Biol Chem 270: 12953–12956
Gottlicher M et al. (1992) Fatty acids activate a chimera of the clofibric acid-activated receptor and the glucocorticoid receptor. Proc Natl Acad Sci USA 89: 4653–4657
Forman BM et al. (1997) Hypolipidemic drugs, polyunsaturated fatty acids, and eicosanoids are ligands for peroxisome proliferator-activated receptors α and δ. Proc Natl Acad Sci USA 94: 4312–4317
Oliver WR Jr et al. (2001) A selective peroxisome proliferator-activated receptor δ agonist promotes reverse cholesterol transport. Proc Natl Acad Sci USA 98: 5306–5311
Drumm K et al. (2003) Albumin and glucose effects on cell growth parameters, albumin uptake and Na+/H+-exchanger isoform 3 in OK cells. Cell Physiol Biochem 13: 199–206
Lee EM et al. (2003) Effects of pathophysiological concentrations of albumin on NHE3 activity and cell proliferation in primary cultures of human proximal tubule cells. Am J Physiol Renal Physiol 285: F748–F757
Panchapakesan U et al. (2005) PPARγ agonists exert antifibrotic effects in renal tubular cells exposed to high glucose. Am J Physiol Renal Physiol [10.1152/ajprenal.00097.2005]
Ghosh SS et al. (2003) PPARγ ligand attenuates PDGF-induced mesangial cell proliferation: role of MAP kinase. Kidney Int 64: 52–62
Routh RE et al. (2002) Troglitazone suppresses the secretion of type I collagen by mesangial cells in vitro. Kidney Int 61: 1365–1376
Weigert C et al. (2003) c-Fos-driven transcriptional activation of transforming growth factor β-1: inhibition of high glucose-induced promoter activity by thiazolidinediones. Biochem Biophys Res Commun 304: 301–307
Xiong Z et al. (2004) Anti-inflammatory effect of PPARγ in cultured human mesangial cells. Ren Fail 26: 497–505
Iwashima Y et al. (1999) Advanced glycation end product-induced peroxisome proliferator-activated receptor γ gene expression in the cultured mesangial cells. Biochem Biophys Res Commun 264: 441–448
Baylis C et al. (2003) Peroxisome proliferator-activated receptor γ agonist provides superior renal protection versus angiotensin-converting enzyme inhibition in a rat model of type 2 diabetes with obesity. J Pharmacol Exp Ther 307: 854–860
Haneda M et al. (2001) Cellular mechanisms in the development and progression of diabetic nephropathy: activation of the DAG–PKC–ERK pathway. Am J Kidney Dis 38: S178–S181
McCarthy KJ et al. (2000) Troglitazone halts diabetic glomerulosclerosis by blockade of mesangial expansion. Kidney Int 58: 2341–2350
Buckingham RE et al. (1998) Peroxisome proliferator-activated receptor-γ agonist, rosiglitazone, protects against nephropathy and pancreatic islet abnormalities in Zucker fatty rats. Diabetes 47: 1326–1334
Zhang Y et al. (2003) Endogenous PPARγ activity ameliorates diabetic nephropathy. J Am Soc Nephrol 14: 392A
Ma LJ et al. (2001) Peroxisome proliferator-activated receptor-γ agonist troglitazone protects against nondiabetic glomerulosclerosis in rats. Kidney Int 59: 1899–1910
Morii T et al. (2003) Association of monocyte chemoattractant protein-1 with renal tubular damage in diabetic nephropathy. J Diabetes Complications 17: 11–15
Haraguchi K et al. (2003) Suppression of experimental crescentic glomerulonephritis by peroxisome proliferator-activated receptor (PPAR) γ activators. Clin Exp Nephrol 7: 27–32
Panzer U et al. (2002) Effects of different PPARγ-agonists on MCP-1 expression and monocyte recruitment in experimental glomerulonephritis. Kidney Int 62: 455–464
[No authors listed] (2001) Effect of fenofibrate on progression of coronary-artery disease in type 2 diabetes: the Diabetes Atherosclerosis Intervention Study, a randomised study. Lancet 357: 905–910
Kim H et al. (2003) Peroxisome proliferator-activated receptor-α agonist treatment in a transgenic model of type 2 diabetes reverses the lipotoxic state and improves glucose homeostasis. Diabetes 52: 1770–1778
Koh EH et al. (2003) Peroxisome proliferator-activated receptor (PPAR)-α activation prevents diabetes in OLETF rats: comparison with PPAR-γ activation. Diabetes 52: 2331–2337
Park CW et al. (2003) A PPAR α agonist improves diabetic nephropathy in db/db mice. J Am Soc Nephrol 14: 393A
Leibowitz MD et al. (2000) Activation of PPARδ alters lipid metabolism in db/db mice. FEBS Lett 473: 333–336
Hegarty BD et al. (2004) Peroxisome proliferator-activated receptor (PPAR) activation induces tissue-specific effects on fatty acid uptake and metabolism in vivo—a study using the novel PPARα/γ agonist tesaglitazar. Endocrinology 145: 3158–3164
Etgen GJ et al. (2002) A tailored therapy for the metabolic syndrome: the dual peroxisome proliferator-activated receptor-α/γ agonist LY465608 ameliorates insulin resistance and diabetic hyperglycemia while improving cardiovascular risk factors in preclinical models. Diabetes 51: 1083–1087
Bakris G et al. (2003) Rosiglitazone reduces urinary albumin excretion in type II diabetes. J Hum Hypertens 17: 7–12
de Zeeuw D et al. (2004) Albuminuria, a therapeutic target for cardiovascular protection in type 2 diabetic patients with nephropathy. Circulation 110: 921–927
Song J et al. (2004) Rosiglitazone activates renal sodium- and water-reabsorptive pathways and lowers blood pressure in normal rats. J Pharm Exp Ther 308: 426–433
Wu L et al. (2004) Beneficial and deleterious effects of rosiglitazone on hypertension development in spontaneously hypertensive rats. Am J Hypertension 17: 749–756
Dobrian AD et al. (2004) Pioglitazone prevents hypertension and reduces oxidative stress in diet-induced obesity. Hypertension 43: 48–56
Czoski-Murray C et al. (2004) Clinical effectiveness and cost-effectiveness of pioglitazone and rosiglitazone in the treatment of type 2 diabetes: a systematic review and economic evaluation. Health Technol Assess 8: iii, ix–x, 1–91
Chiquette E et al. (2004) A meta-analysis comparing the effect of thiazolidinediones on cardiovascular risk factors. Arch Intern Med 164: 2097–2104
Tontonoz P et al. (1998) PPARγ promotes monocyte/macrophage differentiation and uptake of oxidized LDL. Cell 93: 241–252
Matsumoto K et al. (2000) Expression of macrophage (Mphi) scavenger receptor, CD36, in cultured human aortic smooth muscle cells in association with expression of peroxisome proliferator activated receptor-γ, which regulates gain of Mphi-like phenotype in vitro, and its implication in atherogenesis. Arterioscler Thromb Vasc Biol 20: 1027–1032
Chinetti G et al. (2001) PPAR-α and PPAR-γ activators induce cholesterol removal from human macrophage foam cells through stimulation of the ABCA1 pathway. Nat Med 7: 53–58
Li AC et al. (2000) Peroxisome proliferator activated receptor γ ligands inhibit development of atherosclerosis in LDL-receptor deficient mice. J Clin Invest 106: 523–531
Minamikawa J et al. (1998) Potent inhibitory effect of troglitazone on carotid arterial wall thickness in type 2 diabetes. J Clin Endocrinol Metab 83: 1818–1820
Voytovich MH et al. (2005) Short-term treatment with rosiglitazone improves glucose intolerance, insulin sensitivity and endothelial function in renal transplant recipients. Nephrol Dial Transplant 20: 413–418
Nesto RW et al. (2004) Thiazolidinedione use, fluid retention, and congestive heart failure: a consensus statement from the American Heart Association and American Diabetes Association. Diabetes Care 27: 256–263
Guan Y et al. (2005) Thiazolidinediones expand body fluid volume through PPARγ stimulation of ENaC-mediated renal salt absorption. Nat Med 11: 861–866
Zhang H et al. (2005) Collecting duct-specific deletion of peroxisome proliferator-activated receptor γ blocks thiazolidinedione-induced fluid retention. Proc Natl Acad Sci USA 102: 9406–9411
Hong G et al. (2003) PPARγ activation enhances cell surface ENaCα via up-regulation of SGK1 in human collecting duct cells. FASEB J 17: 1966–1968
Chen L et al. (2005) GI262570, a peroxisome proliferator-activated receptor γ agonist, changes electrolytes and water reabsorption from the distal nephron in rats. J Pharmacol Exp Ther 312: 718–725
Zanchi A et al. (2004) Effects of the peroxisomal proliferator-activated receptor-γ agonist pioglitazone on renal and hormonal responses to salt in healthy men. J Clin Endocrinol Metab 89: 1140–1145
Ristow M et al. (1998) Obesity associated with a mutation in a genetic regulator of adipocyte differentiation. N Engl J Med 339: 953–959
Hu E et al. (1996) Inhibition of adipogenesis through MAP kinase-mediated phosphorylation of PPARγ. Science 274: 2100–2103
Agarwal AK et al. (2002) A novel heterozygous mutation in peroxisome proliferator-activated receptor-γ gene in a patient with familial partial lipodystrophy. J Clin Endocrinol Metab 87: 408–411
Barroso I et al. (1999) Dominant negative mutations in human PPARγ associated with severe insulin resistance, diabetes mellitus and hypertension. Nature 402: 880–883
Yen CJ et al. (1997) Molecular scanning of the human peroxisome proliferator activated receptor γ (hPPARγ) gene in diabetic Caucasians: identification of a Pro12Ala PPARγ2 missense mutation. Biochem Biophys Res Commun 241: 270–274
Deeb SS et al. (1998) A Pro12Ala substitution in PPARγ2 associated with decreased receptor activity, lower body mass index and improved insulin sensitivity. Nat Genet 20: 284–287
Altshuler D et al. (2000) The common PPARγ Pro12Ala polymorphism is associated with decreased risk of type 2 diabetes. Nat Genet 26: 76–80
Caramori ML et al. (2003) The human peroxisome proliferator-activated receptor γ2 (PPARγ2) Pro12Ala polymorphism is associated with decreased risk of diabetic nephropathy in patients with type 2 diabetes. Diabetes 52: 3010–3013
Giri S et al. (2004) The 15-deoxy-δ12,14-prostaglandin J2 inhibits the inflammatory response in primary rat astrocytes via down-regulating multiple steps in phosphatidylinositol 3-kinase-Akt-NF-κB-p300 pathway independent of peroxisome proliferator-activated receptor γ. J Immunol 173: 5196–5208
Grau R et al. (2004) Inhibition of activator protein 1 activation, vascular endothelial growth factor, and cyclooxygenase-2 expression by 15-deoxy-δ12,14-prostaglandin J2 in colon carcinoma cells: evidence for a redox-sensitive peroxisome proliferator-activated receptor-γ-independent mechanism. Cancer Res 64: 5162–5171
Castrillo A et al. (2000) Inhibition of IκB kinase and IκB phosphorylation by 15-deoxy-Δ12,14-prostaglandin J2 in activated murine macrophages. Mol Cell Biol 20: 1692–1698
Ueta M et al. (2004) PPARγ ligands attenuate mesangial contractile dysfunction in high glucose. Kidney Int 65: 961–971
Portilla D et al. (2000) Etomoxir-induced PPARα-modulated enzymes protect during acute renal failure. Am J Physiol Renal Physiol 278: F667–F675
Staels B et al. (1998) Activation of human aortic smooth muscle cells is inhibited by PPARα but not by PPARα activators. Nature 393: 790–793
Rubins HB et al. (1999) Gemfibrozil for the secondary prevention of coronary heart disease in men with low levels of high-density lipoprotein cholesterol. Veterans Affairs High Density Lipoprotein Cholesterol Intervention Trial Study Group. N Engl J Med 341: 410–418
Nicholas SB et al. (2001) Expression and function of peroxisome proliferator-activated receptor-γ in mesangial cells. Hypertension 37: 722–727
Law RE et al. (2000) Expression and function of PPARγ in rat and human vascular smooth muscle cells. Circulation 101: 1311–1318
Asano T et al. (2000) Peroxisome proliferator-activated receptor γ1 (PPARγ1) expresses in rat mesangial cells and PPARγ agonists modulate its differentiation. Biochim Biophys Acta 1497: 148–154
Jiang C et al. (1998) PPARγ agonists inhibit production of monocyte inflammatory cytokines. Nature 391: 82–86
Acknowledgements
We thank E Kantzow for her assistance with the literature search. We acknowledge the support of the National Health and Medical Research Council of Australia, Juvenile Diabetes Research Foundation and Merck Laboratories for generously providing us with L805645.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Panchapakesan, U., Chen, XM. & Pollock, C. Drug Insight: thiazolidinediones and diabetic nephropathy—relevance to renoprotection. Nat Rev Nephrol 1, 33–43 (2005). https://doi.org/10.1038/ncpneph0029
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ncpneph0029
This article is cited by
-
The role of expression imbalance between adipose synthesis and storage mediated by PPAR-γ/FSP27 in the formation of insulin resistance in catch up growth
Lipids in Health and Disease (2016)
-
Preserved Na/HCO3 cotransporter sensitivity to insulin may promote hypertension in metabolic syndrome
Kidney International (2015)
-
Paediatrics, insulin resistance and the kidney
Pediatric Nephrology (2015)
-
Role of the EGF receptor in PPARγ-mediated sodium and water transport in human proximal tubule cells
Diabetologia (2013)
-
Inhibition of inflammation by pentosan polysulfate impedes the development and progression of severe diabetic nephropathy in aging C57B6 mice
Laboratory Investigation (2011)