Abstract
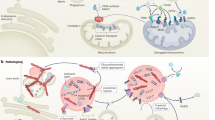
The identification of single genes linked to heritable forms of Parkinson disease (PD) has challenged the previously held view of a nongenetic etiology for this progressive movement disorder. Detailed analyses of individuals with mutations in SNCA, Parkin, PINK1, DJ1 or LRRK2 have greatly advanced our knowledge of preclinical and clinical, morphological, and pathological changes in PD. These genetic breakthroughs have had profound implications for scientists, neurologists and patients alike. Such advances have provided unique opportunities to pursue the mechanisms of neuronal degeneration in models of PD pathogenesis, thereby reinforcing the significance of oxidative stress and mitochondrial dysfunction. With emerging clues from familial variants, researchers have begun to explore factors that lead to the expression of the more common, sporadic disease phenotype (idiopathic PD), including interactions between various genes, modifying effects of susceptibility alleles and epigenetic factors, and the influence of environmental agents and aging on the expression of PD-linked genes. These genetic leads have added to the urgency of developing translational drug treatments, and neurologists and their patients are confronting considerations relating to DNA testing. In this article, we summarize recent progress in establishing a neurogenetic component of PD, emphasize the need for developing PD biomarkers to improve diagnostic accuracy (in both clinical practice and therapeutic trials), and discuss scenarios in which specific DNA tests might be considered for diagnostic purposes. In the absence of consensus guidelines for DNA testing in PD and of any neuroprotective treatment for this nonfatal disorder, we remind ourselves of the omnipresent mandate, 'Primum nil nocere!' ('First, do no harm!').
Key Points
-
Over the past decade, Parkinson disease (PD) has evolved from a textbook example of a mostly nonhereditary disease to a complex disorder with a well-established genetic component in a considerable subset of patients
-
PD is clinically characterized by the cardinal features of bradykinesia, tremor, rigidity, postural instability, and responsiveness to dopaminergic therapy
-
Disease phenotypes associated with the PARK1–9 chromosomal loci follow a typical Mendelian pattern of inheritance, but PARK10 and PARK11 represent susceptibility loci with an as yet undefined mode of transmission
-
Monogenic PD accounts for a minority of cases; most of the other cases are probably caused by complex interactions between several genes, modifying effects of susceptibility alleles and epigenetic factors, and effects on PD-linked-gene expression that are attributable to environmental agents and aging
-
When considering genetic testing in routine PD practice, the academic curiosity of a knowledgeable practitioner has to be weighed against the actual benefit of genetic testing to the subject
-
The PD field now enjoys an embarrassment of riches in genetic leads that also represent a mandate for translational research success
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Litvan I et al. (2003) Movement Disorders Society Scientific Issues Committee report: SIC Task Force appraisal of clinical diagnostic criteria for Parkinsonian disorders. Mov Disord 18: 467–486
Jellinger K (2003) Synucleinopathies. In Neurodegeneration: The Molecular Pathology of Dementia and Movement Disorders, 155–225 (Ed Dickson D) Basel: ISN Neuropath Press
Lang AE and Lozano AM (1998) Parkinson's disease. First of two parts. N Engl J Med 339, 1044–1053
Lang AE and Lozano AM (1998) Parkinson's disease. Second of two parts. N Engl J Med 339: 1130–1143
Braak H et al. (2002) Staging of the intracerebral inclusion body pathology associated with idiopathic Parkinson's disease (preclinical and clinical stages). J Neurol 249 (Suppl 3): 1–5
Klein C et al. (2000) Parkin deletions in a family with adult-onset, tremor-dominant parkinsonism: expanding the phenotype. Ann Neurol 48: 65–71
Lohmann E et al. (2003) How much phenotypic variation can be attributed to parkin genotype? Ann Neurol 54: 176–185
Albanese A et al. (2005) The PINK1 phenotype can be indistinguishable from idiopathic Parkinson disease. Neurology 64: 1958–1960
Hedrich K et al. Clinical spectrum of homo- and heterozygous PINK1 mutations in a large German family with Parkinson's disease: role of a single hit? Arch Neurol, in press
Farrer M et al. (2001) Lewy bodies and parkinsonism in families with parkin mutations. Ann Neurol 50: 293–300
Wszolek ZK et al. (2004) Autosomal dominant parkinsonism associated with variable synuclein and tau pathology. Neurology 62: 1619–1622
Pramstaller PP et al. (2005) Lewy body Parkinson disease in a large pedigree with 77 Parkin mutation carriers. Ann Neurol 58: 411–422
Schlossmacher MG : Alpha-synuclein and synucleinopathies. In Blue Books of Practical Neurology: The Dementias II, Chapter 8 (Eds Growdon JH and Rossor M) Boston: Butterworth and Heinemann, in press
Klein C (2005) Movement disorders: classifications. J Inherit Metab Dis 28: 425–439
Kann M et al. (2002) Role of parkin mutations in 111 community-based patients with early-onset parkinsonism. Ann Neurol 51: 621–625
Kitada T et al. (1998) Mutations in the parkin gene cause autosomal recessive juvenile parkinsonism. Nature 392: 605–608
Hedrich K et al. (2004) Distribution, type, and origin of Parkin mutations: review and case studies. Mov Disord 19: 1146–1157
Hedrich K et al. (2001) The importance of gene dosage studies: mutational analysis of the parkin gene in early-onset parkinsonism. Hum Mol Genet 16: 1649–1656
Schlossmacher MG et al. (2002) Parkin localizes to the Lewy bodies of Parkinson disease and dementia with Lewy bodies. Am J Pathol 160: 1655–1667
Shimura H et al. (2000) Familial Parkinson disease gene product, parkin, is a ubiquitin-protein ligase. Nat Genet 25: 302–305
Sriram SR et al. (2005) Familial-associated mutations differentially disrupt the solubility, localization, binding and ubiquitination properties of parkin. Hum Mol Genet 14: 2571–2586
Cookson MR (2005) The biochemistry of Parkinson's disease. Annu Rev Biochem 74: 29–52
Goldberg MS et al. (2003) Parkin-deficient mice exhibit nigrostriatal deficits but not loss of dopaminergic neurons. J Biol Chem 278: 43628–43635
Palacino JJ et al. (2004) Mitochondrial dysfunction and oxidative damage in parkin-deficient mice. J Biol Chem 279: 18614–18622
Greene JC et al. (2005) Genetic and genomic studies of Drosophila parkin mutants implicate oxidative stress and innate immune responses in pathogenesis. Hum Mol Genet 14: 799–811
Feany MB and Bender WW (2000) A Drosophila model of Parkinson's disease. Nature 404: 394–398
LaVoie MJ et al. (2005) Dopamine covalently modifies and functionally inactivates parkin. Nat Med 11: 1214–1221
Valente EM et al. (2004) Hereditary early-onset Parkinson's disease caused by mutations in PINK1. Science 304: 1158–1160
Valente EM et al. (2004) PINK1 mutations are associated with sporadic early-onset parkinsonism. Ann Neurol 56: 336–341
Rogaeva E et al. (2004) Analysis of the PINK1 gene in a large cohort of cases with Parkinson disease. Arch Neurol 61: 1898–1904
Li Y et al. (2005) Clinicogenetic study of PINK1 mutations in autosomal recessive early-onset parkinsonism. Neurology 64: 1955–1957
Bonifati V et al. (2005) Early-onset parkinsonism associated with PINK1 mutations: frequency, genotypes, and phenotypes. Neurology 65: 87–95
Klein C et al. (2005) PINK1, Parkin, and DJ1 mutations in Italian patients with early-onset parkinsonism. Eur J Hum Genet 13: 1086–1093
Healy DG et al. (2004) PINK1 (PARK6) associated Parkinson disease in Ireland. Neurology 63: 1486–1488
Beilina A et al. (2005) Mutations in PTEN-induced putative kinase 1 associated with recessive parkinsonism have differential effects on protein stability. Proc Natl Acad Sci USA 102: 5703–5708
Silvestri L et al. (2005) Mitochondrial import and enzymatic activity of PINK1 mutants associated to recessive parkinsonism. Hum Mol Genet 14: 3477–3492
Bonifati V et al. (2003) Mutations in the DJ1 gene associated with autosomal recessive early-onset parkinsonism. Science 299: 256–259
Hedrich K et al. (2004) DJ1 (PARK7) mutations are less frequent than Parkin (PARK2) mutations in early-onset Parkinson disease. Neurology 62: 389–394
Canet-Aviles RM et al. (2004) The Parkinson's disease protein DJ1 is neuroprotective due to cysteine-sulfinic acid-driven mitochondrial localization. Proc Natl Acad Sci USA 101: 9103–9108
Kim RH et al. (2005) Hypersensitivity of DJ1-deficient mice to 1-methyl-4-phenyl-1,2,3,6-tetrahydropyrindine (MPTP) and oxidative stress. Proc Natl Acad Sci USA 102: 5215–5220
Goldberg MS et al. (2005) Nigrostriatal dopaminergic deficits and hypokinesia caused by inactivation of the familial Parkinsonism-linked gene DJ1. Neuron 45: 489–496
Polymeropoulos MH et al. (1997) Mutation in the α-synuclein gene identified in families with Parkinson's disease. Science 276: 2045–2047
Singleton AB et al. (2003) α-Synuclein locus triplication causes Parkinson's disease. Science 302: 841
Chartier-Harlin MC et al. (2004) α-synuclein locus duplication as a cause of familial Parkinson's disease. Lancet 364: 1167–1169
Berg D et al. (2005) Alpha-synuclein and Parkinson's disease: implications from the screening of more than 1,900 patients. Mov Disord 20: 1191–1194
Vekrellis K et al. (2004) Neurobiology of α-synuclein. Mol Neurobiol 30: 1–21
Trojanowski JQ and Lee VM (2002) Parkinson's disease and related synucleinopathies are a new class of nervous system amyloidoses. Neurotoxicology 23: 457–460
Schlossmacher MG et al. (2004) Case records of the Massachusetts General Hospital. Weekly clinicopathological exercises. Case 27-2004. A 79-year-old woman with disturbances in gait, cognition, and autonomic function. N Engl J Med 351: 912–922
Zimprich A et al. (2004) Mutations in LRRK2 cause autosomal-dominant parkinsonism with pleomorphic pathology. Neuron 44: 601–607
Paisan-Ruiz C et al. (2004) Cloning of the gene containing mutations that cause PARK8-linked Parkinson's disease. Neuron 44: 595–600
Funayama M et al. (2005) An LRRK2 mutation as a cause for the parkinsonism in the original PARK8 family. Ann Neurol 57: 918–921
Zabetian C et al. (2005) A clinic-based study of the LRRK2 gene in Parkinson disease yields new mutations. Neurology 65: 741–744
Farrer M et al. (2005) LRRK2 mutations in Parkinson disease. Neurology 65: 738–740
Paisan-Ruiz C et al. (2005) LRRK2 gene in Parkinson disease. Neurology 65: 696–700
Mata IF et al. (2005) LRRK2 pathogenic substitutions in Parkinson's disease. Neurogenetics 6: 171–177
Kachergus J et al. (2005) Identification of a novel LRRK2 mutation linked to autosomal dominant parkinsonism: evidence of a common founder across European populations. Am J Hum Genet 76: 672–680
Lesage S et al. (2005) LRRK2 haplotype analyses in European and North African families with Parkinson disease: a common founder for the G2019S mutation dating from the 13th century. Am J Hum Genet 77: 330–332
Li YJ et al. (2004) Apolipoprotein E controls the risk and age at onset of Parkinson disease. Neurology 62: 2005–2009
Lincoln SJ et al. (2003) Parkin variants in North American Parkinson's disease: cases and controls. Mov Disord 18: 1306–1311
Hilker R et al. (2001) Positron emission tomographic analysis of the nigrostriatal dopaminergic system in familial parkinsonism associated with mutations in the parkin gene. Ann Neurol 49: 367–376
Khan NL et al. (2002) Clinical and subclinical dopaminergic dysfunction in PARK6-linked parkinsonism: an 18F-dopa PET study. Ann Neurol 52: 849–853
Walter U et al. (2004) Brain parenchyma sonography detects preclinical parkinsonism. Mov Disord 19: 1445–1449
Buhmann C et al. (2005) Motor reorganization in asymptomatic carriers of a single mutant Parkin allele: a human model for presymptomatic parkinsonism. Brain 128: 2281–2290
Pankratz N et al. (2003) Genome-wide linkage analysis and evidence of gene-by-gene interactions in a sample of 362 multiplex Parkinson disease families. Hum Mol Genet 12: 2599–2608
Tan EK et al. (2000) Variability and validity of polymorphism association studies in Parkinson's disease. Neurology 55: 533–538
Skipper L et al. (2004) Linkage disequilibrium and association of MAPT H1 in Parkinson disease. Am J Hum Genet 75: 669–677
Eblan MJ et al. (2005) The glucocerebrosidase gene and Parkinson's disease in Ashkenazi Jews. N Engl J Med 352: 728–731
Liu Y et al. (2002) The UCH-L1 gene encodes two opposing enzymatic activities that affect α-synuclein degradation and Parkinson's disease susceptibility. Cell 111: 209–218
Maraganore DM et al. (2004) UCHL1 is a Parkinson's disease susceptibility gene. Ann Neurol 55: 512–521
Lucking CB et al. (2000) Association between early-onset Parkinson's disease and mutations in the parkin gene. French Parkinson's Disease Genetics Study Group. N Engl J Med 342: 1560–1567
McInerney-Leo A et al. (2005) Genetic testing in Parkinson's disease. Mov Disord 20: 1–10
Hornykiewicz O and Birkmayer W et al (1961) The effect of L-3,4-dihydroxyphenylalanine (=DOPA) on Parkinsonian akinesia [German]. Wien Klin Wochenschr 73: 787–788
el-Agnaf OMA et al Detection of alpha-synuclein oligomers in human plasma as a biomarker candidate for Parkinson disease. FASEB J, in press
Ozelius LJ et al. (2006) LRRK2 G2019S as a cause of Parkinson's disease in Ashkenazi Jews. N Engl J Med 354: 424–425
Lesage S et al. (2006) LRRK2 G2019S as a cause of Parkinson's disease in North African Arabs. N Engl J Med 354: 422–423
Acknowledgements
We thank our patients and their families for encouragement and support. The authors wish to express their gratitude to their colleagues M Farrer, A Lang and L Sudarsky for critical comments on earlier versions of the manuscript. CK received support through a Lichtenberg Grant from the Volkswagen Foundation, the Deutsche Forschungsgemeinschaft, and a Research Grant from the University of Lübeck; MGS received support from the National Institute of Neurological Disorders and Stroke/National Institutes of Health, Michael J Fox Foundation, and Multiple System Atrophy Fund at Brigham and Women's Hospital.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Klein, C., Schlossmacher, M. The genetics of Parkinson disease: implications for neurological care. Nat Rev Neurol 2, 136–146 (2006). https://doi.org/10.1038/ncpneuro0126
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ncpneuro0126
This article is cited by
-
Triple herbal extract DA-9805 exerts a neuroprotective effect via amelioration of mitochondrial damage in experimental models of Parkinson’s disease
Scientific Reports (2018)
-
A workflow for the integrative transcriptomic description of molecular pathology and the suggestion of normalizing compounds, exemplified by Parkinson’s disease
Scientific Reports (2018)
-
Short-term temporal memory in idiopathic and Parkin-associated Parkinson’s disease
Scientific Reports (2018)
-
Imaging genetics approach to Parkinson’s disease and its correlation with clinical score
Scientific Reports (2017)
-
Integrating Patient Concerns into Parkinson’s Disease Management
Current Neurology and Neuroscience Reports (2017)